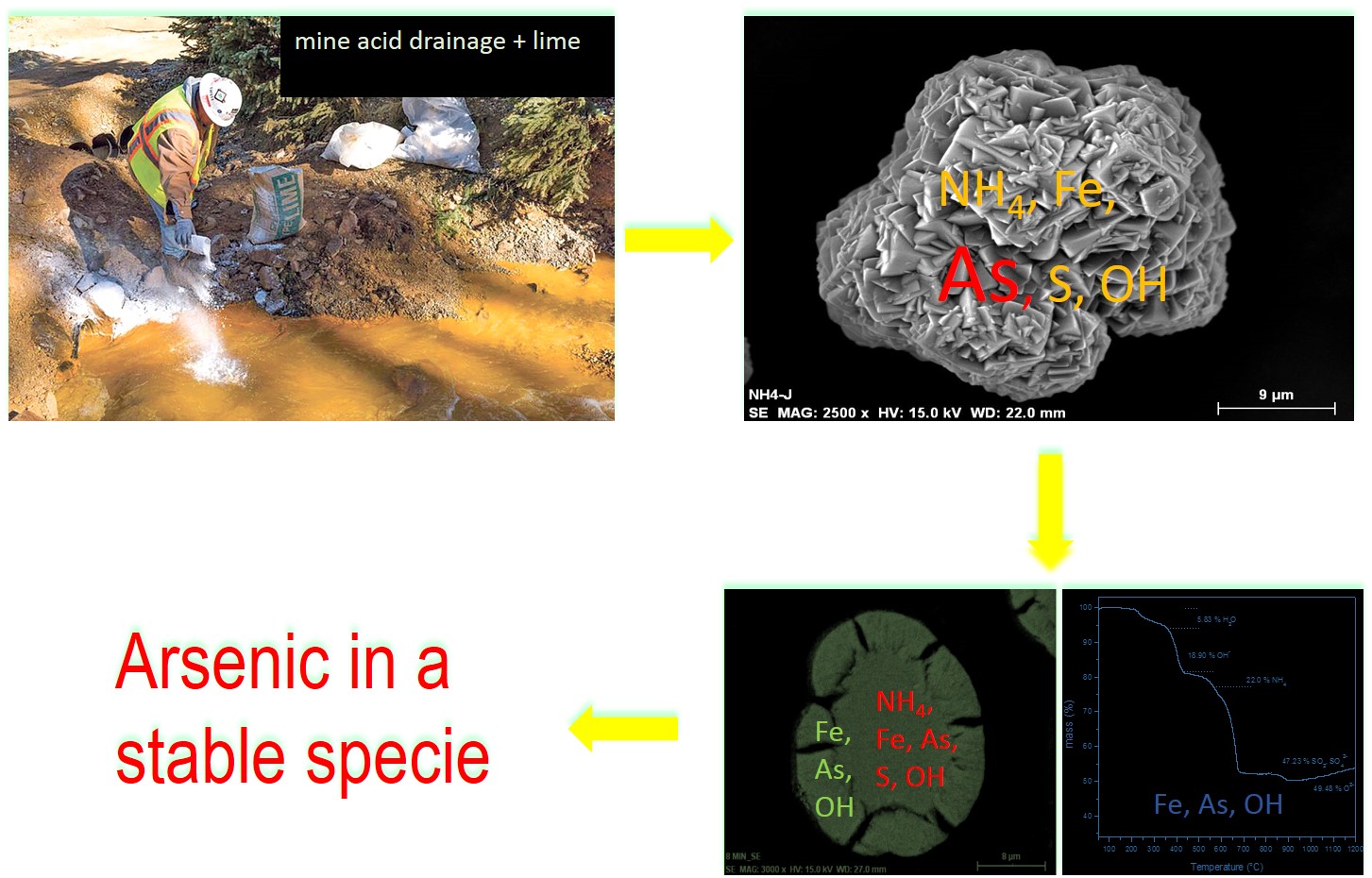
Arsenic, an element of environmental impact, can be incorporated into jarosite–type compounds and remain stabilised within the structure under a wide range of environmental conditions. In this study, a sample of ammonium–arsenic jarosite was synthesised by precipitation in sulphate medium at controlled pH of 1.2–1.8. The behaviour of arsenic during the thermal and chemical decomposition of jarosite was analysed; the degradation in alkaline medium of jarosite was also studied. According to the results, the synthesised jarosite is composed of joined rhombohedral crystals, forming tightly spherical shaped particles, 37–54 μm size. The ammonium jarosite produced possessed a high arsenic concentration; its calculated stoichiometry being (NH4)Fe2.45[(SO4)1.80(AsO4)0.20][(OH)4.15(H20)1.85]. It was found that arsenic is stabilised in the jarosite structure; upon heating, it remains in residual solids above 700°C, whilst in alkaline medium an incongruent dissolution takes place, with the arsenic retained in the solid phase along with iron. These solids, when exposed to high temperatures (1200°C), transform into a type of iron oxide known as hematite, so with arsenic it is retained an iron compound forming a stable compound which withstands high temperatures.