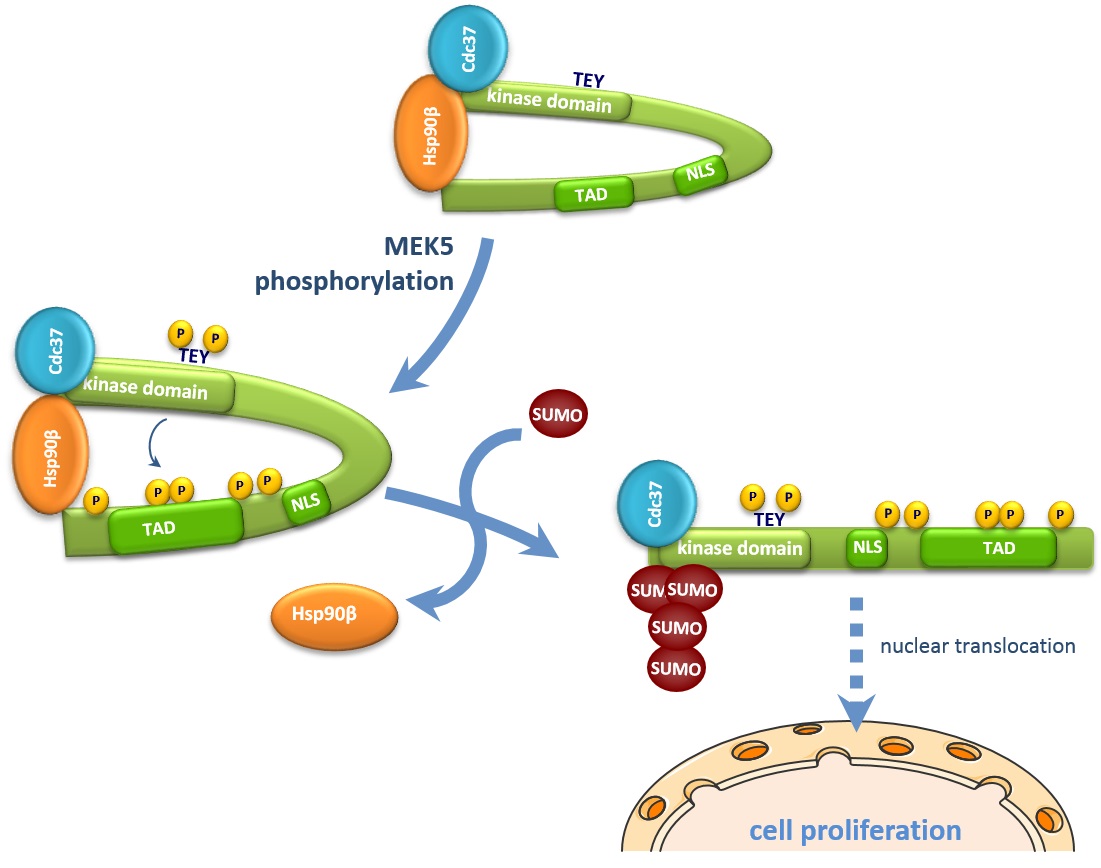
The MAP kinase ERK5 contains an N-terminal kinase domain and a unique C-terminal tail including a nuclear localization signal and a transcriptional activation domain. ERK5 is activated in response to growth factors and stresses, and regulates transcription at the nucleus by either phosphorylation or interaction with transcription factors. MEK5-ERK5 pathway plays an important role regulating cancer cell proliferation and survival. Therefore, it is important to define the precise molecular mechanisms implicated in ERK5 nucleo-cytoplasmic shuttling. We previously described that the molecular chaperone Hsp90 stabilizes and anchors ERK5 at the cytosol, and that ERK5 nuclear shuttling requires Hsp90 dissociation. Here, we show that MEK5 or Cdc37 overexpression -mechanisms that induce nuclear ERK5- induced ERK5 SUMO-2 modification at residues Lys6/Lys22 in cancer cells. We also show that overexpression of the SUMO protease SENP2 completely abolished endogenous ERK5 nuclear localization in response to EGF stimulation. Furthermore, mutation of these SUMO sites abolished the ability of ERK5 to translocate to the nucleus and to promote prostatic cancer PC-3 cell proliferation. These results allow us to propose a more precise mechanism: in response to MEK5 activation, ERK5 SUMOylation favors the dissociation of Hsp90 from the complex, allowing ERK5 nuclear shuttling and activation of transcription.