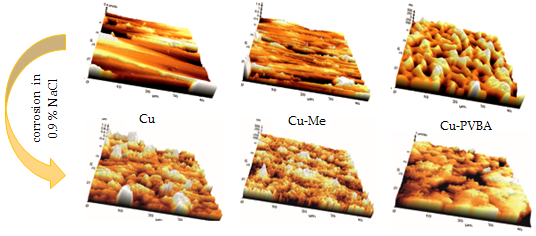
Poly(vinyl butyral-co-vinyl alcohol-co-vinyl acetate) named further PVBA was investigated as protective coating for copper corrosion in 0.9 % NaCl solution using electrochemical measurements such as, electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization associated with Atomic Force Microscopy (AFM). The PVBA coating on the copper surface (Cu-PVBA) was modeled in methanol containing PVBA. Its inhibitory properties against corrosion was comparatively discussed with those of the copper sample treated in methanol without polymer (Cu-Me) and of untreated sample (standard copper). A protective performance of PVBA coating of 80 % was computed from electrochemical measurements, for copper corrosion in NaCl solution. Also, AFM images designed a specific surface morphology of coated surface with PVBA, clearly highlighting a polymer film adsorbed on the copper surface, which presents certain deterioration after corrosion, but metal surface was not significantly affected compared to those of untreated samples or treated in methanol, in the absence of PVBA.