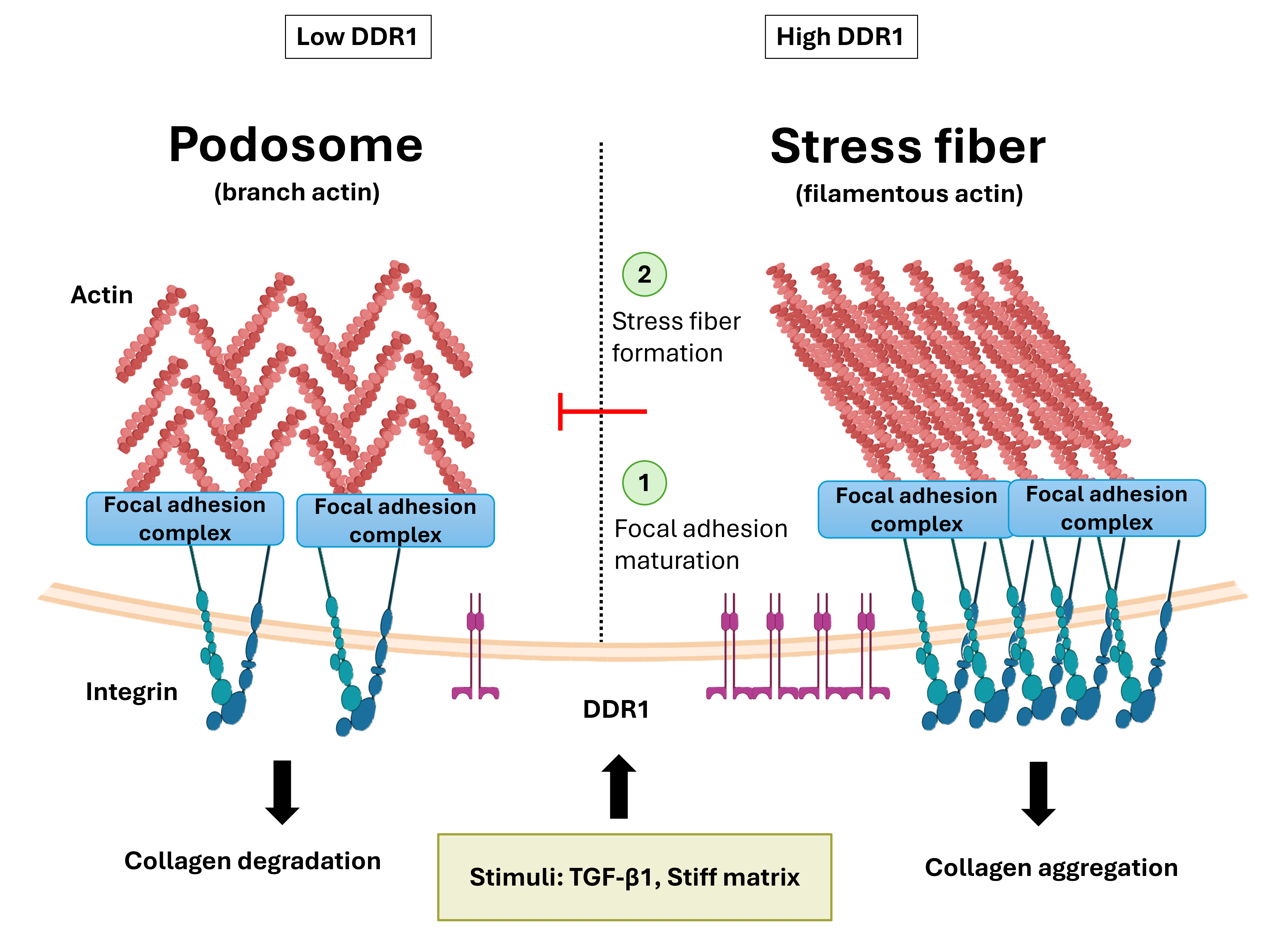
Discoidin domain receptor 1 (DDR1) has been implicated in fibrotic progression in multiple organs, including the kidney; however, its role in regulating cytoskeletal organization and matrix remodeling in renal fibroblasts remains unclear. Here, we investigated how DDR1 expression is regulated by profibrotic stimulation and extracellular matrix stiffness, and how DDR1 influences cytoskeletal organization and collagen remodeling. Single-cell RNA sequencing of murine kidneys subjected to unilateral ureteral obstruction (UUO) revealed enrichment of Ddr1 expression in transitional fibroblast populations during early activation. In vitro, transforming growth factor-β1 (TGF-β1) increased DDR1 expression, but DDR1 depletion did not affect canonical myofibroblast marker expression. Instead, DDR1 depletion suppressed stress fiber assembly while promoting actin-rich podosome formation associated with matrix degradation. Functionally, DDR1-deficient cells exhibited impaired focal adhesion maturation, enhanced collagen degradation, reduced gel contraction, and decreased collagen matrix stiffness as measured by atomic force microscopy. Furthermore, extracellular matrix stiffness dynamically regulated DDR1 expression, suggesting a bidirectional relationship between DDR1 expression and matrix mechanics. Together, these findings identify DDR1 as a modulator of cytoskeletal remodeling that governs the balance between matrix-degradative and contractile remodeling programs in renal fibroblasts.