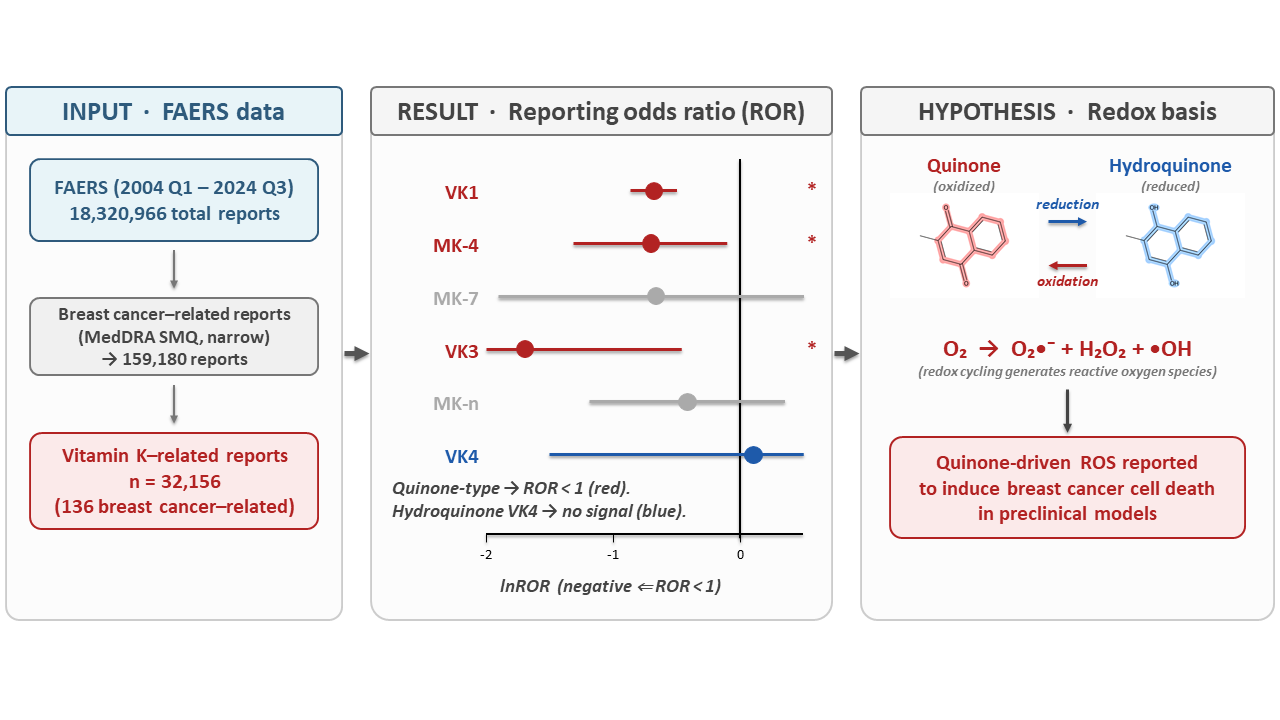
Background/Objective: Vitamin K (VK) comprises a family of quinone compounds with potential involvement in cell death-related pathways through their redox properties. However, consistent findings have not been obtained regarding the clinical significance of VK in breast cancer (BC). Thus, we used the FDA Adverse Event Reporting System (FAERS) to examine the co-reporting patterns of BC-related adverse-event terms among VK-related reports. Methods: Reporting disproportionality analysis was conducted using FAERS data spanning the first quarter of 2004 to the third quarter of 2024. BC-related reports were defined using all valid Preferred Terms included in the relevant narrow-scope Standardized MedDRA Query (SMQ). Reporting odds ratios (RORs) and proportional reporting ratios were calculated for all VK types and each homolog, followed by exploratory comparisons with other compounds containing quinone structures. Results: In total, 32,156 VK-related reports were identified, including 136 BC-related reports. VK-related reports showed significantly lower reporting disproportionality for breast cancer-related reports (ROR = 0.486, 95% confidence interval = 0.411–0.575). In homolog-specific analyses, similar trends were observed for the quinone-type homologs phytomenadione, menatetrenone, and menadione, whereas no significant reporting disproportionality was detected for the hydroquinone-type homolog menadiol. Conclusions: The differences in reporting patterns among quinone-type VK homologs, hydroquinone-type VK, and other quinone-containing compounds suggest that differences in redox properties may be partially related to the structure of reporting disproportionality. Although this study did not demonstrate causality or clinical efficacy, it provides a hypothesis-generating basis for linking basic, epidemiological, and clinical research using FAERS data. Future validation through mechanistic research and analytical epidemiological studies with stricter control of confounding is warranted.