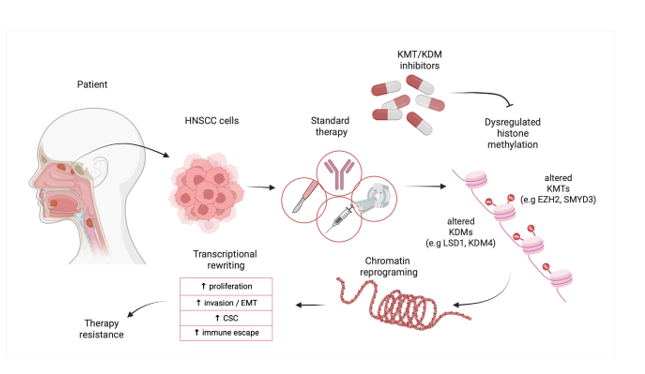
Despite advances in multimodal treatment, head and neck squamous cell carcinoma (HNSCC) remains a major clinical problem owing to its high recurrence rate and frequent development of treatment resistance. Abnormal histone modifications, particularly lysine methylation regulated by methyltransferases (KMTs) and demethylases (KDMs), have emerged as key drivers of HNSCC initiation, progression, and cellular plasticity. This review aims to comprehensively evaluate the role of selected KMTs and KDMs in HNSCC biology, with a focus on their contribution to resistance to immunotherapy, radiotherapy, and cytotoxic chemotherapy. We summarize and critically analyze preclinical and clinical studies investigating histone methylation dynamics in HNSCC, with particular emphasis on enzymes such as KMT2C/D, EZH2, NSD1/NSD2, SMYD3, G9a/EHMT2, LSD1, KDM2A/B, KDM3, KDM4, KDM5, KDM6, KDM7, and KDM8. Particular attention is given to pharmacological approaches targeting these proteins: we discuss smallmolecule inhibitors of EZH2, LSD1, KDM4/5/6, and other KMT/KDMs that are currently in preclinical development or earlyphase clinical trials, and we highlight completed and ongoing studies testing EZH1/2 inhibitors and epigenetic combinations in patients with recurrent and metastatic HNSCC. Deregulation of specific KMTs and KDMs reshapes histone methylation at key residues, thereby controlling cell cycle progression, epithelial-mesenchymal transition (EMT), stem cell phenotypes, DNA damage responses, and interactions with the immune system in HNSCC. Targeting disrupted histone methylation pathways may partially reverse epigenetic reprogramming of HNSCC cells and represents a promising strategy to improve treatment efficacy in advanced disease. We also summarize preclinical evidence and limited clinical data on targeting histone methylation in HNSCC and discuss therapeutic implications.