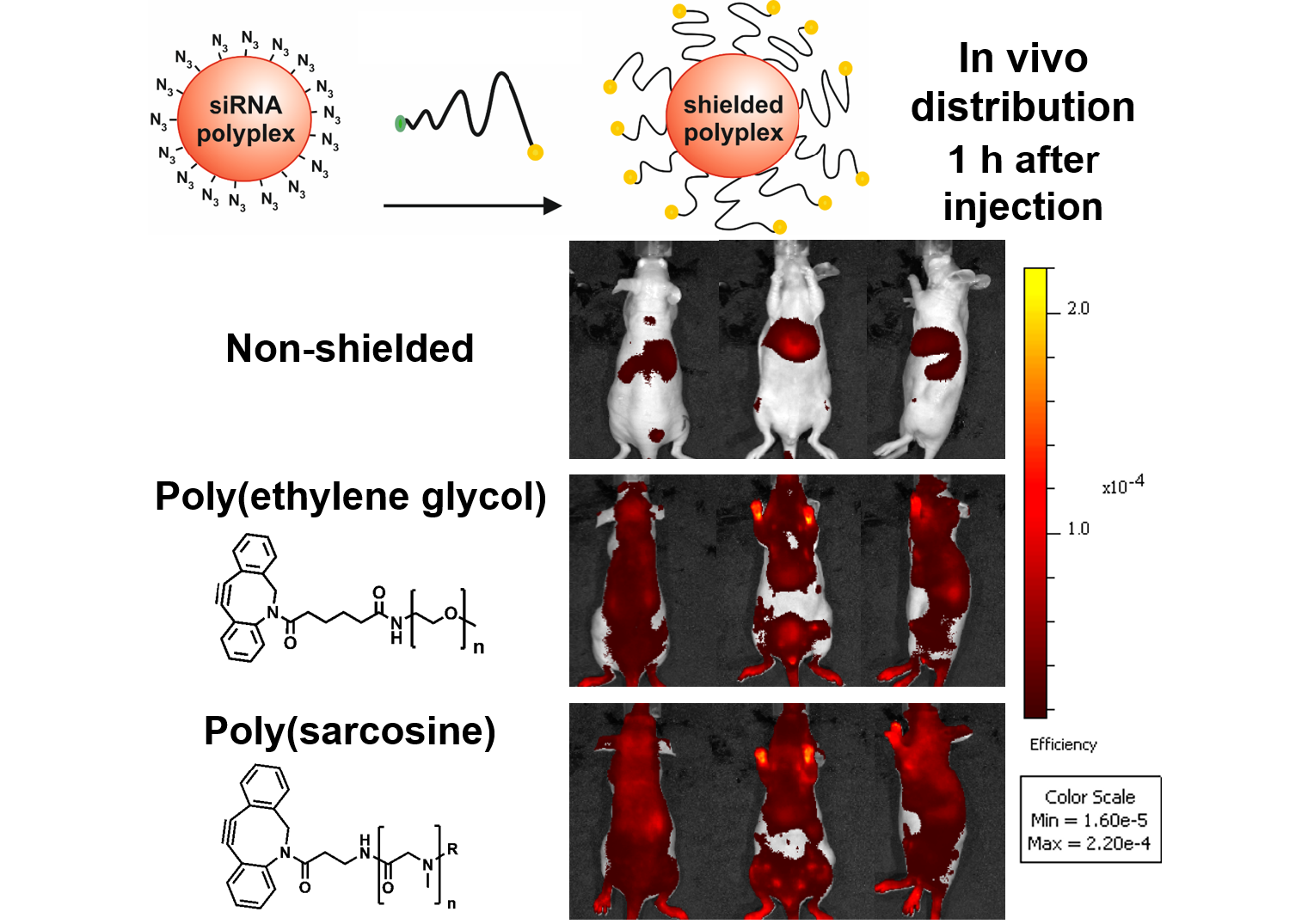
Shielding agents are commonly used to shield polyelectrolyte complexes, e.g. polyplexes, from agglomeration, precipitation in complex media, like blood, and thus enhance their circulation times in vivo. Since up to now primarily poly(ethylene glycol) (PEG) has been investigated to shield non-viral carriers for systemic delivery, we report on the use of polysarcosine (pSar) as a potential alternative for steric stabilization. A redox-sensitive, cationizable lipo-oligomer structure (containing two cholanic acids attached via a bioreducible disulfide linker to an oligoaminoamide backbone in T-shape configuration) was equipped with azide-functionality by solid phase supported synthesis. After mixing with small interfering RNA (siRNA), lipopolyplexes formed spontaneously and were further surface-functionalized with polysarcosines. Polysarcosine was synthesized by living controlled ring-opening polymerization using an azide-reactive dibenzo-aza-cyclooctyne-amine as an initiator. The shielding ability of the resulting formulations was investigated with biophysical assays and by near-infrared fluorescence bioimaging in mice. The modification of ~100 nm lipopolyplexes was only slightly increased upon functionalization. Cellular uptake into cells was strongly reduced by the pSar shielding. Moreover, polysarcosine-shielded polyplexes showed enhanced blood circulation times in bioimaging studies compared to unshielded polyplexes and similar to PEG-shielded polyplexes. Therefore, polysarcosine is a promising alternative for the shielding of non-viral, lipo-cationic polyplexes.