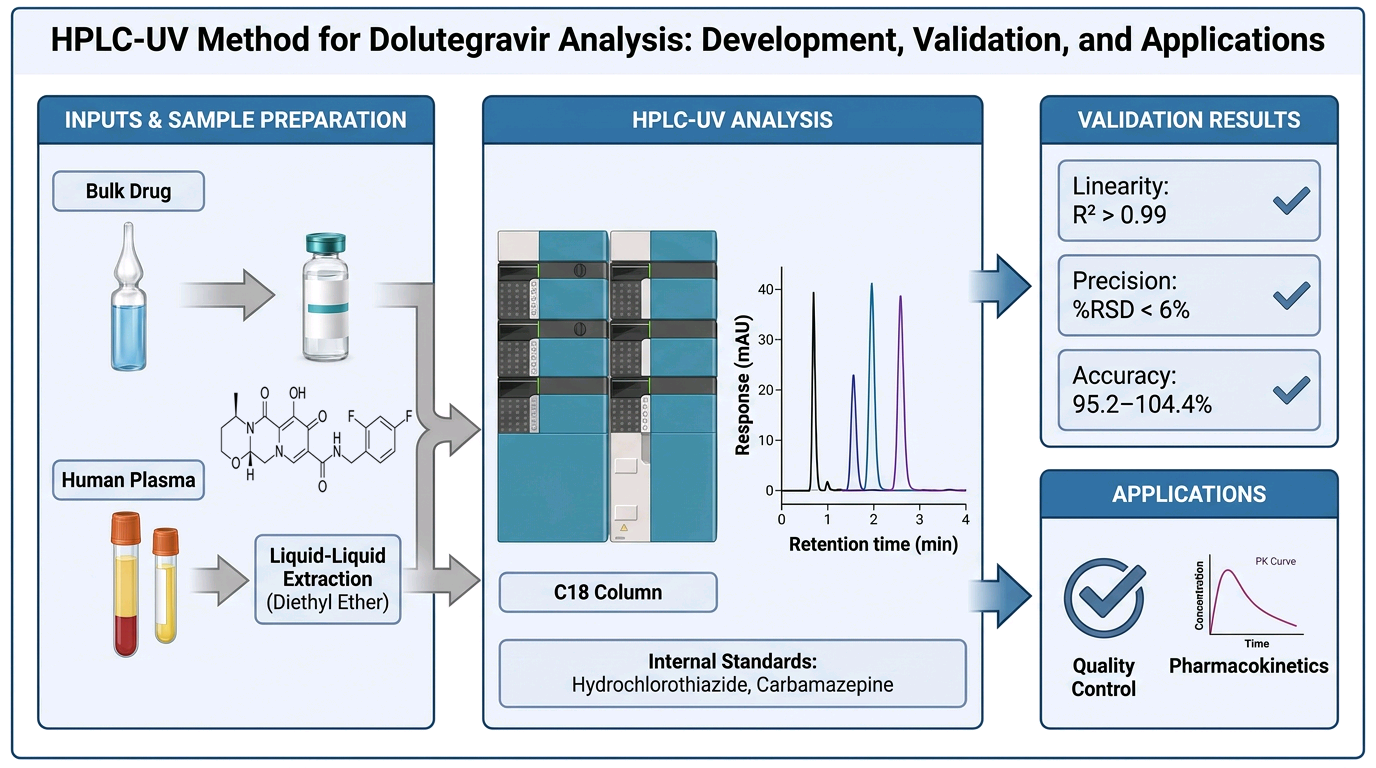
Background/Objectives: Dolutegravir is an integrase strand transfer inhibitor widely used in HIV therapy due to its high resistance barrier and favourable safety profile. As pharmacopoeial coverage continues to evolve and local formulation efforts increase, relia-ble analytical methods are required to ensure drug quality, safety, and efficacy. This study aimed to develop and validate two simple, sensitive, and cost-effective high-performance liquid chromatography with UV detection (HPLC-UV) methods for quantifying dolute-gravir in bulk form and human plasma. Methods: Reverse-phase HPLC methods were developed and validated according to ICH Q2(R2) guidelines. Hydrochlorothiazide and carbamazepine were used as internal standards for bulk and plasma analysis, respective-ly. Chromatographic separation was achieved on C18 columns. Key validation parameters included linearity, accuracy, precision, limit of detection (LOD), limit of quantitation (LOQ), robustness, and system suitability. Plasma samples were prepared using liquid–liquid extraction with diethyl ether. Results: The bulk method showed excellent linearity over 0.5–100 µg/mL with an R2 of 0.9997, while the plasma method was linear over 0.8–10 µg/mL with an R2 of 0.9956. Accuracy ranged from 95.2% to 104.4% for bulk and showed acceptable recoveries for plasma samples. Precision was satisfactory with %RSD values below 6% across all methods. LOD and LOQ were 0.42 µg/mL and 1.26 µg/mL for bulk analysis, and 0.55 µg/ml and 1.68 µg/ml, for plasma analysis respectively. Robustness and system suitability tests confirmed method reliability. Conclusions: The developed HPLC-UV methods are simple, reproducible, and suitable for routine quality control of dolutegravir in pharmaceutical formulations and for its quantification in human plasma for pharmacokinetic applications.