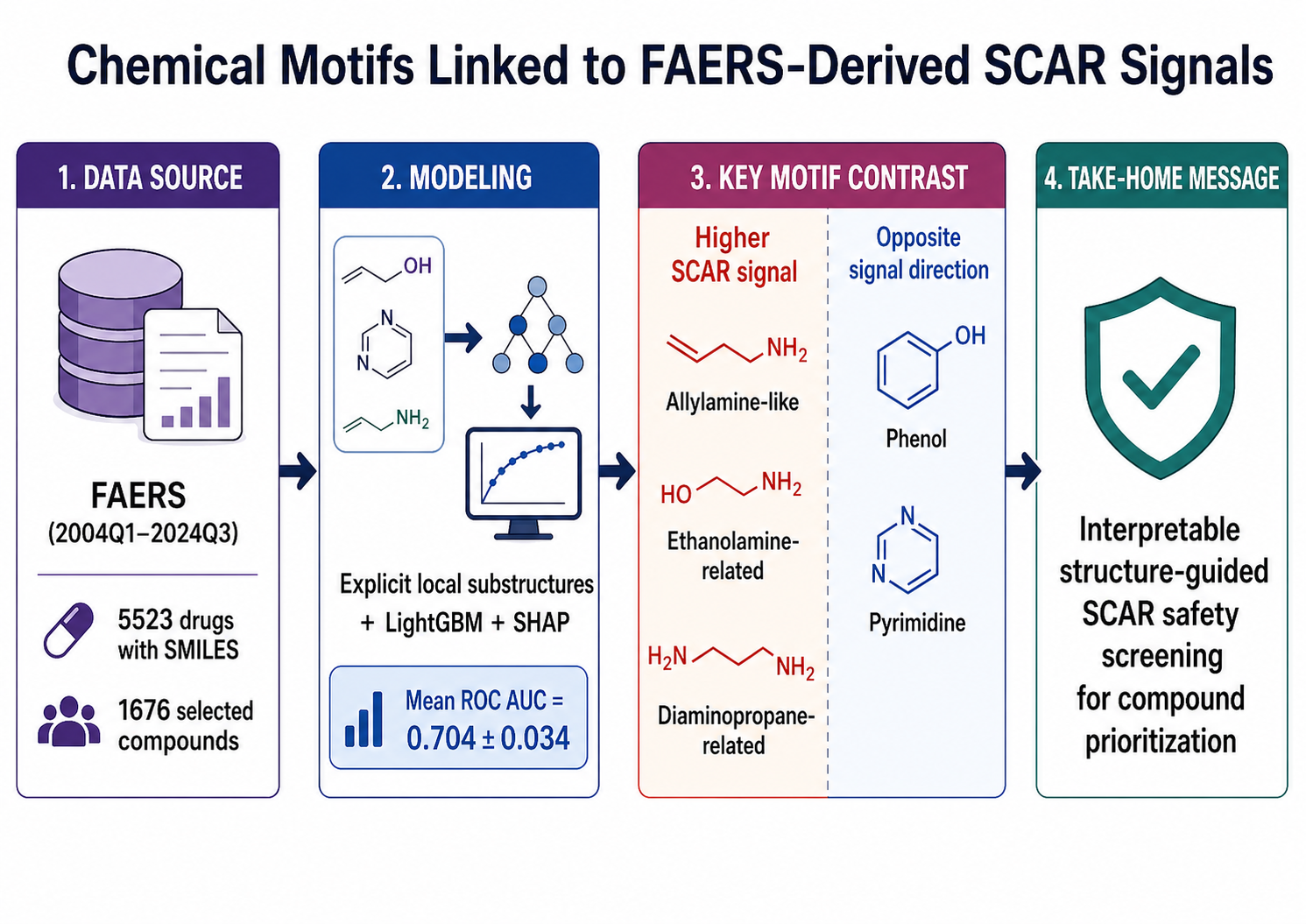
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening drug hypersensitivity syndromes. Although pharmacovigilance can identify drugs disproportionately reported with SCARs, it does not reveal which local chemistries recur among them. To address this, we assessed whether drugs with disproportionate SCAR reporting in the FDA Adverse Event Reporting System (FAERS) share interpretable chemical motifs. We screened FAERS data from 2004Q1 to 2024Q3, identified 5523 drugs with available Simplified Molecular-Input Line-Entry System (SMILES) representations, and constructed a signal-enriched dataset of 1676 compounds with nominally significant broad-SCAR associations after excluding predefined therapeutic/supportive confounders. Compounds were assigned to positive-signal [natural logarithm of reporting odds ratio (lnROR) > 0, n = 1219] or nonpositive-signal (lnROR ≤ 0, n = 457) classes and encoded with 9753 explicitly mappable atom-centered local substructure descriptors. A light gradient boosting machine classifier evaluated using repeated nested cross-validation (six-fold outer × 50 repeats) achieved moderate but stable internal discrimination (mean area under the receiver operating characteristic curve = 0.7041 ± 0.0337). SHapley Additive exPlanations (SHAP) analysis revealed a clear fragment-level contrast: allylamine-like, ethanolamine, and diaminopropane-related motifs were associated with higher positive-signal class probability, whereas phenol and pyrimidine motifs were associated with corresponding lower probability. The phenol fragment (Oc1ccccc1) was the most influential feature overall (mean |SHAP| = 0.1727), followed by an allylamine-like fragment (0.1031). These findings suggest that broad-SCAR concern detected in the FAERS is not chemically random within the selected dataset. Overall, the proposed framework should be viewed not as a direct predictor of absolute clinical SCAR risk but as an interpretable structure-guided safety-screening approach to prioritize compounds and motif families for further SCAR-focused evaluation.