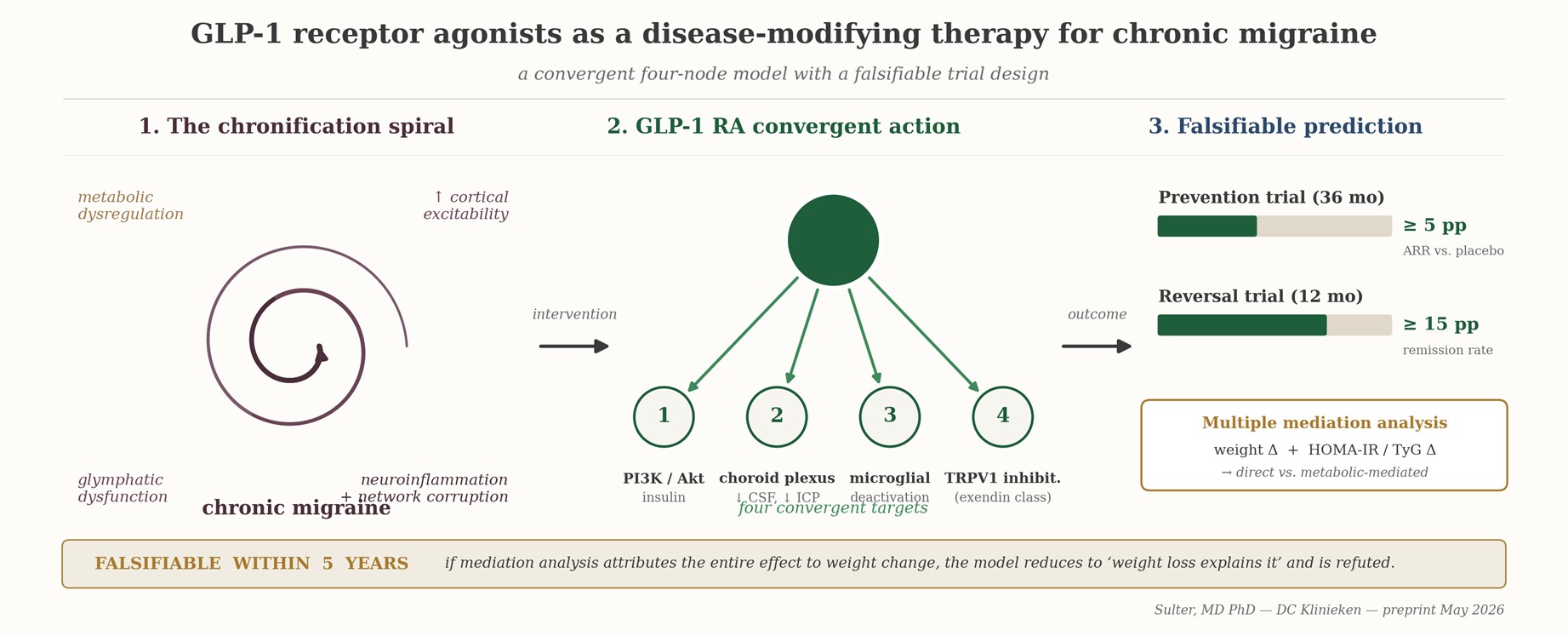
Chronic migraine is increasingly understood as a network-level disorder in which trigeminovascular nociception is sustained by metabolic, inflammatory, and macro-network dysfunction rather than by an isolated headache mechanism. Glucagon-like peptide-1 receptor agonists (GLP-1RAs), originally developed for type 2 diabetes and obesity, engage receptors expressed in choroid plexus, cortex, hippocampus, thalamus, and hypothalamus. Evidence converges on four mechanistic intersections between GLP-1 signalling and the chronic-migraine cascade: restoration of cerebral insulin signalling and cortical energy supply; modulation of cerebrospinal-fluid secretion and intracranial pressure with downstream relevance for glymphatic dynamics (most clearly validated in idiopathic intracranial hypertension, with explicit caveats for normotensive migraine); suppression of microglial activation in pain-relevant circuits (established preclinically, with one pilot human imaging study); and direct trigeminal-nociceptor effects via TRPV1 inhibition demonstrated for exendin-derived peptides. A 2026 real-world cohort of approximately 11,000 chronic-migraine adults provides a preliminary clinical signal. We propose, and operationalise as falsifiable, the hypothesis that GLP-1RAs are disease-modifying rather than purely symptomatic in chronic migraine, and we describe a placebo-controlled trial with parallel multiple mediation analysis on weight change AND HOMA-IR/TyG delta that can refute the claim within five years. A biochemical scheiding between exendin-based agonists (exenatide, lixisenatide) and human GLP-1 analogues (semaglutide, liraglutide, dulaglutide) generates a falsifiable sub-hypothesis on peripheral TRPV1 inhibition that is testable retrospectively on data that already exist.