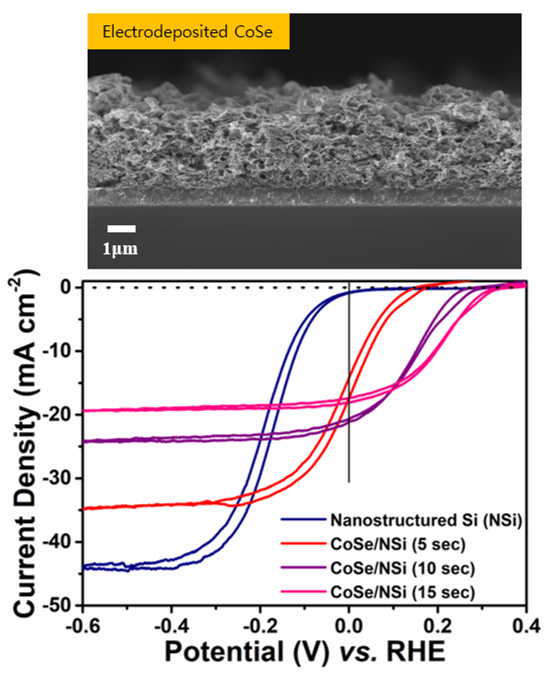
Photoelectrochemical water splitting is a promising field for sustainable energy production using hydrogen. Development of efficient catalysts is essential for resourceful hydrogen production. The most efficient catalysts reported to date have been extremely precious rare-earth metals. One of the biggest hurdles in this research area is the difficulty of developing highly efficient catalysts comparable to the noble metal catalysts. Here, we report that non-noble metal dichalcogenide (Co3Se4) catalysts made using a facile one-pot electrodeposition method, showed highly efficient photoelectrochemical activity on a Si photocathode. To enhance light collection and enlarge its surface area even further, we implemented surface nano-structuring on the Si surface. The nano-structured Si photoelectrode has an effective area greater than that of planar silicon and a wider absorption spectrum. Consequently, this approach exhibits reduced overvoltage as well as increased photo-catalytic activity. Such results show the importance of controlling the optimized interface between the surface structure of the photoelectrode and the electrodeposited co-catalyst on it to improve catalytic activity. This should enable other electrochemical reactions in a variety of energy conversion systems.