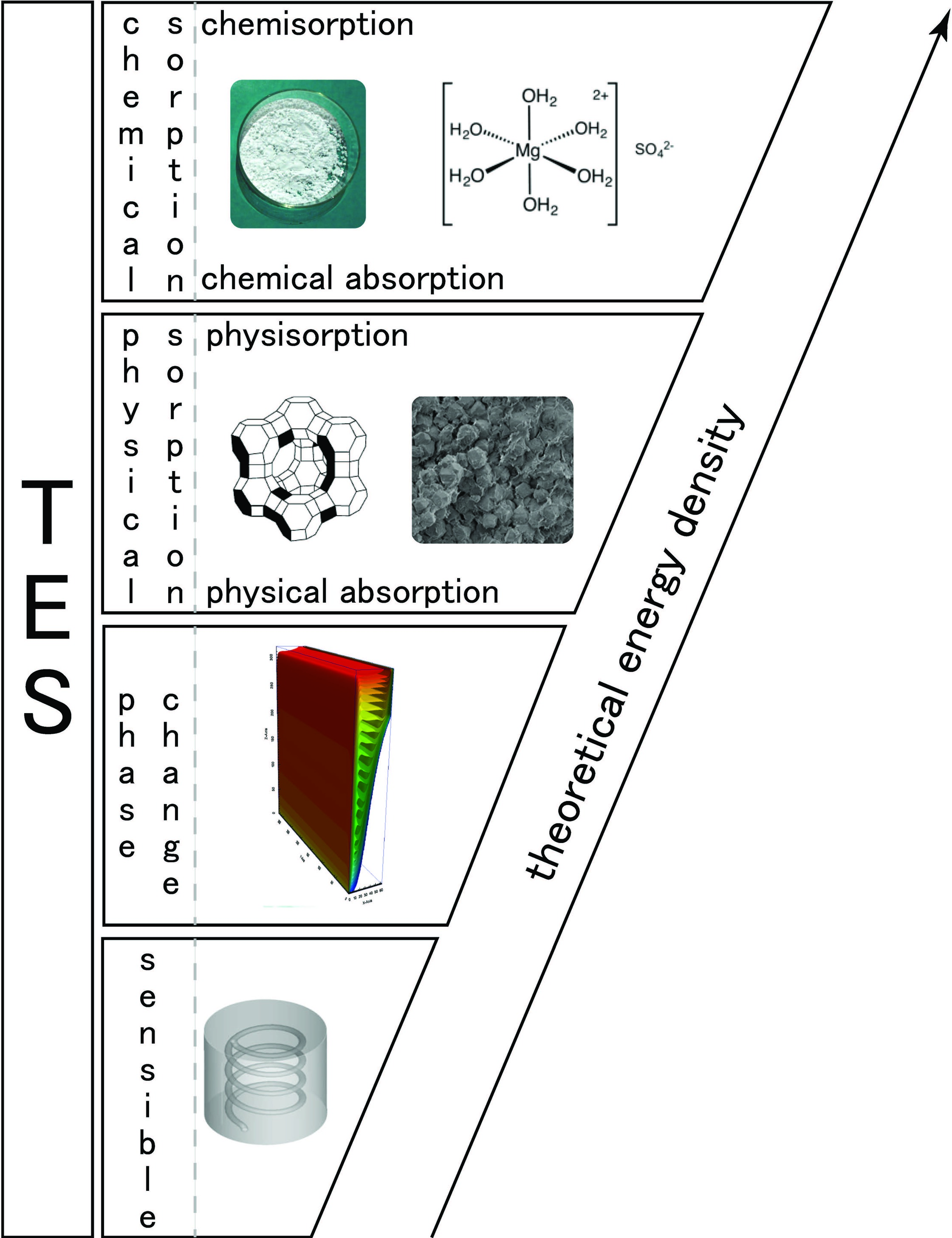
The theoretical limits of water sorbate based chemical sorption heat storage are investigated in this study. First, a classification of \textit{thermochemical heat storage} is proposed based on bonding typology. Then, thermodynamics of chemical solid/gas sorption is introduced. The analysis of the reaction enthalpy from the literature indicates that this value is only slightly varying for one mole of water. Using this observation, and with the help of thermodynamical considerations, it is possible to derive conclusions on energy efficiency of closed and open heat storage systems. Whatever the salt, the main results are 1) the energy required for evaporation of water is, at least, 65% of the available energy of reaction and 2) the maximum theoretical energy efficiency of the system is about 1.8.