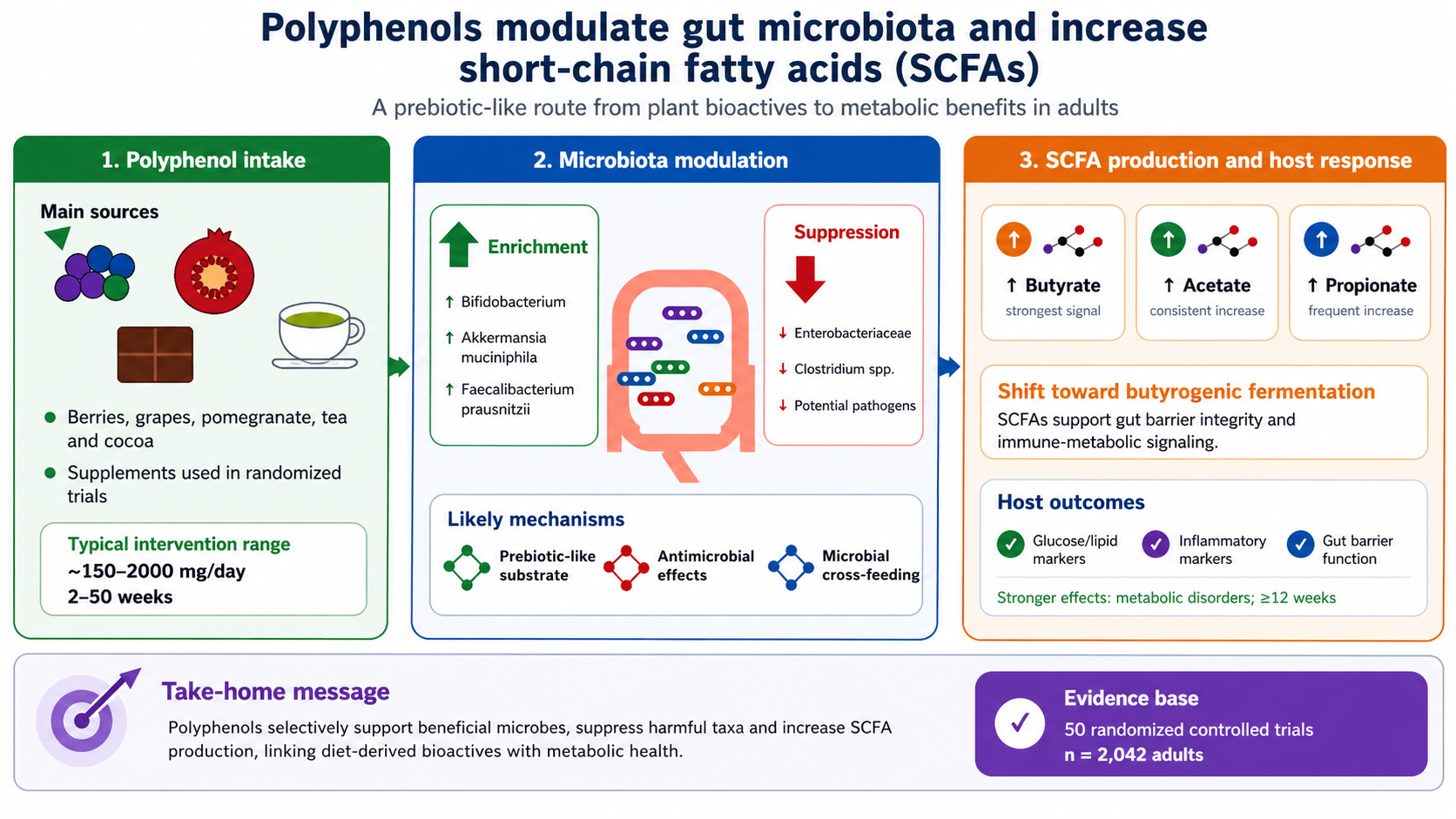
Background: Polyphenols interact bidirectionally with the gut microbiota and may enhance short‑chain fatty acid (SCFA) production, yet evidence from human randomized controlled trials (RCTs) has not been comprehensively synthesized. Objectives: This systematic review and meta‑analysis evaluated the effects of polyphenol supplementation on gut microbiota composition, microbial diversity, and fecal SCFA concentrations in adults, and examined moderators of these effects. Methods: Five databases were searched through October 2023 for RCTs assessing oral polyphenol supplementation in adults. Eligible studies reported outcomes related to gut microbiota composition or fecal SCFAs. Random‑effects meta‑analyses were conducted for SCFA outcomes, and subgroup analyses examined effects by polyphenol class, dose, duration, health status, and analytical methods. Risk of bias was assessed using the Cochrane RoB 2 tool, and certainty of evidence using GRADE. Results: Fifty RCTs (n = 2,042 participants) were included. Polyphenol supplementation increased total SCFAs in 70.6% of studies and significantly increased butyrate concentrations (pooled SMD = 0.48; 95% CI: 0.32–0.64; I² = 58%). Acetate and propionate increased in 75% and 71.4% of studies, respectively. A consistent shift toward a more butyrogenic fermentation profile was observed. Polyphenols enriched beneficial genera, including Bifidobacterium (81.8%), Akkermansia muciniphila (50%), and Faecalibacterium prausnitzii (45.5%), while reducing potentially pathogenic taxa such as Enterobacteriaceae and Clostridium spp. Improvements in alpha diversity were reported in 66.7% of studies, and beta diversity separation in 87.5%. Effects were stronger in individuals with metabolic disorders and in interventions lasting ≥12 weeks. Conclusions: Polyphenol supplementation consistently enhances beneficial gut bacteria, increases SCFA production, particularly butyrate, and improves microbial diversity in adults. These findings support classifying polyphenols as bioactive prebiotics that meet ISAPP criteria. This term underscores their distinction from traditional prebiotics within our proposed five-phase model: whereas fibers are characterized by their fermentability, polyphenols integrate direct antimicrobial activity against pathogenic species (e.g., Staphylococcus aureus, Enterobacteriaceae) with their role as selective substrates for beneficial microbes. This dual mechanism of action suggests that polyphenols do not merely supplement the microbiota but actively reshape it by pruning harmful taxa while fertilizing beneficial ones. Polyphenol-rich strategies represent promising microbiota-focused approaches; however, while shifts in microbial profiles coincide with better metabolic health, further research is required to bridge the correlation-causation gap and confirm whether these microbial changes directly drive clinical outcomes.