Submitted:
12 January 2023
Posted:
13 January 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
1.1. Experimental design
1.2. Time-of-day (circadian) effects
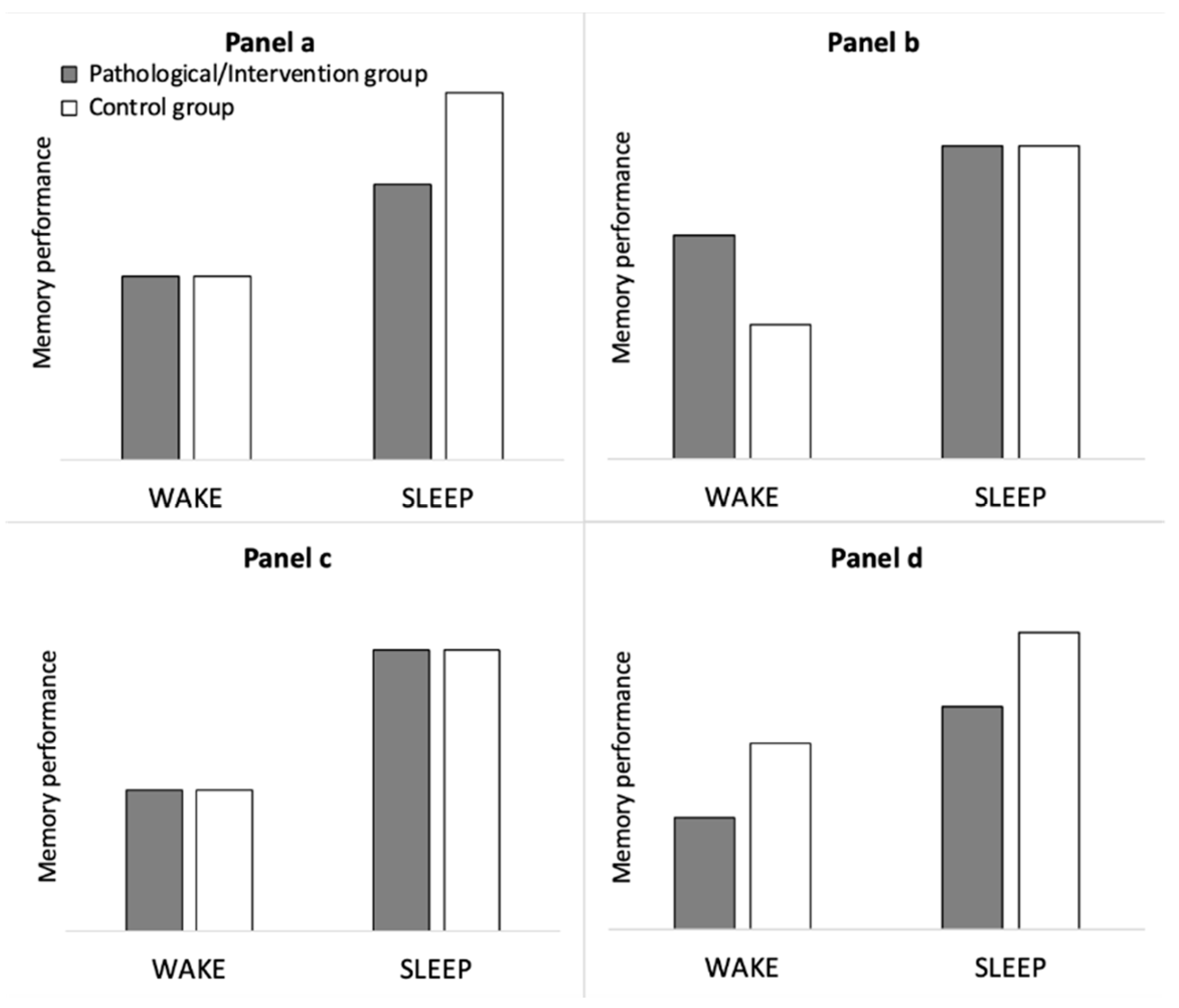
1.3. Controls in overnight studies
1.4. Control conditions in napping studies
1.5. Intervals between encoding and sleep
1.6. Baseline measures and feedback
2. Task complexity
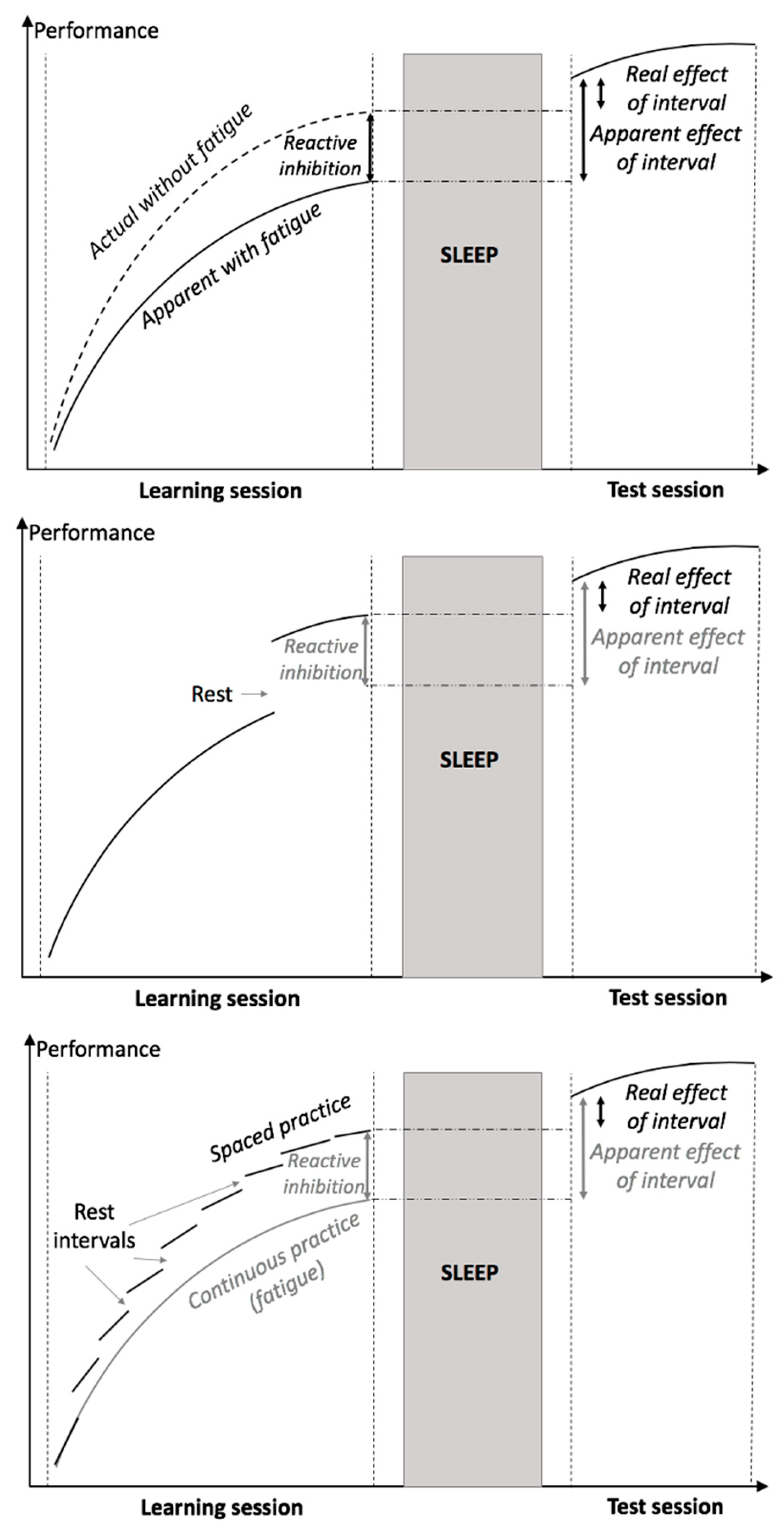
3. Fatigue effect in repetitive tasks
4. Data analysis practices
4.1. Sample size and result interpretation
4.2. Spurious correlations
4.3. Individual differences
5. Conclusion
5.1. Data deposition
5.2. Pre-registration
Acknowledgements
Competing Interests
References
- Palagini, L. & Rosenlicht, N. Sleep, dreaming, and mental health: a review of historical and neurobiological perspectives. Sleep Med. Rev. 15, 179-186 (2011). [CrossRef]
- Simor, P., Sifuentes-Ortega, R. & Peigneux, P. ESRS European sleep medicine textbook: chapter: A. 5. Sleep and psychology (cognitive and emotional processes). (2021).
- Walker, M. P. The role of sleep in cognition and emotion. Ann. N. Y. Acad. Sci. 1156, 168-197 (2009). [CrossRef]
- Shochat, T. Impact of lifestyle and technology developments on sleep. Nature and science of sleep 4, 19 (2012). [CrossRef]
- Bixler, E. Sleep and society: an epidemiological perspective. Sleep Med. 10, S3-S6 (2009). [CrossRef]
- Matricciani, L. et al. Past, present, and future: trends in sleep duration and implications for public health. Sleep health 3, 317-323 (2017). [CrossRef]
- Hale, L. & Guan, S. Screen time and sleep among school-aged children and adolescents: a systematic literature review. Sleep Med. Rev. 21, 50-58 (2015). [CrossRef]
- Ackermann, S. & Rasch, B. Differential effects of non-REM and REM sleep on memory consolidation? Curr. Neurol. Neurosci. Rep. 14, 430 (2014).
- Antony, J. W., Schönauer, M., Staresina, B. P. & Cairney, S. A. Sleep spindles and memory reprocessing. Trends Neurosci. 42, 1-3 (2019). [CrossRef]
- Boyce, R., Williams, S. & Adamantidis, A. REM sleep and memory. Curr. Opin. Neurobiol. 44, 167-177 (2017).
- Diekelmann, S. & Born, J. The memory function of sleep. Nat. Rev. Neurosci. 11, 114-126 (2010). [CrossRef]
- Diekelmann, S., Wilhelm, I. & Born, J. The whats and whens of sleep-dependent memory consolidation. Sleep Med. Rev. 13, 309-321 (2009).
- Feld, G. B. & Born, J. Sculpting memory during sleep: concurrent consolidation and forgetting. Curr. Opin. Neurobiol. 44, 20-27 (2017). [CrossRef]
- Lewis, P. A. & Durrant, S. J. Overlapping memory replay during sleep builds cognitive schemata. Trends in cognitive sciences 15, 343-351 (2011). [CrossRef]
- Mednick, S. C., Cai, D. J., Shuman, T., Anagnostaras, S. & Wixted, J. T. An opportunistic theory of cellular and systems consolidation. Trends Neurosci. 34, 504-514 (2011). [CrossRef]
- Saletin, J. M. & Walker, M. P. Nocturnal mnemonics: sleep and hippocampal memory processing. Front. Neurol. 3, 59 (2012). [CrossRef]
- Siegel, D. J. Memory: an overview, with emphasis on developmental, interpersonal, and neurobiological aspects. J. Am. Acad. Child Adolesc. Psychiatry 40, 997-1011 (2001). [CrossRef]
- Stickgold, R. & Walker, M. P. Memory consolidation and reconsolidation: what is the role of sleep? Trends Neurosci. 28, 408-415 (2005).
- Stickgold, R. & Walker, M. P. Sleep-dependent memory triage: Evolving generalization through selective processing. Nat. Neurosci. 16, 139-145 (2013). [CrossRef]
- Tononi, G. & Cirelli, C. Sleep function and synaptic homeostasis. Sleep Med. Rev. 10, 49-62, (2006). [CrossRef]
- Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12-34 (2014). [CrossRef]
- Walker, M. P. A refined model of sleep and the time course of memory formation. Behav. Brain Sci. 28, 51-104 (2005). [CrossRef]
- Schönauer, M., Grätsch, M. & Gais, S. Evidence for two distinct sleep-related long-term memory consolidation processes. Cortex 63, 68-78 (2015). [CrossRef]
- Krakauer, J. W., Ghazanfar, A. A., Gomez-Marin, A., MacIver, M. A. & Poeppel, D. Neuroscience needs behavior: correcting a reductionist bias. Neuron 93, 480-490 (2017). [CrossRef]
- Niv, Y. The primacy of behavioral research for understanding the brain. Behav. Neurosci. (2021). [CrossRef]
- Gais, S., Lucas, B. & Born, J. Sleep after learning aids memory recall. Learn. Mem. 13, 259-262 (2006). [CrossRef]
- Walker, M. P., Brakefield, T., Hobson, J. A. & Stickgold, R. Dissociable stages of human memory consolidation and reconsolidation. Nature 425, 616-620 (2003). [CrossRef]
- King, B. R., Hoedlmoser, K., Hirschauer, F., Dolfen, N. & Albouy, G. Sleeping on the motor engram: the multifaceted nature of sleep-related motor memory consolidation. Neurosci. Biobehav. Rev. 80, 1-22 (2017). [CrossRef]
- Rasch, B. & Born, J. About sleep's role in memory. Physiol. Rev. 93, 681-766, (2013). [CrossRef]
- Born, J. & Wilhelm, I. System consolidation of memory during sleep. Psychol. Res. 76, 192-203 (2012). [CrossRef]
- Vorster, A. P. & Born, J. Sleep and memory in mammals, birds and invertebrates. Neurosci. Biobehav. Rev. 50, 103-119 (2015). [CrossRef]
- Pan, S. C. & Rickard, T. C. Sleep and motor learning: is there room for consolidation? Psychol. Bull. 141, 812 (2015). [CrossRef]
- Mantua, J. Sleep Physiology Correlations and Human Memory Consolidation: Where Do We Go From Here? Sleep 41, zsx204 (2018). [CrossRef]
- Vertes, R. P. & Siegel, J. M. Time for the sleep community to take a critical look at the purported role of sleep in memory processing. Sleep 28, 1228-1229 (2005). [CrossRef]
- Dastgheib, M., Kulanayagam, A. & Dringenberg, H. C. Is the role of sleep in memory consolidation overrated? Neurosci. Biobehav. Rev., 104799 (2022).
- Cordi, M. J. & Rasch, B. How robust are sleep-mediated memory benefits? Curr. Opin. Neurobiol. 67, 1-7 (2021).
- Voderholzer, U. et al. Sleep restriction over several days does not affect long-term recall of declarative and procedural memories in adolescents. Sleep Med. 12, 170-178 (2011). [CrossRef]
- Robertson, E. M., Pascual-Leone, A. & Press, D. Z. Awareness modifies the skill-learning benefits of sleep. Curr. Biol. 14, 208-212, (2004). [CrossRef]
- Song, S., Howard, J. H., Jr. & Howard, D. V. Sleep does not benefit probabilistic motor sequence learning. J. Neurosci. 27, 12475-12483, (2007). [CrossRef]
- Viczko, J., Sergeeva, V., Ray, L. B., Owen, A. M. & Fogel, S. M. Does sleep facilitate the consolidation of allocentric or egocentric representations of implicitly learned visual-motor sequence learning? Learn. Mem. 25, 67-77 (2018). [CrossRef]
- Wilson, J. K., Baran, B., Pace-Schott, E. F., Ivry, R. B. & Spencer, R. Sleep modulates word-pair learning but not motor sequence learning in healthy older adults. Neurobiol. Aging 33, 991-100 (2012). [CrossRef]
- Nemeth, D. et al. Sleep has no critical role in implicit motor sequence learning in young and old adults. Exp. Brain Res. 201, 351-358, (2010). [CrossRef]
- Csabi, E., Varszegi-Schulz, M., Janacsek, K., Malecek, N. & Nemeth, D. The consolidation of implicit sequence memory in obstructive sleep apnea. PLoS One 9, e109010 (2014). [CrossRef]
- Ujma, P. P. Meta-analytic evidence suggests no correlation between sleep spindles and memory. (2022). [CrossRef]
- Payne, J. D. et al. The role of sleep in false memory formation. Neurobiol Learn Mem 92, 327-334 (2009). [CrossRef]
- Peigneux, P. & Smith, C. Memory processing in relation to sleep. Principles and practice of sleep medicine 5 (2010).
- Ellenbogen, J. M., Payne, J. D. & Stickgold, R. The role of sleep in declarative memory consolidation: passive, permissive, active or none? Curr. Opin. Neurobiol. 16, 716-722 (2006). [CrossRef]
- Schmidt, C., Collette, F., Cajochen, C. & Peigneux, P. A time to think: circadian rhythms in human cognition. Cogn. Neuropsychol. 24, 755-789 (2007). [CrossRef]
- Barner, C., Schmid, S. R. & Diekelmann, S. Time-of-day effects on prospective memory. Behav. Brain Res. 376, 112179 (2019). [CrossRef]
- Scullin, M. K. & McDaniel, M. A. Remembering to execute a goal: sleep on it! Psychol. Sci. 21, 1028-1035 (2010).
- Keisler, A., Ashe, J. & Willingham, D. T. Time of day accounts for overnight improvement in sequence learning. Learn. Mem. 14, 669-672 (2007). [CrossRef]
- Taillard, J., Gronfier, C., Bioulac, S., Philip, P. & Sagaspe, P. (s Note: MDPI stays neutral with regard to jurisdictional claims in published …, 2021).
- Dresler, M. et al. Neuroscience-driven discovery and development of sleep therapeutics. Pharmacology & therapeutics 141, 300-334 (2014). [CrossRef]
- Genzel, L. & Robertson, E. M. To Replay, Perchance to Consolidate. PLoS Biol. 13, e1002285. (2015). [CrossRef]
- Tucker, M. et al. The relative impact of sleep and circadian drive on motor skill acquisition and memory consolidation. Sleep 40, zsx036 (2017). [CrossRef]
- Fenn, K. M., Nusbaum, H. C. & Margoliash, D. Consolidation during sleep of perceptual learning of spoken language. Nature 425, 614-616 (2003). [CrossRef]
- Hallgato, E., Győri-Dani, D., Pekár, J., Janacsek, K. & Nemeth, D. The differential consolidation of perceptual and motor learning in skill acquisition. Cortex 49, 1073-1081 (2013). [CrossRef]
- Talamini, L. M., Nieuwenhuis, I. L., Takashima, A. & Jensen, O. Sleep directly following learning benefits consolidation of spatial associative memory. Learn. Mem. 15, 233-237 (2008). [CrossRef]
- Tucker, M., McKinley, S. & Stickgold, R. Sleep optimizes motor skill in older adults. J. Am. Geriatr. Soc. 59, 603-609 (2011). [CrossRef]
- Gais, S., Rasch, B., Dahmen, J. C., Sara, S. & Born, J. The memory function of noradrenergic activity in non-REM sleep. J Cogn Neurosci 23, 2582-2592. (2011). [CrossRef]
- Mednick, S. C., Nakayama, K. & Stickgold, R. Sleep-dependent learning: a nap is as good as a night. Nat Neurosci 6, 697-698. (2003). [CrossRef]
- Payne, J. D. et al. Napping and the selective consolidation of negative aspects of scenes. Emotion 15, 176 (2015). [CrossRef]
- Backhaus, J. et al. Impaired declarative memory consolidation during sleep in patients with primary insomnia: influence of sleep architecture and nocturnal cortisol release. Biol. Psychiatry 60, 1324-1330 (2006). [CrossRef]
- Ahuja, S. et al. Role of normal sleep and sleep apnea in human memory processing. Nature and science of sleep 10, 255 (2018). [CrossRef]
- Cellini, N. Memory consolidation in sleep disorders. Sleep Med. Rev. 35, 101-112 (2017). [CrossRef]
- Wallace, A. & Bucks, R. S. Memory and obstructive sleep apnea: a meta-analysis. Sleep 36, 203-220 (2013). [CrossRef]
- Csábi, E., Benedek, P., Janacsek, K., Katona, G. & Nemeth, D. Sleep disorder in childhood impairs declarative but not nondeclarative forms of learning. J. Clin. Exp. Neuropsychol. 35, 677-685 (2013). [CrossRef]
- Rosenzweig, I. et al. Sleep apnoea and the brain: a complex relationship. The Lancet Respiratory Medicine 3, 404-414 (2015). [CrossRef]
- Wulff, K., Gatti, S., Wettstein, J. G. & Foster, R. G. Sleep and circadian rhythm disruption in psychiatric and neurodegenerative disease. Nat. Rev. Neurosci. 11, 589-599 (2010). [CrossRef]
- Li, J., Vitiello, M. V. & Gooneratne, N. S. Sleep in normal aging. Sleep Med. Clin. 13, 1-11 (2018). [CrossRef]
- Mander, B. A., Winer, J. R. & Walker, M. P. Sleep and human aging. Neuron 94, 19-36 (2017). [CrossRef]
- Nissen, C. et al. Sleep-related memory consolidation in primary insomnia. J. Sleep Res. 20, 129-136 (2011). [CrossRef]
- Lahl, O., Wispel, C., Willigens, B. & Pietrowsky, R. An ultra short episode of sleep is sufficient to promote declarative memory performance. J Sleep Res 17, 3-10. (2008). [CrossRef]
- Diekelmann, S., Biggel, S., Rasch, B. & Born, J. Offline consolidation of memory varies with time in slow wave sleep and can be accelerated by cuing memory reactivations. Neurobiol. Learn. Mem. 98, 103-111 (2012). [CrossRef]
- Nishida, M. & Walker, M. P. Daytime naps, motor memory consolidation and regionally specific sleep spindles. PLoS One 2, e341 (2007). [CrossRef]
- Schönauer, M., Geisler, T. & Gais, S. Strengthening procedural memories by reactivation in sleep. J. Cogn. Neurosci. 26, 143-153 (2014). [CrossRef]
- Gais, S., Plihal, W., Wagner, U. & Born, J. Early sleep triggers memory for early visual discrimination skills. Nat Neurosci 3, 1335-1339. (2000). [CrossRef]
- Plihal, W. & Born, J. Effects of early and late nocturnal sleep on declarative and procedural memory. J Cogn Neurosci 9, 534-547. (1997). [CrossRef]
- Mednick, S. C., Mednick, S. C., Cai, D. J., Kanady, J. & Drummond, S. P. A. Comparing the benefits of caffeine, naps and placebo on verbal, motor and perceptual memory. Behav. Brain Res. 193, 79 (2008). [CrossRef]
- Sopp, M. R., Michael, T. & Mecklinger, A. Effects of early morning nap sleep on associative memory for neutral and emotional stimuli. Brain Res 1698, 29-42. (2018). [CrossRef]
- Plihal, W., Weaver, S., Molle, M., Fehm, H. L. & Born, J. Sensory processing during early and late nocturnal sleep. Electroencephalogr Clin Neurophysiol 99, 247-256. (1996). [CrossRef]
- Yaroush, R., Sullivan, M. J. & Ekstrand, B. R. Effect of sleep on memory. II. Differential effect of the first and second half of the night. J Exp Psychol 88, 361-366. (1971). [CrossRef]
- Wamsley, E. J. Memory consolidation during waking rest. Trends in cognitive sciences 23, 171-173 (2019). [CrossRef]
- Cousins, J. N., Wong, K. F., Raghunath, B. L., Look, C. & Chee, M. W. The long-term memory benefits of a daytime nap compared with cramming. Sleep 42, zsy207 (2019). [CrossRef]
- McDevitt, E. A., Duggan, K. A. & Mednick, S. C. REM sleep rescues learning from interference. Neurobiol Learn Mem 122, 51-62. (2015). [CrossRef]
- Piosczyk, H. et al. The effect of sleep-specific brain activity versus reduced stimulus interference on declarative memory consolidation. J. Sleep Res. 22, 406-413 (2013). [CrossRef]
- Schichl, M., Ziberi, M., Lahl, O. & Pietrowsky, R. The influence of midday naps and relaxation-hypnosis on declarative and procedural memory performance. Sleep and Hypnosis 13, 7-14 (2011).
- Mednick, S. C., Makovski, T., Cai, D. & Jiang, Y. V. Sleep and rest facilitate implicit memory in a visual search task. Vision Res. 49, 2557-2565 (2009). [CrossRef]
- Simor, P. et al. Deconstructing procedural memory: Different learning trajectories and consolidation of sequence and statistical learning. Front. Psychol. 9, 2708 (2019). [CrossRef]
- Schapiro, A. C., McDevitt, E. A., Rogers, T. T., Mednick, S. C. & Norman, K. A. Human hippocampal replay during rest prioritizes weakly learned information and predicts memory performance. Nature communications 9, 3920 (2018). [CrossRef]
- den Berg van, N. H. et al. Sleep Enhances Consolidation of Memory Traces for Complex Problem-Solving Skills. Cereb. Cortex, (2021). [CrossRef]
- Van den Berg, N. et al. Sleep preferentially enhances memory for a cognitive strategy but not the implicit motor skills used to acquire it. Neurobiol. Learn. Mem. 161, 135-142 (2019). [CrossRef]
- Mikutta, C. et al. Phase-amplitude coupling of sleep slow oscillatory and spindle activity correlates with overnight memory consolidation. J. Sleep Res.. (2019). [CrossRef]
- Cordi, M. J., Diekelmann, S., Born, J. & Rasch, B. No effect of odor-induced memory reactivation during REM sleep on declarative memory stability. Frontiers in systems neuroscience 8, 157. (2014). [CrossRef]
- Prehn-Kristensen, A. et al. Transcranial Oscillatory Direct Current Stimulation During Sleep Improves Declarative Memory Consolidation in Children With Attention-deficit/hyperactivity Disorder to a Level Comparable to Healthy Controls. Brain stimulation 7, 793-799. (2014). [CrossRef]
- Xue, G. et al. Greater neural pattern similarity across repetitions is associated with better memory. Science 330, 97-101. (2010). [CrossRef]
- Young, D. R. & Bellezza, F. S. Encoding variability, memory organization, and the repetition effect. Journal of Experimental Psychology: Learning, Memory, and Cognition 8, 545-559 (1982).
- Ebbinghaus, H. Memory: a contribution to experimental psychology. Annals of neurosciences 20, 155-156. (2013). [CrossRef]
- Chan, J. C. & McDermott, K. B. The testing effect in recognition memory: a dual process account. J. Exp. Psychol. Learn. Mem. Cogn. 33, 431-437. (2007). [CrossRef]
- Soderstrom, N. C., Kerr, T. K. & Bjork, R. A. The Critical Importance of Retrieval--and Spacing--for Learning. Psychol Sci 27, 223-230 (2016). [CrossRef]
- Karpicke, J. D. & Roediger, H. L., 3rd. The critical importance of retrieval for learning. Science 319, 966-968. (2008). [CrossRef]
- Wiklund-Hornqvist, C., Jonsson, B. & Nyberg, L. Strengthening concept learning by repeated testing. Scand. J. Psychol. 55, 10-16. (2014). [CrossRef]
- Klinzing, J. G., Rasch, B., Born, J. & Diekelmann, S. Sleep's role in the reconsolidation of declarative memories. Neurobiol Learn Mem 136, 166-173. (2016). [CrossRef]
- Fowler, M. J., Sullivan, M. J. & Ekstrand, B. R. Sleep and memory. Science 179, 302-304. (1973). [CrossRef]
- Fenn, K. M. & Hambrick, D. Z. Individual differences in working memory capacity predict sleep-dependent memory consolidation. J. Exp. Psychol. Gen. 141, 404-410. (2012). [CrossRef]
- Wilhelm, I., Diekelmann, S. & Born, J. Sleep in children improves memory performance on declarative but not procedural tasks. Learn Mem 15, 373-377 (2008). [CrossRef]
- Marshall, L., Mölle, M., Hallschmid, M. & Born, J. Transcranial Direct Current Stimulation during Sleep Improves Declarative Memory. J. Neurosci. 24, 9985-9992. (2004). [CrossRef]
- Payne, J. D. et al. Memory for semantically related and unrelated declarative information: the benefit of sleep, the cost of wake. PLoS One 7, e33079. (2012). [CrossRef]
- Rasch, B., Buchel, C., Gais, S. & Born, J. Odor cues during slow-wave sleep prompt declarative memory consolidation. Science 315, 1426-1429 (2007). [CrossRef]
- Buschke, H. Selective reminding for analysis of memory and learning. Journal of Verbal Learning and Verbal Behavior 12, 543-550 (1973). [CrossRef]
- Quan, S. F., Budhiraja, R. & Kushida, C. A. Associations Between Sleep Quality, Sleep Architecture and Sleep Disordered Breathing and Memory After Continuous Positive Airway Pressure in Patients with Obstructive Sleep Apnea in the Apnea Positive Pressure Long-term Efficacy Study (APPLES). Sleep science (Sao Paulo, Brazil) 11, 231-238. (2018). [CrossRef]
- Uguccioni, G. et al. Sleep-related declarative memory consolidation and verbal replay during sleep talking in patients with REM sleep behavior disorder. PLoS One 8, e83352. (2013). [CrossRef]
- Mazza, S. et al. Relearn Faster and Retain Longer. Psychol Sci 27, 1321-1330. (2016). [CrossRef]
- Roediger III, H. L. & Karpicke, J. D. The power of testing: basic research and implications for educational practice. Perspect. Psychol. Sci. 1, 181-210 (2006). [CrossRef]
- McDermott, K. B. Practicing Retrieval Facilitates Learning. Annu Rev Psychol 72, 609-633, (2021). [CrossRef]
- Jacoby, L. L. A process dissociation framework: Separating automatic from intentional uses of memory. JMemL 30, 513-541 (1991). [CrossRef]
- Sigman, M. & Dehaene, S. Parsing a cognitive task: a characterization of the mind's bottleneck. PLoS Biol. 3, e37 (2005).
- Cohen, D. A., Pascual-Leone, A., Press, D. Z. & Robertson, E. M. Off-line learning of motor skill memory: A double dissociation of goal and movement. PNAS 102, 18237-18241 (2005). [CrossRef]
- Conte, F. & Ficca, G. Caveats on psychological models of sleep and memory: a compass in an overgrown scenario. Sleep Med. Rev. 17, 105-121 (2013). [CrossRef]
- Stickgold, R. Parsing the role of sleep in memory processing. Curr. Opin. Neurobiol. 23, 847-853 (2013). [CrossRef]
- Schendan, H. E., Searl, M., Melrose, R. & Stern, C. An FMRI study of the role of the medial temporal lobe in implicit and explicit sequence learning. Neuron 37, 1013-1025 (2003). [CrossRef]
- Fischer, S., Drosopoulos, S., Tsen, J. & Born, J. Implicit Learning-Explicit Knowing: A Role for Sleep in Memory System Interaction. J. Cogn. Neurosci. 18, 311 (2006). [CrossRef]
- Freedberg, M., Toader, A. C., Wassermann, E. M. & Voss, J. L. Competitive and cooperative interactions between medial temporal and striatal learning systems. Neuropsychologia 136, 107257, (2020). [CrossRef]
- Brown, R. M. & Robertson, E. M. Off-line processing: Reciprocal interactions between declarative and procedural memories. J. Neurosci. 27, 10468-10475 (2007). [CrossRef]
- Feld, G. B., Weis, P. P. & Born, J. The Limited Capacity of Sleep-Dependent Memory Consolidation. Front Psychol 7, 1368, (2016). [CrossRef]
- Diekelmann, S., Born, J. & Wagner, U. Sleep enhances false memories depending on general memory performance. Behav Brain Res 208, 425-429, (2010). [CrossRef]
- Abel, M. & Bauml, K. H. Retrieval-induced forgetting, delay, and sleep. Memory 20, 420-428, (2012). [CrossRef]
- Cairney, S. A., Durrant, S. J., Power, R. & Lewis, P. A. Complementary roles of slow-wave sleep and rapid eye movement sleep in emotional memory consolidation. Cereb Cortex 25, 1565-1575, (2015). [CrossRef]
- Rudoy, J. D., Voss, J. L., Westerberg, C. E. & Paller, K. A. Strengthening individual memories by reactivating them during sleep. Science 326, 1079-1079 (2009). [CrossRef]
- Song, S. & Cohen, L. G. Practice and sleep form different aspects of skill. Nature communications 5, 3407 (2014). [CrossRef]
- Albouy, G. et al. Daytime sleep enhances consolidation of the spatial but not motoric representation of motor sequence memory. PLoS One 8, e52805 (2013). [CrossRef]
- Nissen, M. J. & Bullemer, P. Attentional requirements of learning: Evidence from performance measures. Cogn. Psychol. 19, 1-32 (1987). [CrossRef]
- Brawn, T. P., Fenn, K. M., Nusbaum, H. C. & Margoliash, D. Consolidating the effects of waking and sleep on motor-sequence learning. The Journal of neuroscience 30, 13977-13982 (2010). [CrossRef]
- Rickard, T. C., Cai, D. J., Rieth, C. A., Jones, J. & Ard, M. C. Sleep does not enhance motor sequence learning. Journal of Experimental Psychology: Learning, Memory, and Cognition; Journal of Experimental Psychology: Learning, Memory, and Cognition 34, 834 (2008).
- Cai, D. J. & Rickard, T. C. Reconsidering the role of sleep for motor memory. Behav. Neurosci. 123, 1153 (2009). [CrossRef]
- Nettersheim, A., Hallschmid, M., Born, J. & Diekelmann, S. The role of sleep in motor sequence consolidation: stabilization rather than enhancement. J. Neurosci. 35, 6696-6702 (2015). [CrossRef]
- Rieth, C. A., Cai, D. J., McDevitt, E. A. & Mednick, S. C. The role of sleep and practice in implicit and explicit motor learning. Behav. Brain Res. 214, 470-474 (2010). [CrossRef]
- Török, B., Janacsek, K., Nagy, D. G., Orbán, G. & Nemeth, D. Measuring and filtering reactive inhibition is essential for assessing serial decision making and learning. J. Exp. Psychol. Gen. 146, 529 (2017). [CrossRef]
- Ioannidis, J. P. Why most published research findings are false. PLoS Med. 2, e124 (2005). [CrossRef]
- Maxwell, S. E., Lau, M. Y. & Howard, G. S. Is psychology suffering from a replication crisis? What does “failure to replicate” really mean? American Psychologist 70, 487 (2015). [CrossRef]
- OpenScienceCollaboration. Estimating the reproducibility of psychological science. Science 349 (2015).
- Rickard, T. C., Pan, S. C. & Gupta, M. W. Severe Publication Bias Contributes to Illusory Sleep Consolidation in the Motor Sequence Learning Literature. Journal of Experimental Psychology: Learning, Memory, and Cognition (in press). [CrossRef]
- Wagner, U., Hallschmid, M., Rasch, B. & Born, J. Brief sleep after learning keeps emotional memories alive for years. Biol. Psychiatry 60, 788-790 (2006). [CrossRef]
- Csabi, E. et al. Declarative and Non-declarative Memory Consolidation in Children with Sleep Disorder. Front. Hum. Neurosci. 9, 709, (2015). [CrossRef]
- Association, A. P. Publication Manual of the American Psychological Association, (2020). (American Psychological Association, 2019).
- Dienes, Z. How Bayes factors change scientific practice. J. Math. Psychol. 72, 78-89 (2016). [CrossRef]
- Dienes, Z., Coulton, S. & Heather, N. Using Bayes factors to evaluate evidence for no effect: examples from the SIPS project. Addiction 113, 240-246 (2018). [CrossRef]
- Dienes, Z. & Mclatchie, N. Four reasons to prefer Bayesian analyses over significance testing. Psychon. Bull. Rev. 25, 207-218 (2018). [CrossRef]
- Wagenmakers, E.-J. et al. Bayesian inference for psychology. Part I: Theoretical advantages and practical ramifications. Psychon. Bull. Rev. 25, 35-57 (2018). [CrossRef]
- Rouder, J. N. Optional stopping: No problem for Bayesians. Psychon. Bull. Rev. 21, 301-308 (2014). [CrossRef]
- Schönbrodt, F. D., Wagenmakers, E.-J., Zehetleitner, M. & Perugini, M. Sequential hypothesis testing with Bayes factors: Efficiently testing mean differences. Psychol. Methods 22, 322 (2017). [CrossRef]
- Brown, H. & Maylor, E. A. Memory consolidation effects on memory stabilization and item integration in older adults. Psychon. Bull. Rev. 24, 1032-1039 (2017). [CrossRef]
- Rouder, J. N., Speckman, P. L., Sun, D., Morey, R. D. & Iverson, G. Bayesian t tests for accepting and rejecting the null hypothesis. Psychon. Bull. Rev. 16, 225-237, (2009). [CrossRef]
- Jarosz, A. F. & Wiley, J. What are the odds? A practical guide to computing and reporting Bayes factors. The Journal of Problem Solving 7, 2 (2014). [CrossRef]
- Ellis, P. D. The essential guide to effect sizes: Statistical power, meta-analysis, and the interpretation of research results. (Cambridge university press, 2010).
- Scullin, M. K. Sleep, memory, and aging: the link between slow-wave sleep and episodic memory changes from younger to older adults. Psychol. Aging 28, 105 (2013). [CrossRef]
- Ujma, P. P. Sleep spindles and general cognitive ability–A meta-analysis. Sleep Spindles & Cortical Up States 2, 1-17 (2021). [CrossRef]
- Ackermann, S., Hartmann, F., Papassotiropoulos, A., de Quervain, D. J. & Rasch, B. No associations between interindividual differences in sleep parameters and episodic memory consolidation. Sleep 38, 951-959 (2015). [CrossRef]
- Abdi, H. Bonferroni and Šidák corrections for multiple comparisons. Encyclopedia of measurement and statistics 3, 103-107 (2007).
- Forstmeier, W., Wagenmakers, E. J. & Parker, T. H. Detecting and avoiding likely false-positive findings–a practical guide. Biological Reviews 92, 1941-1968 (2017). [CrossRef]
- Bódizs, R. et al. Prediction of general mental ability based on neural oscillation measures of sleep. J. Sleep Res. 14, 285-292, (2005). [CrossRef]
- Fang, Z. et al. Sleep Spindles and Intellectual Ability: Epiphenomenon or Directly Related? J Cogn Neurosci 29, 167-182, (2017). [CrossRef]
- Fang, Z., Ray, L. B., Owen, A. M. & Fogel, S. M. Brain Activation Time-Locked to Sleep Spindles Associated With Human Cognitive Abilities. Front Neurosci 13, 46, (2019). [CrossRef]
- Fogel, S. M. & Smith, C. T. Learning-dependent changes in sleep spindles and Stage 2 sleep. J. Sleep Res. 15, 250-255 (2006). [CrossRef]
- Ujma, P. P. et al. A comparison of two sleep spindle detection methods based on all night averages: individually adjusted vs. fixed frequencies. Front. Hum. Neurosci. 9, 52 (2015).
- Poldrack, R. A. & Gorgolewski, K. J. OpenfMRI: Open sharing of task fMRI data. Neuroimage 144, 259-261 (2017). [CrossRef]
- Gorgolewski, K., Esteban, O., Schaefer, G., Wandell, B. & Poldrack, R. OpenNeuro—a free online platform for sharing and analysis of neuroimaging data. Organization for Human Brain Mapping. Vancouver, Canada 1677 (2017).
- Simor, P., Gombos, F., Blaskovich, B. & Bódizs, R. Long-range alpha and beta and short-range gamma EEG synchronization distinguishes phasic and tonic REM periods. Sleep 41, zsx210 (2017). [CrossRef]
- Simor, P., van Der Wijk, G., Gombos, F. & Kovács, I. The paradox of rapid eye movement sleep in the lights of oscillatory activity and cortical synchronization during phasic and tonic microstates. Neuroimage, 116066 (2019). [CrossRef]
- Nosek, B. A. et al. Promoting an open research culture. Science 348, 1422-1425 (2015). [CrossRef]
- Foster, E. D. & Deardorff, A. Open science framework (OSF). Journal of the Medical Library Association: JMLA 105, 203 (2017). [CrossRef]
- Nosek, B. A., Ebersole, C. R., DeHaven, A. C. & Mellor, D. T. The preregistration revolution. Proceedings of the National Academy of Sciences 115, 2600-2606 (2018). [CrossRef]
- Lindsay, D. S., Simons, D. J. & Lilienfeld, S. O. Research preregistration 101. APS observer 29 (2016).
- Hardwicke, T. E. & Wagenmakers, E.-J. Preregistration: A pragmatic tool to reduce bias and calibrate confidence in scientific research. (2021).
- DeAngelis, C. D. et al. Clinical trial registration: a statement from the International Committee of Medical Journal Editors. Arch. Dermatol. 141, 76-77 (2005). [CrossRef]
- Soderberg, C. K. et al. Initial evidence of research quality of registered reports compared with the standard publishing model. Nature Human Behaviour 5, 990-997 (2021). [CrossRef]

| Wider issues | Specific issues | Solutions |
|---|---|---|
| Non-optimal experimental designs may lead to inaccurate conclusions due to confounding variables | Time-of-day (circadian) effects | Using multiple control conditions: morning-evening; evening-morning; morning alone; evening alone; evening-morning and evening-evening with sleep deprivation (with and without recovery sleep). Napping conditions. Assessing sleepiness and vigilance. |
| Overnight studies (pathological population or intervention) | Including a morning-evening condition and/or an evening-morning deprivation condition. Including a control group. |
|
| Napping studies | Considering time of day and duration of nap. Including a quiet-wake control condition. Monitoring the nap with polysomnography. |
|
| Time interval between memory encoding and sleep onset | Controlling for duration of and participant’s activity during the interval between end of task and sleep. Monitoring the activities during the interval with actigraphy and/or questionnaires. |
|
| Baseline measurements and feedback effects (declarative memory) | Using a selective reminding procedure, possibly combined with a predefined learning criterion. | |
| Task complexity. Every task involves multiple cognitive processes that need to be disentangled to better understand the specific effect of sleep | – | Disentangling and contrasting different cognitive processes and aspects of memory involved in a given task. |
| Fatigue effect in repetitive non-declarative memory tasks may lead to a spurious beneficial effect of sleep by negatively affecting performance after a longer practice | – | Using appropriate experimental designs, e.g., including post-rest performance at the end of the training session as a baseline, and promoting learning through spaced rather than massed practice. Using appropriate data analysis methods, such as curve fitting and computational modeling. |
| Inappropriate data analysis practices, including use of small sample sizes and inappropriate analyses/reporting, may lead to spurious correlations and incorrect conclusions. | Small sample size and reporting only significant results | Determining the required sample size a priori. Using Bayesian analyses to decide when to stop data collection. Reporting Bayes Factors and effect sizes. |
| Spurious correlations between sleep parameters and memory consolidation | Planning correlation analyses of interest in advance, correcting for multiple comparisons, and reporting non-significant planned comparisons. | |
| Not controlling for individual differences in general cognitive abilities in correlational studies of sleep and memory | Including neurocognitive assessments of general cognitive abilities as covariates. Including a baseline night of sleep with an appropriate control task. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




