Submitted:
06 May 2026
Posted:
07 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Methods
2.2.1. MRI Acquisition
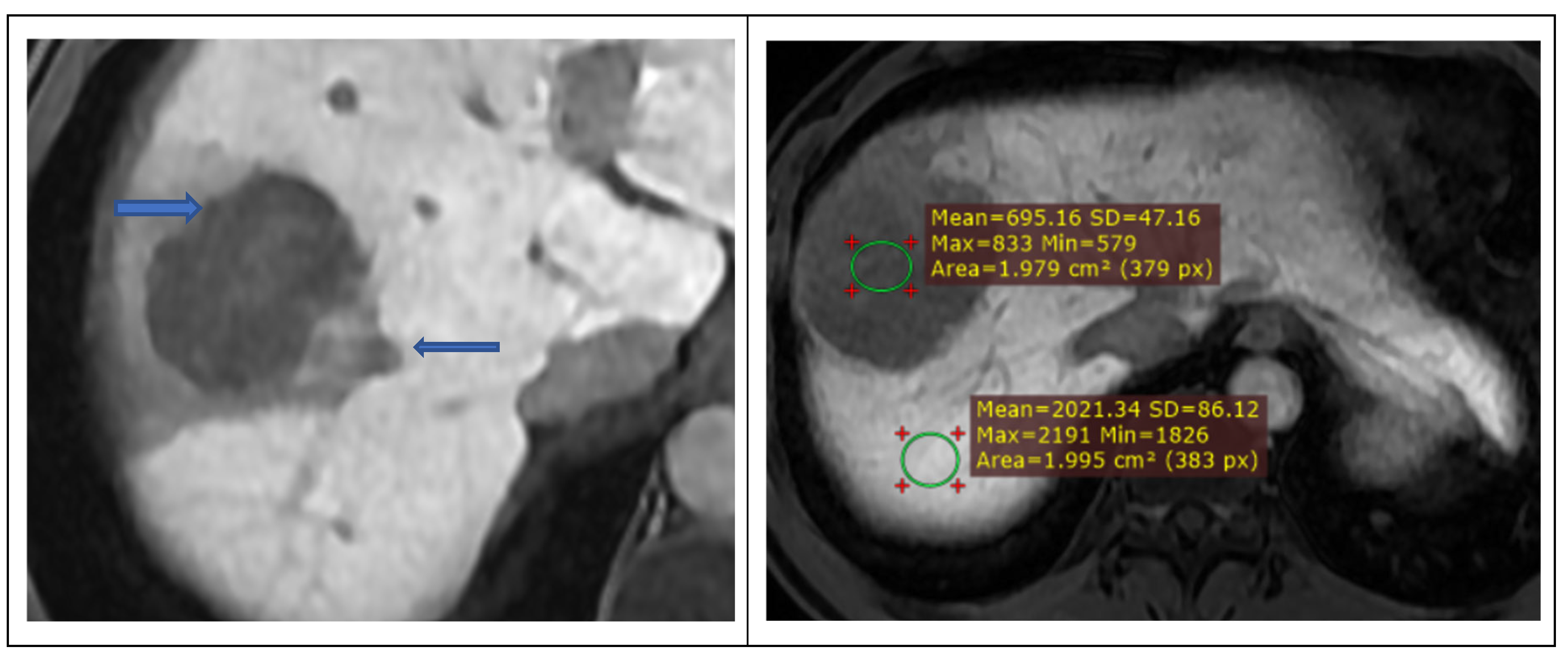
2.2.2. Image Analysis
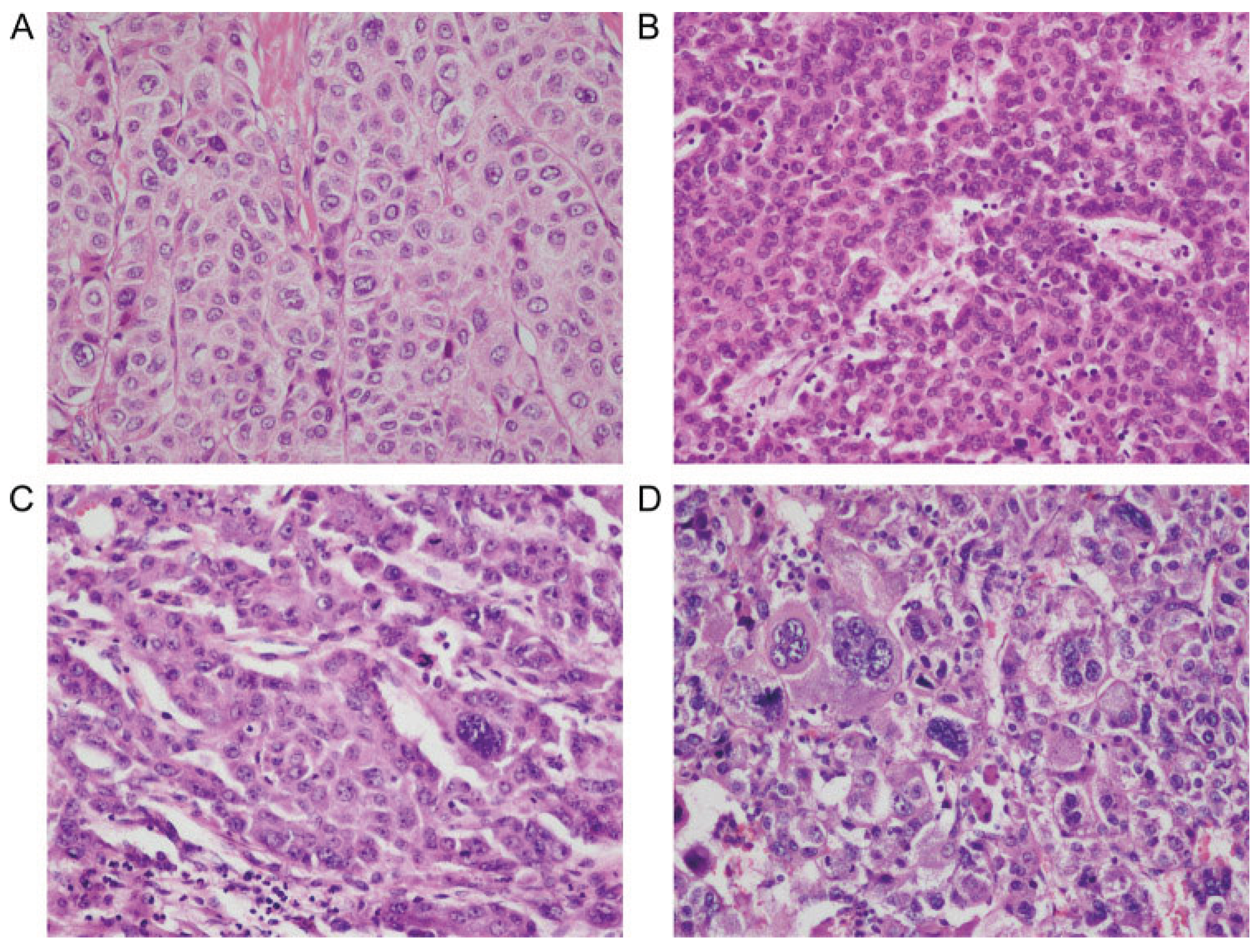
2.2.3. Histopathology
2.2.4. Clinical and Biochemical Variables
2.2.5. Pathologic Assessment
2.3. Statistical Analysis
2.4. Ethical Considerations
3. Results
| Variable | Grade I–II (n=24) | Grade III–IV (n=51) | p-value |
| Demographic characteristics, mean ± SD | |||
| Age (years) | 54.46 ± 12.65 | 57.35 ± 10.64 | 0.39 |
| Sex, n (%) | |||
| Male | 22 (91.7) | 44 (86.3) | 0.77 |
| Female | 2 (8.3) | 7 (13.7) | 0.77 |
| Medical history and risk factors, n (%) | |||
| Hepatitis C | 3 (12.5) | 7 (13.7) | 0.90 |
| Hepatitis B | 20 (83.3) | 40 (78.4) | 0.76 |
| Underlying cirrhosis | 8 (33.3) | 13 (25.5) | 0.58 |
| Biochemical markers, mean ± SD or median (IQR) | |||
| AFP (ng/mL) | 9.2 (4.6–110.2) | 42.4 (10.1–361.2) | 0.10 |
| Bilirubin (µmol/L) | 13.02 ± 4.75 | 11.90 ± 5.87 | 0.28 |
| Albumin (g/L) | 43.92 ± 2.57 | 41.72 ± 3.06 | 0.002 |
| Liver functional reserve | |||
| ALBI score, mean ± SD | -3.01 ± 0.20 | -2.90 ± 0.36 | 0.11 |
| ALBI grade, n (%) | |||
| Grade 1 | 24 (100) | 43 (84.3) | 0.05 |
| Grade 2–3 | 0 (0.0) | 8 (15.7) |
| Imaging feature | Grade I–II (n=24) | Grade III–IV (n=51) | p-value |
| Tumor size | |||
| Largest tumor diameter (mm), mean ± SD | 45.88 ± 26.02 | 55.02 ± 24.27 | 0.02 |
| Number of tumors per patient | 27 | 61 | |
| Tumor number, n (%) | |||
| 1 lesion | 21 (87.5) | 43 (84.3) | 0.72 |
| ≥2 lesions | 3 (12.5) | 8 (15.7) | |
| APHE, n (%) | |||
| Present | 23 (95.8) | 46 (90.2) | 0.66 |
| Absent | 1 (4.2) | 5 (9.8) | |
| Washout, n (%) | |||
| Present | 16 (66.7) | 45 (88.2) | 0.06 |
| Absent | 8 (33.3) | 6 (11.8) | |
| Capsule appearance, n (%) | |||
| Present | 14 (58.3) | 30 (58.8) | 0.97 |
| Absent | 10 (41.7) | 21 (41.2) | |
| Hepatobiliary phase tumor signal, n (%) | |||
| Hypointense | 7 (29.2) | 36 (70.6) | 0.002 |
| Hyperintense | 11 (45.8) | 11 (21.6) | |
| Isointense / non-specific | 6 (25.0) | 4 (7.8) | |
| Tumor-to-liver signal ratio (LLR), mean ± SD | 0.62 ± 0.29 | 0.47 ± 0.13 | 0.03 |
| Peritumoral arterial hyperintensity, n (%) | |||
| Present | 0 (0.0%) | 20 (39.2%) | <0.001 |
| Absent | 24 (100%) | 31 (60.8%) | |
| Peritumoral HBP hypointensity, n (%) | |||
| Present | 1 (4.2%) | 32 (62.7%) | <0.001 |
| Absent | 23 (95.8%) | 19 (37.3%) | |
| Quantitative index | |||
| HBP LLR, mean ± SD | 0.62 ± 0.29 | 0.47 ± 0.13 | 0.03 |
| Predictor | B coefficient | SE | OR | 95% CI | p-value |
| Peritumoral HBP hypointensity | |||||
| No | 1.00 | Reference | <0.001 | ||
| Yes | 3.66 | 1.06 | 38.74 | 4.83–310.39 | |
| HBP tumor signal intensity | 1.15 | 0.37 | 3.16 | 1.52–6.54 | 0.002 |
| Tumor size | 0.18 | 0.12 | 1.19 | 0.94–1.52 | 0.149 |
| HBP LLR | 0.38 | 0.15 | 1.46 | 1.09–1.95 | 0.01 |
| Elevated AFP (≥20 ng/mL) | |||||
| No | 1.00 | Reference | 0.04 | ||
| Yes | 1.03 | 0.51 | 2.80 | 1.03–7.60 | |
| Liver function (ALBI) | |||||
| ALBI score | 1.20 | 0.85 | 3.32 | 0.62–17.71 | 0.149 |
| Predictor | B coefficient | SE | OR | 95% CI | p-value |
| Peritumoral HBP hypointensity | |||||
| No | 1.00 | Reference | 0.002 | ||
| Yes | 3.43 | 1.11 | 30.89 | 3.54–269.66 | |
| HBP tumor signal intensity | 0.72 | 0.44 | 2.06 | 0.87–4.87 | 0.10 |
| Elevated AFP (≥20 ng/mL) | |||||
| No | 1.00 | Reference | 0.38 | ||
| Yes | -0.58 | 0.66 | 0.56 | 0.15–2.04 | |
| Liver function (ALBI) | |||||
| ALBI score | 1.03 | 1.01 | 2.81 | 0.39–20.32 | 0.31 |
| Constant | 2.36 | 3.03 | 0.44 | ||
| Predictive model | AUC | Se (%) | Sp (%) | PPV (%) | NPV (%) | Accuracy (%) |
| Individual parameters | ||||||
| Tumor size (cm) | 0.67 | 90.2 | 45.8 | 78.0 | 68.8 | 76.0 |
| APHE | 0.53 | 9.8 | 95.8 | 83.3 | 33.3 | 37.3 |
| Peritumoral HBP hypointensity | 0.79 | 62.7 | 95.8 | 97.0 | 54.8 | 73.3 |
| HBP tumor signal intensity | 0.72 | 70.6 | 70.8 | 83.7 | 53.1 | 70.7 |
| Elevated AFP (≥20 ng/mL) | 0.57 | 76.5 | 37.5 | 72.2 | 42.9 | 64.0 |
| ALBI grade 2–3 | 0.58 | 15.7 | 100 | 100 | 35.8 | 42.7 |
| Combined models | ||||||
| 2-parameter MRI model | 0.84 | 62.7 | 95.8 | 97.0 | 54.8 | 73.3 |
| 4-parameter combined model | 0.87 | 74.5 | 87.5 | 92.7 | 61.8 | 78.7 |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Laversanne, M.; Sung, H.; et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2024, 74(3), 229–263. [Google Scholar] [CrossRef] [PubMed]
- Huy Do, S. Epidemiology of Hepatitis B and C Virus Infections and Liver Cancer in Vietnam. Euroasian J. Hepato-Gastroenterol. 2015, 5(1), 49–51. [Google Scholar] [CrossRef] [PubMed]
- Martins-Filho, S.N.; Paiva, C.; Azevedo, R.S.; Alves, V.A.F. Histological Grading of Hepatocellular Carcinoma-A Systematic Review of Literature. Front. Med. 2017, 4, 193. [Google Scholar] [CrossRef]
- Edmondson, H.A.; Steiner, P.E. Primary carcinoma of the liver: a study of 100 cases among 48,900 necropsies. Cancer 1954, 7(3), 462–503. [Google Scholar] [CrossRef]
- Rodríguez-Perálvarez, M.; Luong, T.V.; Andreana, L.; Meyer, T.; Dhillon, A.P.; Burroughs, A.K. A systematic review of microvascular invasion in hepatocellular carcinoma: diagnostic and prognostic variability. Ann. Surg. Oncol. 2013, 20(1), 325–39. [Google Scholar] [CrossRef]
- Van Beers, B.E.; Pastor, C.M.; Hussain, H.K. Primovist, Eovist: what to expect? J. Hepatol. 2012, 57(2), 421–9. [Google Scholar] [CrossRef]
- Kitao, A.; Matsui, O.; Yoneda, N.; et al. The uptake transporter OATP8 expression decreases during multistep hepatocarcinogenesis: correlation with gadoxetic acid enhanced MR imaging. Eur. Radiol. 2011, 21(10), 2056–66. [Google Scholar] [CrossRef]
- Lee, S.; Kim, S.H.; Lee, J.E.; Sinn, D.H.; Park, C.K. Preoperative gadoxetic acid-enhanced MRI for predicting microvascular invasion in patients with single hepatocellular carcinoma. J. Hepatol. 2017, 67(3), 526–534. [Google Scholar] [CrossRef]
- Zhou, Z.; Cao, S.; Chen, C.; et al. A Novel Nomogram for the Preoperative Prediction of Edmondson-Steiner Grade III-IV in Hepatocellular Carcinoma Patients. J. Hepatocell. Carcinoma 2023, 10, 1399–1409. [Google Scholar] [CrossRef]
- Samban, S.S.; Hari, A.; Nair, B.; et al. An Insight Into the Role of Alpha-Fetoprotein (AFP) in the Development and Progression of Hepatocellular Carcinoma. Mol. Biotechnol. 2024, 66(10), 2697–2709. [Google Scholar] [CrossRef] [PubMed]
- Demirtas, C.O.; D’Alessio, A.; Rimassa, L.; Sharma, R.; Pinato, D.J. ALBI grade: Evidence for an improved model for liver functional estimation in patients with hepatocellular carcinoma. JHEP Rep. Innov. Hepatol. 2021, 3(5), 100347. [Google Scholar] [CrossRef]
- Choi, J.Y.; Lee, J.M.; Sirlin, C.B. CT and MR imaging diagnosis and staging of hepatocellular carcinoma: part II. Extracellular agents, hepatobiliary agents, and ancillary imaging features. Radiology 2014, 273(1), 30–50. [Google Scholar] [CrossRef] [PubMed]
- Chernyak, V.; Fowler, K.J.; Kamaya, A.; et al. Liver Imaging Reporting and Data System (LI-RADS) Version 2018: Imaging of Hepatocellular Carcinoma in At-Risk Patients. Radiology 2018, 289(3), 816–830. [Google Scholar] [CrossRef]
- Ahn, S.Y.; Lee, J.M.; Joo, I.; et al. Prediction of microvascular invasion of hepatocellular carcinoma using gadoxetic acid-enhanced MR and (18)F-FDG PET/CT. Abdom. Imaging 2015, 40(4), 843–51. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.A.; Kim, M.J.; Jeon, H.M.; et al. Prediction of microvascular invasion of hepatocellular carcinoma: usefulness of peritumoral hypointensity seen on gadoxetate disodium-enhanced hepatobiliary phase images. J. Magn. Reson. Imaging JMRI 2012, 35(3), 629–34. [Google Scholar] [CrossRef]
- Bai, D.S.; Zhang, C.; Chen, P.; Jin, S.J.; Jiang, G.Q. The prognostic correlation of AFP level at diagnosis with pathological grade, progression, and survival of patients with hepatocellular carcinoma. Sci. Rep. 2017, 7(1), 12870. [Google Scholar] [CrossRef] [PubMed]
- Edoo, M.I.A.; Chutturghoon, V.K.; Wusu-Ansah, G.K.; et al. Serum Biomarkers AFP, CEA and CA19-9 Combined Detection for Early Diagnosis of Hepatocellular Carcinoma. Iran. J. Public Health 2019, 48(2), 314–322. [Google Scholar] [PubMed]
- Yan, Y.; Si, Z.; Chun, C.; et al. Multiphase MRI-Based Radiomics for Predicting Histological Grade of Hepatocellular Carcinoma. J. Magn. Reson. Imaging JMRI 2024, 60(5), 2117–2127. [Google Scholar] [CrossRef]
- Mao, Y.; Wang, J.; Zhu, Y.; et al. Gd-EOB-DTPA-enhanced MRI radiomic features for predicting histological grade of hepatocellular carcinoma. Hepatobiliary Surg. Nutr. 2022, 11(1), 13–24. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Li, X.; Liu, J.; et al. The Combination of Gd-EOB-DTPA Enhanced T1 Mapping with Apparent Diffusion Coefficient could Improve the Diagnostic Efficacy of Hepatocellular Carcinoma Grading. Curr. Med. Imaging 2024, 20, e15734056259418. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, C.; Gu, S.; et al. Anatomical versus non-anatomical resection for solitary hepatocellular carcinoma without macroscopic vascular invasion: A propensity score matching analysis. J. Gastroenterol. Hepatol. 2017/04/01 2017, 32(4), 870–878. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Chen, M.; Cheng, A.L.; et al. Atezolizumab plus bevacizumab versus active surveillance in patients with resected or ablated high-risk hepatocellular carcinoma (IMbrave050): a randomised, open-label, multicentre, phase 3 trial. Lancet 2023, 402(10415), 1835–1847. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




