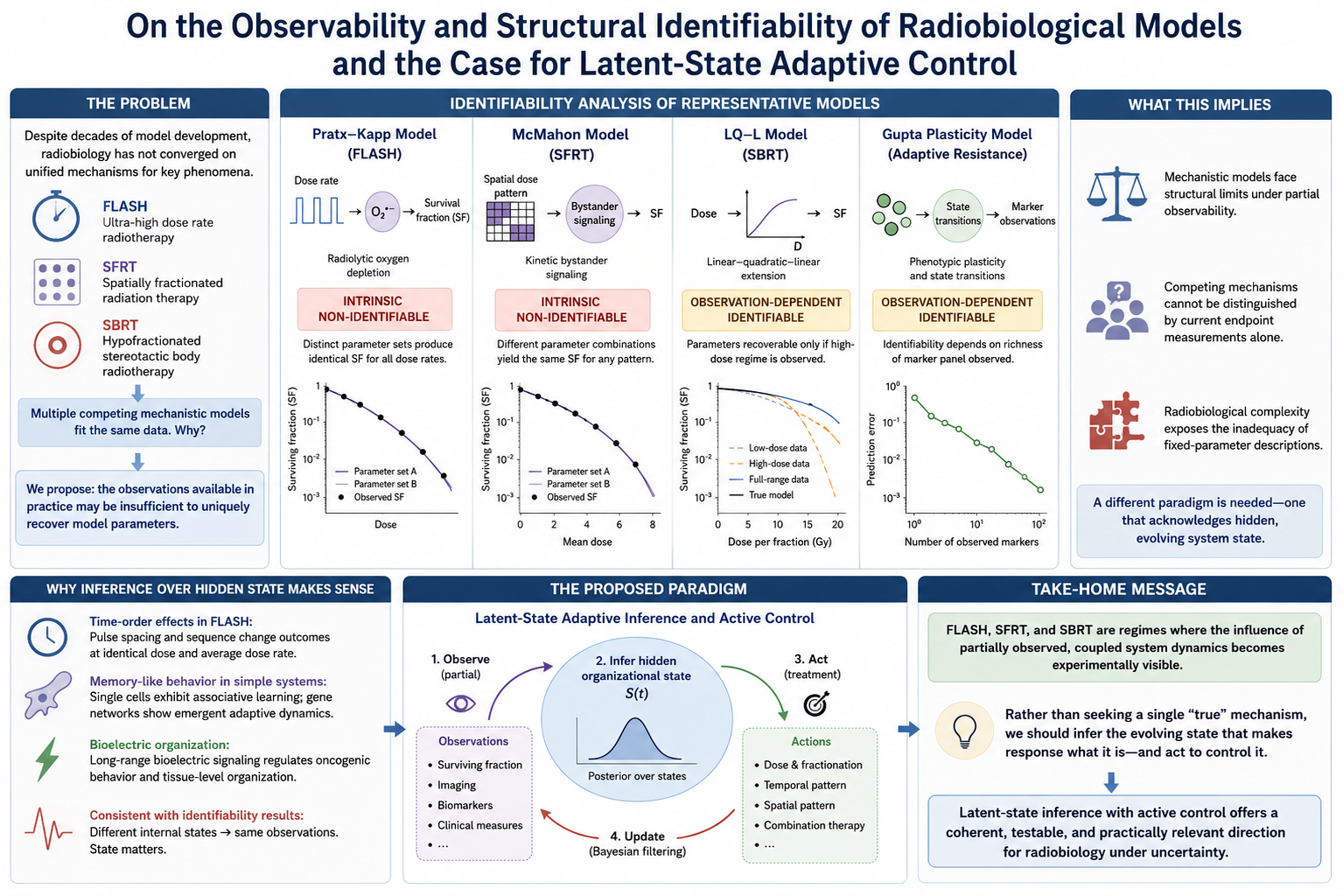
Mechanistic models in radiobiology have proliferated across FLASH ultra-high dose-rate radiotherapy, spatially fractionated radiation therapy (SFRT), and stereotactic body radiotherapy (SBRT), yet the field has not converged on unified mechanistic explanations despite decades of model development. We propose that this proliferation partly reflects a structural property of the model-observation pairing itself: clinically accessible measurements may be insufficient to uniquely recover the parameters governing the underlying biological dynamics. Using generating-series structural identifiability analysis, we examine four representative models spanning the temporal, spatial, magnitude, and adaptive-state dimensions of radiobiological response: the Pratx-Kapp radiolytic oxygen depletion model (FLASH), the McMahon kinetic-bystander model (SFRT), the LQ-L extension (SBRT), and the Gupta phenotypic-plasticity model of adaptive resistance. The Pratx-Kapp and McMahon models exhibit intrinsic non-identifiability under conventional surviving-fraction observation, while the LQ-L and Gupta models exhibit observation-dependent identifiability conditional on dose-range coverage and marker-panel richness. These findings suggest that increasing radiobiological complexity progressively exposes the limitations of fixed-parameter mechanistic descriptions under partial observability. As a constructive response, we propose, as a hypothesis, that adaptive latent-state inference frameworks operating over a coupled multi-layer organizational state may provide a complementary operational paradigm for radiobiology under uncertainty.