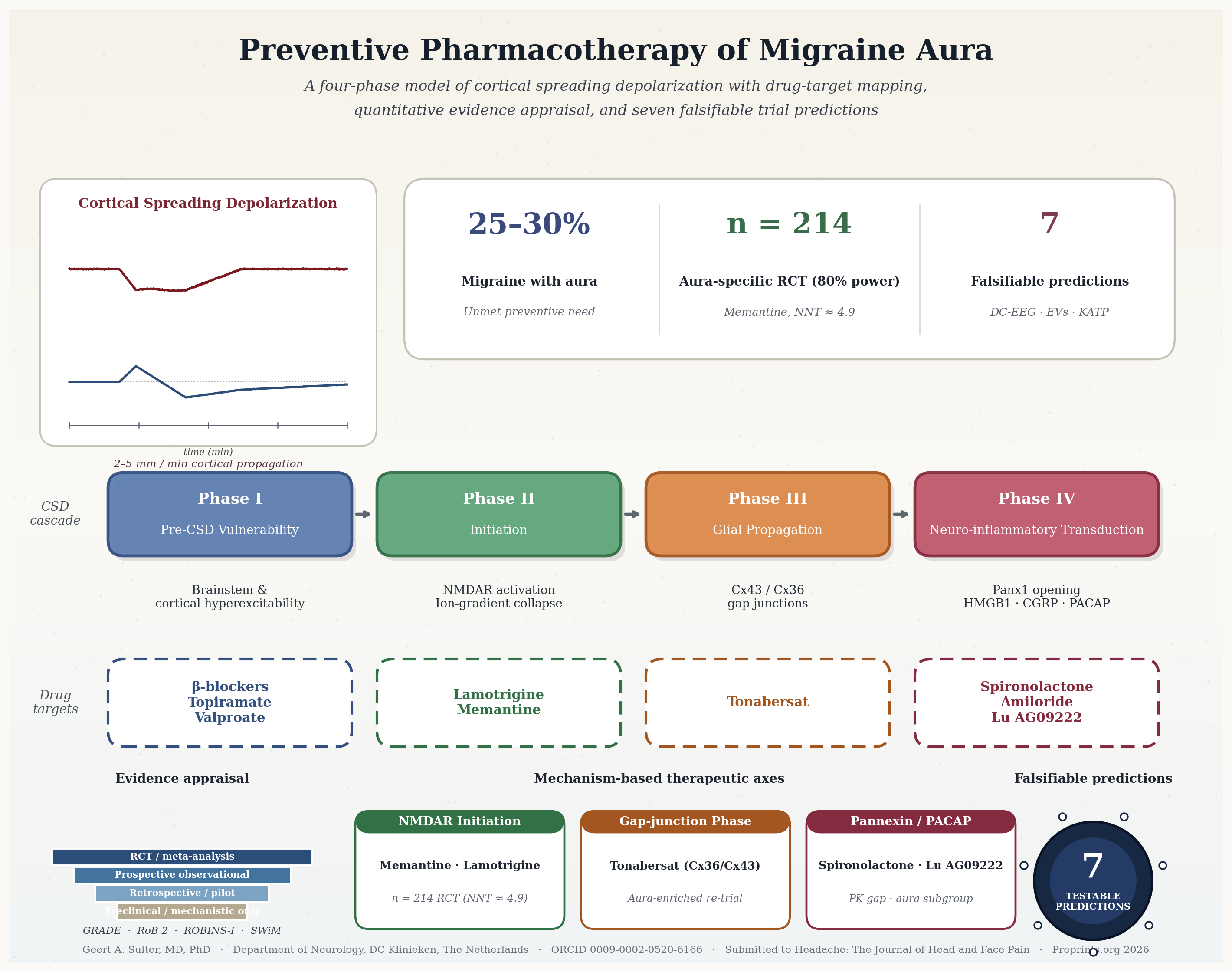
Objective: To synthesise the pathobiology of cortical spreading depolarization (CSD) and critically appraise current and emerging pharmacological strategies specifically targeting migraine aura prevention. Background: Migraine with aura affects 25–30% of patients, and the aura phenomenon remains a substantial unmet preventive need. Calcitonin gene-related peptide (CGRP) monoclonal antibodies do not readily cross the blood–brain barrier and frequently fail to suppress CSD, the neurophysiological substrate of aura.Methods: A literature search of PubMed, Embase, and the Cochrane Library (inception through January 2026) identified studies on CSD pathophysiology, preclinical CSD suppression, and clinical efficacy of candidate agents. Evidence quality was assessed with GRADE; risk of bias with Cochrane RoB 2 (RCTs) and ROBINS-I (observational); narrative synthesis followed SWiM. De novo quantitative estimations (post-hoc power analyses, sample-size projections, worst-case sensitivity analyses) were used as methodological tools, not as original empirical data. Results: CSD pathogenesis is organised into four phases: pre-CSD vulnerability, initiation, glial propagation, and neuro-inflammatory transduction. Lamotrigine and memantine target initiation and have the most advanced clinical evidence; both lack aura-specific RCTs. A 2024 network meta-analysis ranked memantine favourably (50% responder rate OR 5.58, 95% CI 2.41–12.92) but no contributing trial stratified by aura. An a priori sample-size calculation indicates 214 enrolled patients (170 evaluable; NNT≈4.9; n/(1−d) for 20% attrition) for a definitive aura-specific memantine RCT. Tonabersat—a Cx36/Cx43 gap-junction modulator—reduced aura attacks from 3.2 to 1.0 per 12 weeks in Phase 2; a worst-case intention-to-treat sensitivity analysis confirms that this signal survives even 16.6% unaccounted attrition. Spironolactone (pannexin-1 inhibition) and amiloride (ASIC1a) remain preclinical or pilot-stage. Tissue-selective KATP antagonists (Kir6.1/SUR2B) and the anti-PACAP-38 antibody Lu AG09222 represent the most promising pipeline agents. Conclusion: The therapeutic gap for migraine aura prevention reflects correctable methodological choices, not a lack of biological tractability. Four mechanism-based drug classes—NMDA-receptor antagonists, pannexin-1 inhibitors, gap-junction modulators, and KATP antagonists—offer entry points for aura-specific prevention. Adequately powered, aura-enriched RCTs with validated CSD biomarkers (DC-EEG co-registered against electrocorticography; neuron-derived extracellular vesicles) and pre-specified falsifiability thresholds are now the rate-limiting step. Seven testable methodological predictions are proposed.