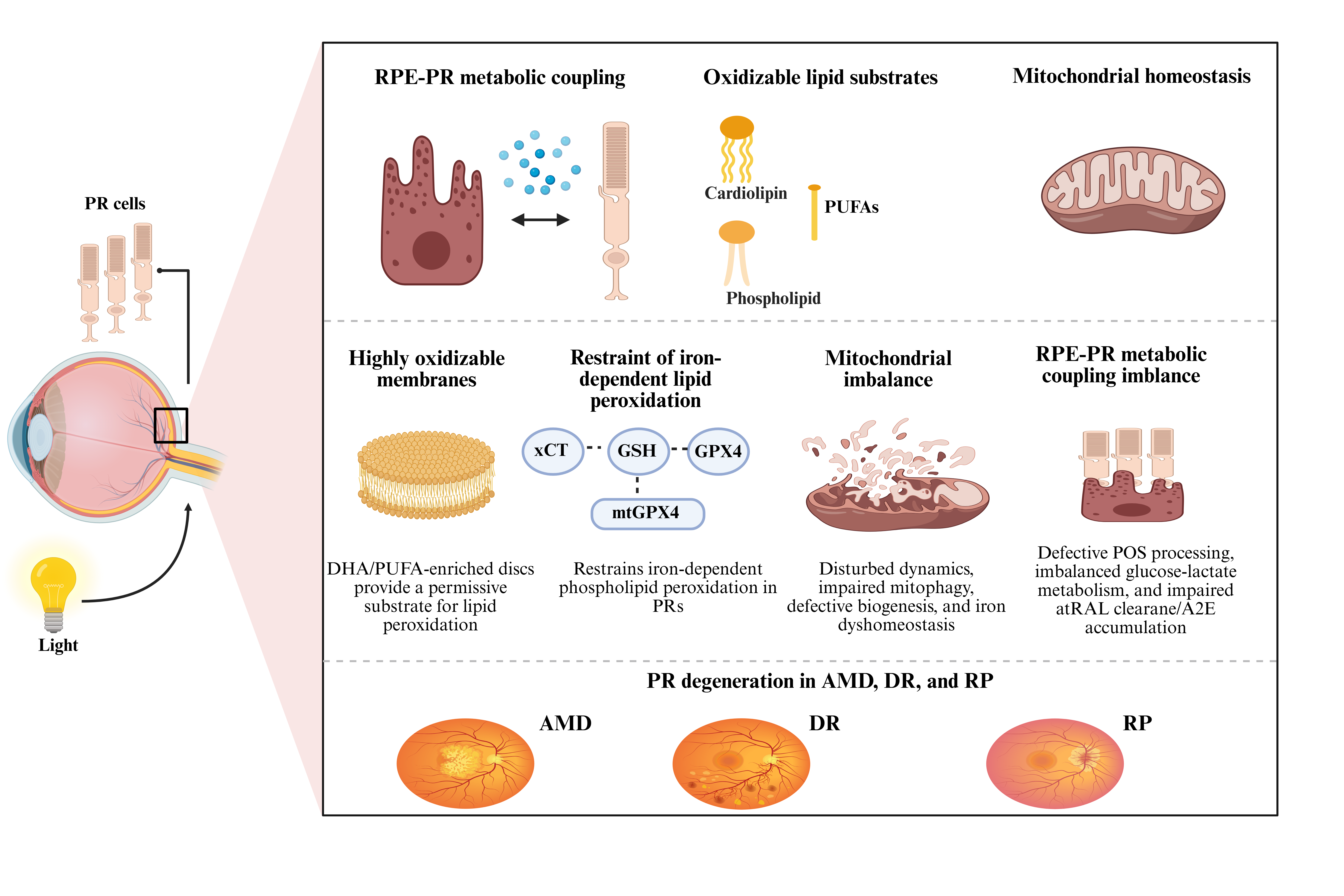
Photoreceptor (PR) degeneration is a shared pathological feature of multiple blinding retinal diseases. This review examines the mechanisms underlying PR vulnerability to lipid-peroxidation-driven injury, with emphasis on three interconnected features: the marked enrichment of docosahexaenoic acid (DHA) and other polyunsaturated fatty acids (PUFAs) in PR outer-segment disc membranes; the chronically high metabolic demand of PRs and the specialized spatial organization of their mitochondria; and retinal pigment epithelium (RPE)–PR metabolic coupling, including outer-segment renewal and phagocytic turnover, glucose transport and lactate shuttling, and the visual cycle. We also summarize antioxidant defense systems centered on the cystine/glutamate antiporter (xCT)–glutathione (GSH)–glutathione peroxidase 4 (GPX4) axis and mitochondrial GPX4 (mtGPX4), which restrict iron-dependent lipid peroxidation in PRs. We propose that highly oxidizable membrane lipid substrates, mitochondrial homeostatic imbalance, and impaired RPE–PR metabolic coupling may collectively shape PR susceptibility to ferroptosis-associated injury. From a therapeutic perspective, this framework supports multitarget strategies designed to interrupt lipid-peroxidation propagation, stabilize mitochondrial redox homeostasis and quality-control mechanisms, and restore RPE–PR metabolic support and local iron-buffering capacity.