Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
Key Messages
- What is already known on this topic?
- 2.
- What this study adds
- 3.
- How this study might affect research, practice, or policy
1. Introduction
2. Methodology
2.1. Setting
2.2. Navrongo Health and Demographic Surveillance System
2.3. Study Design
2.4. Case Ascertainment
2.5. Variable Definitions
- The case definition of pneumonia was based on the cause of death identified through verbal autopsies. According to the WHO (2022), the case definition for pneumonia includes recognising respiratory symptoms such as cough, difficulty breathing, rapid breathing, chest pain, and abnormal lung sounds during interviews with family members or caregivers of the deceased. These symptoms are used to classify the cause of death as pneumonia when there is no clinical or laboratory confirmation.
- The WHO case definition for meningitis includes a sudden onset of fever, neck stiffness, altered consciousness, and other meningeal signs (e.g., headache, confusion, photophobia). In infants: bulging fontanelle, unusual behaviour (irritability), or poor feeding.
2.6. Data Sources
2.7. Outcome Definitions and Ascertainment
2.8. Exposure Definitions and Ascertainment
2.9. Data Analysis
2.10. Study Design
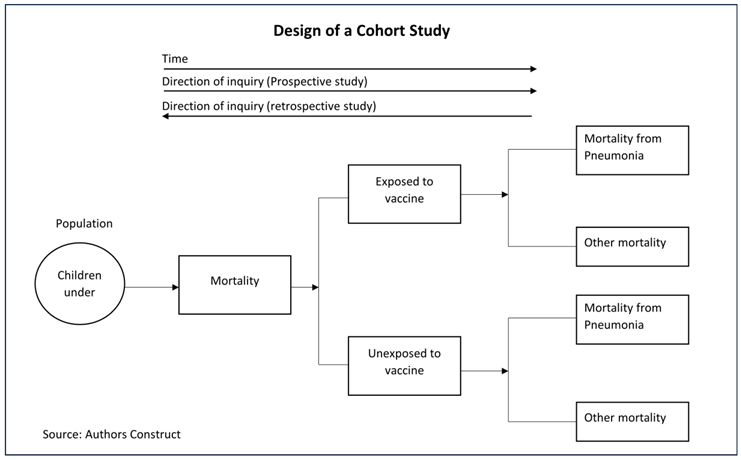
3. Results
3.1. Background Characteristics
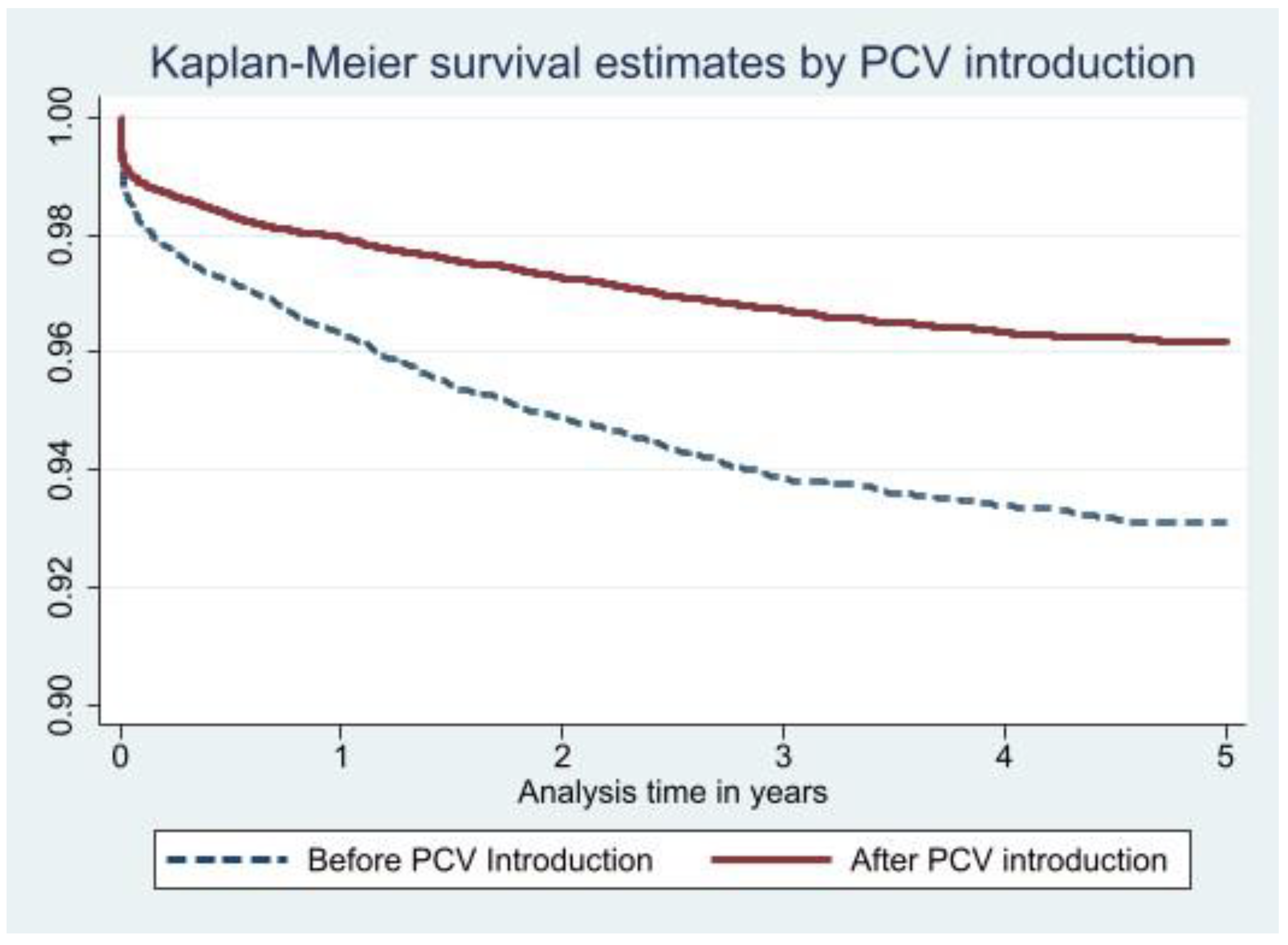
Objective 1: All-cause mortality in children under five before and after PCV13 introduction
3.2. Mortality Due to Pneumonia, Meningitis, and Diarrhoea Before and After PCV13 Immunisation
3.3. Mortality Rate of Pneumonia in Children Under Five Before and After PCV13 Immunisation
3.4. Mortality Rate of Meningitis in Children Under Five Before and After PCV13 Vaccination
Objective 2: Cause-specific pneumonia and meningitis under-five death rates since the introduction of PCV-13
Objective 3: To estimate vaccine coverage of PCV-13 in children under five in the Kassena Nankana districts
| Survey Year | PCV1 | PCV2 | PCV3 | Rota1 | Rota2 |
|---|---|---|---|---|---|
| 2013 | 25.3 | 23.4 | 19.6 | 25.8 | 21.2 |
| 2014 | 94.1 | 92.9 | 85.9 | 93.9 | 90.9 |
| 2015 | 97.2 | 97 | 95.4 | 96.6 | 96.2 |
| 2016 | 96.8 | 96.5 | 95.4 | 96.3 | 95.7 |
| 2017 | 96.1 | 96.1 | 95.7 | 95.6 | 95.8 |
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| NHDSS | Navrongo Health and Demographic Surveillance System |
| EPI | Expanded Programme on Immunisation |
| PCV | Pneumococcal Vaccine |
| MCV | Measles Containing Vaccine |
References
- Sanyang, Y. Prevalence of under-five years of age mortality by infectious diseases in West African region. Int. J. Afr. Nurs. Sci. 2019, 11. [Google Scholar] [CrossRef]
- Unicef. One is too many. Ending child deaths from pneumonia and diarrhea Every death counts. 2016.
- O’Brien, K.L.1; Wolfson, L.J.; Watt, J.P.; Henkle, E.; Deloria-Knoll, M.; McCall, N.; Lee, E.; Mulholland, K.; Levine, O.S. C.T. Hib and Pneumococcal Global Burden of Disease Study Team. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates; Lancet, 2009. [Google Scholar]
- Centers for Disease Control and Prevention. Vaccine preventable deaths and the Global Immunization Vision and Strategy. In MMWR Morb Mortal Wkly Rep.; 2006. [Google Scholar]
- WHO. Pneumonia. 2019. [Google Scholar]
- World Health Organization. Pneumococcal conjugate vaccine for childhood immunization–WHO position paper. Wkly. Epidemiol. Rec. PubMed. 2007, 82, 93–104. [Google Scholar]
- O’Brien, K.L.; Wolfson, L.J.; Watt, J.P.; Henkle, E.; Deloria-Knoll, M.; et al. Burden of disease caused by Streptococcus pneumoniae in children younger than 5 years: global estimates. Lancet 2009, 374, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.M.; Owusu, R.; Nonvignon, J. Sustainability of pneumococcal conjugate vaccination in Ghana: a cost-effectiveness analysis in the context of donor transition. Front Public Health 2024, 12. [Google Scholar] [CrossRef] [PubMed]
- Robbins, J.B.; Schneerson, R.; Gotschlich, E.C.; et al. Meningococcal meningitis in sub-Saharan Africa: the case for mass and routine vaccination with available polysaccharide vaccines. Bull. World Health Organ. 2003, 81, 745–50. [Google Scholar]
- 2019 Pneumonia & Diarrhea Progress Report Progress Report Card.
- VIEW-hub Report: Global Vaccine Introduction and Implementation J A N U A R Y 2 0 2 3 Introduction Trends Over Time.
- Gatera, M.; Uwimana, J.; Manzi, E.; et al. Use of administrative records to assess pneumococcal conjugate vaccine impact on pediatric meningitis and pneumonia hospitalizations in Rwanda. PubMed 2016, 34. [Google Scholar] [CrossRef]
- Izu, A.; Solomon, F.; Nzenze, S.A.; et al. Pneumococcal conjugate vaccines and hospitalization of children for pneumonia: a time-series analysis, South Africa, 2006–2014; Bull World Health Organ. PubMed, 2017; p. 95. [Google Scholar]
- Moïsi, J.; Alassani, I.; Tall, H.; et al. Using the indirect cohort approach to estimate PCV13 effectiveness against meningitis and pneumonia endpoints in Northern Togo. Program and abstracts of the 10th International Symposium on Pneumonia and Pneumococcal Diseases (Glasgow, Scotland), Geneva, Switzerland, 2016; p. 181. [Google Scholar]
- McCollum, E.D.; Nambiar, B.; Deula, R.; et al. Impact of the 13-valent pneumococcal conjugate vaccine on clinical and hypoxemic childhood pneumonia over three years in Central Malawi: an observational study. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Silaba, M.; Ooko, M.; Bottomley, C.; et al. The impact of 10-valent pneumococcal conjugate vaccine (PCV10) on the incidence of radiologically confirmed pneumonia and on pneumonia hospitalizations among children in Kilifi, Kenya. Program and abstracts of the 10th International Symposium on Pneum, 2016. [Google Scholar]
- Oduro, A.R.; Wak, G.; Azongo, D.; et al. Profile of the Navrongo health and demographic surveillance system. Int. J. Epidemiol. 2012, 41, 968–76. [Google Scholar] [CrossRef]
- THE NHDSS OF THE NAVRONGO HEALTH RESEARCH CENTRE - NAVRONGO HEALTH RESEARCH CENTRE.
- Waight, P.A.; Andrews, N.J.; Ladhani, S.N.; et al. Effect of the 13-valent pneumococcal conjugate vaccine on invasive pneumococcal disease in England and Wales 4 years after its introduction: an observational cohort study. Lancet Infect. Dis. 2015, 15, 535–43. [Google Scholar] [CrossRef]
- Moore, M.R.; Link-Gelles, R.; Schaffner, W.; et al. Effectiveness of 13-valent pneumococcal conjugate vaccine for prevention of invasive pneumococcal disease in children in the USA: a matched case-control study. Lancet Respir. Med. 2016, 4, 399–406. [Google Scholar] [CrossRef]
- Deceuninck, G.; Brousseau, N.; Lefebvre, B.; et al. Effectiveness of thirteen-valent pneumococcal conjugate vaccine to prevent serotype 3 invasive pneumococcal disease in Quebec in children, Canada. Vaccine 2023, 41, 5486–9. [Google Scholar] [CrossRef]
- Kambiré, D.; Soeters, H.M.; Ouédraogo-Traoré, R.; et al. Early impact of 13-valent pneumococcal conjugate vaccine on pneumococcal meningitis—Burkina Faso, 2014–2015. J. Infect. 2018, 76, 270–9. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Ang, G.; Akksilp, K.; et al. Re-evaluating the impact and cost-effectiveness of pneumococcal conjugate vaccine introduction in 112 low-income and middle-income countries in children younger than 5 years: a modelling study. Lancet Glob. Health 2024, 12, e1485–97. [Google Scholar] [CrossRef]
- Aku, F.Y. Meningitis outbreak caused by vaccine-preventable bacterial pathogens—northern Ghana, 2016. MMWR Morb. Mortal. Wkly. Rep. 2017, 66. [Google Scholar] [CrossRef]
- Faye, P.M.; Sonko, M.A.; Diop, A.; et al. Impact of 13-Valent Pneumococcal Conjugate Vaccine on Meningitis and Pneumonia Hospitalizations in Children aged . Clin. Infect. Dis. 2019, 69, S66–71. [Google Scholar] [CrossRef] [PubMed]
- Use of administrative records to assess pneumococcal conjugate vaccine impact on pediatric meningitis and pneumonia hospitalizations in Rwanda - ScienceDirect.
- Dayie, N.T.K.D.; Tettey, E.Y.; Newman, M.J.; et al. Pneumococcal carriage among children under five in Accra, Ghana, five years after the introduction of pneumococcal conjugate vaccine. BMC Pediatr. 2019, 19, 316. [Google Scholar] [CrossRef]
- Gatera, M.; Uwimana, J.; Manzi, E.; et al. Use of administrative records to assess pneumococcal conjugate vaccine impact on pediatric meningitis and pneumonia hospitalizations in Rwanda. Vaccine 2016, 34, 5321–8. [Google Scholar] [CrossRef] [PubMed]
- Izu, A.; Solomon, F.; Nzenze, S.A.; et al. Pneumococcal conjugate vaccines and hospitalization of children for pneumonia: A time-series analysis, South Africa, 2006–2014. Bull. World Health Organ. 2017, 95, 618–28. [Google Scholar] [CrossRef]
- Moïsi, J.; Alassani, I.; Tall, H. Using the indirect cohort approach to estimate PCV13 effectiveness against meningitis and pneumonia endpoints in Northern Togo; ISPPD: Geneva, Switzerland, 2016; p. 181. [Google Scholar]
- McCollum, E.D.; Nambiar, B.; Deula, R.; et al. Impact of the 13-valent pneumococcal conjugate vaccine on clinical and hypoxemic childhood pneumonia over three years in central Malawi: An observational study. PLoS ONE 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Silaba, M.; Ooko, M.; Bottomley, C. The impact of 10-valent pneumococcal conjugate vaccine (PCV10) on the incidence of radiologically confirmed pneumonia and on pneumonia hospitalizations among children in Kilifi, Kenya. Program and abstracts of the 10th International Symposium on Pneumonia and Pneumococcal Diseases (Glasgow, Scotland, 2016. [Google Scholar]
- Kambiré, D.; Soeters, H.M.; Ouédraogo-Traoré, R.; et al. Early impact of 13-valent pneumococcal conjugate vaccine on pneumococcal meningitis—Burkina Faso, 2014–2015. J. Infect. 2018, 76, 270–9. [Google Scholar] [CrossRef] [PubMed]
| Variable | Number (%) | Death of child (%) |
|---|---|---|
| Sex children | ||
| Female | 24,064 (49.8) | 858 (3.6) |
| Male | 24,287 (50.2) | 1,014 (4.2) |
| Mother’s Age | ||
| <19 | 5,919 (12.2) | 243(4.1) |
| 20-34 | 33,295 (68.9) | 1,170(3.5) |
| 35+ | 9,137 (18.9) | 459(5.0) |
| Maternal Education | ||
| No formal Education | 10,245(21.2) | 549(5.4) |
| Primary/JSS | 26,370(54.5) | 1031(7.8) |
| Secondary/Tertiary | 9,025(18.7) | 229(4.8) |
| Missing | 2,711(5.6) | 63(2.3) |
| Place of Residence of mother | ||
| Urban | 7,316 (15.1) | 221 (3.0) |
| Rural | 41,035 (84.9) | 1,651 (4.0) |
| Delivery Place | ||
| Health Facility | 32,560 (67.3) | 1,137(3.5) |
| Home/Elsewhere | 8,511 (17.6) | 576(6.8) |
| Missing | 7,280 (15.1) | 159(2.2) |
| Wealth Quantiles | ||
| Poorest | 12,177(28.3) | 591(4.9) |
| Poorer | 9,886(23.0) | 412(4.2) |
| Poor | 8,054(18.7) | 334(4.2) |
| Less Poor | 8,051(18.7) | 268(3.3) |
| Least poor | 4,881(11.3) | 125(2.6) |
| Multiple | ||
| Singleton | 46,022(95.2) | 1,718(3.7) |
| Multiple | 2,329(4.8) | 154(6.6) |
| Period | ||
| Before | 23,002(47.6) | 1,232(5.4) |
| After | 25,349(52.4) | 640(2.5) |
| Total | 48,351 | 1,872(3.9) |
| Variable | Before PCV13 immunization (2007-2012) | After PCV13 immunization (2012-2017) | ||
|---|---|---|---|---|
| Deaths | MR (95%CI) | Deaths | MR (95%CI) | |
| Sex | ||||
| Female | 470 | 19.8 (18.1 - 21.7) | 338 | 7.6 (6.9 - 8.4) |
| Male | 559 | 23.4 (21.5 - 25.4) | 455 | 8.8 (8.1 - 9.7) |
| Mother’s Age | ||||
| <19 | 139 | 26.6 (22.5 - 31.4) | 104 | 8.9 (7.3 - 10.8) |
| 20-34 | 619 | 19.4 (18.0 - 21.0) | 551 | 7.8 (7.2 - 8.5) |
| 35+ | 271 | 25.6 (22.7 - 28.8) | 188 | 9.2 (7.9 - 10.6) |
| Maternal Education | ||||
| No formal Education | 356 | 24.5 (22.0 - 27.1) | 193 | 9.2 (8.0 - 10.5) |
| Primary/JSS | 549 | 42.5 (37.7 - 48.0) | 482 | 16.7 (14.7 - 18.9) |
| Secondary/Tertiary | 110 | 32.0 (24.2 - 42.3) | 119 | 11.3 (8.7 - 14.8) |
| Missing | 14 | 39.4 (23.3 - 66.5) | 49 | 11.6 (8.7 - 15.3) |
| Place of Residence | ||||
| Urban | 121 | 18.7(15.7 - 22.4) | 100 | 6.5(5.4 - 7.9) |
| Rural | 908 | 22.0(20.6 - 23.5) | 743 | 8.5(7.9 - 9.2) |
| Delivery Place | ||||
| Health Facility | 482 | 21.1(19.3 - 23.1) | 655 | 8.6(8.0 - 9.3) |
| Home/Elsewhere | 445 | 23.4(21.3 - 25.6) | 131 | 9.3(7.8 - 11.0) |
| Missing | 102 | 17.7(14.6 - 21.5) | 57 | 4.6(3.5 - 5.9) |
| Wealth Quantiles | ||||
| Poorest | 331 | 26.1(23.4 - 29.1) | 260 | 10.1(8.9 - 11.4) |
| Poorer | 225 | 22.3(19.6 - 25.4) | 187 | 8.9(7.7 - 10.3) |
| Poor | 175 | 21.5(18.6 - 25.0) | 159 | 9.3(8.0 - 10.9) |
| Less Poor | 138 | 17.6(14.9 - 20.8) | 130 | 7.5(6.3 - 8.9) |
| least poor | 61 | 13.9(10.8 - 17.9) | 64 | 6.0(4.7 - 7.6) |
| Multiple | ||||
| Singleton | 946 | 20.7(19.5 - 22.1) | 772 | 7.9(7.3 - 8.5) |
| Multiple | 83 | 40.8(32.9 - 50.6) | 71 | 16.1(12.8 - 20.4) |
| Period | ||||
| Before | 1,029 | 21.6(20.3 - 22.9) | 203 | 4.8(4.2 - 5.5) |
| After | 640 | 10.6(9.8 - 11.5) | ||
| Cause of Death | Before PCV13 introduction | After PCV13 introduction | ||
|---|---|---|---|---|
| Death | MR (95%CI)/1000 pyrs | Death | MR (95%CI)/1000 pyrs | |
| Pneumonia | 54 | 11.3(8.7 - 14.8) | 88 | 5.9(4.8 - 7.2) |
| Meningitis | 17 | 3.6(2.2 - 5.7) | 31 | 2.1(1.5 - 2.9) |
| Pneumonia and Meningitis | 71 | 14.9(11.8 - 18.8) | 119 | 7.9(6.6 - 9.5) |
| Diarrhoea | 73 | 15.3(12.2 - 19.3) | 107 | 7.1(5.9 - 8.6) |
| Variable | Before PCV13 immunization | After PCV13 immunization | ||
|---|---|---|---|---|
| Deaths | MR (95%CI) | Deaths | MR (95%CI) | |
| Sex | ||||
| Female | 27 | 1.1(0.8 - 1.7) | 45 | 0.6(0.4 - 0.8) |
| Male | 27 | 1.1(0.8 - 1.6) | 43 | 0.6(0.4 - 0.8) |
| mother’s Age | ||||
| <19 | 5 | 1.0(0.4 - 2.3) | 11 | 0.7(0.4 - 1.2) |
| 20-34 | 35 | 1.1(0.8 - 1.5) | 56 | 0.5(0.4 - 0.7) |
| 35+ | 14 | 1.3(0.8 - 2.2) | 21 | 0.7(0.4 - 1.0) |
| Maternal Education | ||||
| No formal Education | 20 | 1.4(0.9 - 2.1) | 27 | 0.8(0.5 - 1.1) |
| Primary/JSS | 25 | 1.8(1.0 - 3.3) | 44 | 1.0(0.7 - 1.6) |
| Secondary/Tertiary | 8 | 2.2(0.8 - 6.6) | 11 | 0.8(0.3 - 2.0) |
| Missing | 1 | 2.8(0.4 - 20.0) | 6 | 1.3(0.6 - 2.9) |
| Place of Residence | ||||
| Urban | 8 | 1.2(0.6 - 2.5) | 13 | 0.6(0.3 - 1.0) |
| Rural | 46 | 1.1(0.8 - 1.5) | 75 | 0.6(0.5 - 0.7) |
| Delivery Place | ||||
| Health Facility | 24 | 1.0(0.7 - 1.6) | 52 | 0.5(0.4 - 0.7) |
| Home/Elsewhere | 23 | 1.2(0.8 - 1.8) | 28 | 0.8(0.6 - 1.2) |
| Missing | 7 | 1.2(0.6 - 2.6) | 8 | 0.4(0.2 - 0.9) |
| Wealth Quantiles | ||||
| Poorest | 17 | 1.3(0.8 - 2.2) | 27 | 0.7(0.5 - 1.0) |
| Poorer | 12 | 1.2(0.7 - 2.1) | 20 | 0.6(0.4 - 1.0) |
| Poor | 8 | 1.0(0.5 - 2.0) | 13 | 0.5(0.3 - 0.9) |
| Less Poor | 6 | 0.8(0.3 - 1.7) | 10 | 0.4(0.2 - 0.7) |
| least poor | 7 | 1.6(0.8 - 3.4) | 12 | 0.8(0.5 - 1.4) |
| Multiple | ||||
| Singleton | 51 | 1.1(0.9 - 1.5) | 81 | 0.6(0.5 - 0.7) |
| Multiple | 3 | 1.5(0.5 - 4.6) | 7 | 1.1(0.5 - 2.3) |
| Variable | Before PCV13 immunization | After PCV13 immunization | ||
|---|---|---|---|---|
| Deaths | MR (95%CI) | Deaths | MR (95%CI) | |
| Sex | ||||
| Female | 10 | 0.4(0.2 - 0.8) | 17 | 0.2(0.1 - 0.4) |
| Male | 7 | 0.3(0.1 - 0.6) | 14 | 0.2(0.1 - 0.3) |
| Mother’s Age | ||||
| <19 | 2 | 0.4(0.1 - 1.5) | 6 | 0.4(0.2 - 0.8) |
| 20-34 | 11 | 0.3(0.2 - 0.6) | 20 | 0.2(0.1 - 0.3) |
| 35+ | 4 | 0.4(0.1 - 1.0) | 5 | 0.2(0.1 - 0.4) |
| Maternal Education | ||||
| No formal Education | 5 | 0.3(0.1 - 0.8) | 8 | 0.2(0.1 - 0.4) |
| Primary/JSS | 8 | 0.7(0.2 - 1.7) | 17 | 0.4(0.2 - 0.8) |
| Secondary/Tertiary | 3 | 0.7(0.2 - 2.0) | 4 | 0.2(0.1 - 0.6) |
| Missing | 1 | 2.8(0.4 - 20.0) | 2 | 0.4(0.1 - 1.7) |
| Place of Residence | ||||
| Urban | 2 | 0.3(0.1 - 1.2) | 3 | 0.1(0.0 - 0.4) |
| Rural | 15 | 0.4(0.2 - 0.6) | 28 | 0.2(0.2 - 0.3) |
| Delivery Place | ||||
| Health Facility | 7 | 0.3(0.1 - 0.6) | 20 | 0.2(0.1 - 0.3) |
| Home/Elsewhere | 9 | 0.5(0.2 - 0.9) | 9 | 0.3(0.1 - 0.5) |
| Missing | 1 | 0.2(0.0 - 1.2) | 2 | 0.1(0.0 - 0.4) |
| Wealth Quantiles | ||||
| Poorest | 5 | 0.4(0.2 - 0.9) | 10 | 0.3(0.1 - 0.5) |
| Poorer | 3 | 0.3(0.1 - 0.9) | 5 | 0.2(0.1 - 0.4) |
| Poor | 3 | 0.4(0.1 - 1.1) | 4 | 0.2(0.1 - 0.4) |
| Less Poor | 4 | 0.5(0.2 - 1.4) | 6 | 0.2(0.1 - 0.5) |
| least poor | 1 | 0.2(0.0 - 1.6) | 3 | 0.2(0.1 - 0.6) |
| Multiple | ||||
| Singleton | 17 | 0.4(0.2 - 0.6) | 30 | 0.2(0.1 - 0.3) |
| Multiple | 0 | 0 | 1 | 0.2(0.0 - 1.1) |
| Variable | Before PCV13 immunization | After PCV13 immunization | ||
|---|---|---|---|---|
| Deaths | MR (95%CI) | Deaths | MR (95%CI) | |
| Sex | ||||
| Female | 37 | 1.6(1.1 - 2.2) | 62 | 0.8(0.6 - 1.1) |
| Male | 34 | 1.4(1.0 - 2.0) | 57 | 0.8(0.6 - 1.0) |
| Mother’s Age | ||||
| <19 | 7 | 1.3(0.6 - 2.8) | 17 | 1.0(0.6 - 1.6) |
| 20-34 | 46 | 1.4(1.1 - 1.9) | 76 | 0.7(0.6 - 0.9) |
| 35+ | 18 | 1.7(1.1 - 2.7) | 26 | 0.8(0.6 - 1.2) |
| Maternal Education | ||||
| No formal Education | 25 | 1.7(1.2 - 2.5) | 35 | 1.0(0.7 - 1.4) |
| Primary/JSS | 33 | 2.5(1.5 - 4.1) | 61 | 1.4(1.0 - 2.1) |
| Secondary/Tertiary | 11 | 2.9(1.3 -7.4) | 15 | 1.0(0.5 - 2.3) |
| Missing | 2 | 5.6(1.4 - 22.5) | 8 | 1.7(0.9 - 3.5) |
| Place of Residence | ||||
| Urban | 10 | 1.5(0.8 - 2.9) | 16 | 0.7(0.5 - 1.2) |
| Rural | 61 | 1.5(1.2 - 1.9) | 103 | 0.8(0.7 - 1.0) |
| Delivery Place | ||||
| Health Facility | 31 | 1.4(1.0 - 1.9) | 72 | 0.7(0.6 - 0.9) |
| Home/Elsewhere | 32 | 1.7(1.2 - 2.4) | 37 | 1.1(0.8 - 1.5) |
| Missing | 8 | 1.4(0.7 - 2.8) | 10 | 0.5(0.3 - 1.0) |
| Wealth Quantiles | ||||
| Poorest | 22 | 1.7(1.1 - 2.6) | 37 | 1.0(0.7 - 1.3) |
| Poorer | 15 | 1.5(0.9 - 2.5) | 25 | 0.8(0.5 - 1.2) |
| Poor | 11 | 1.4(0.7 - 2.4) | 17 | 0.7(0.4 - 1.1) |
| Less Poor | 10 | 1.3(0.7 - 2.4) | 16 | 0.6(0.4 - 1.0) |
| least poor | 8 | 1.8(0.9 - 3.7) | 15 | 1.0(0.6 - 1.7) |
| Multiple | ||||
| Singleton | 68 | 1.5(1.2 - 1.9) | 111 | 0.8(0.6 - 0.9) |
| Multiple | 3 | 1.5(0.5 - 4.6) | 8 | 1.2(0.6 - 2.5) |
| Survey Year | BCG | OPV0 | OPV1 | OPV2 | OPV3 | Penta1 | Penta2 | Penta3 | Measles 1 |
|---|---|---|---|---|---|---|---|---|---|
| 2007 | 98.9 | 70.2 | 99.3 | 98.7 | 97.8 | 99.5 | 99.2 | 98.1 | 93.2 |
| 2008 | 97.8 | 72.6 | 99.3 | 98.5 | 97.9 | 99.4 | 99.0 | 98.2 | 95.3 |
| 2009 | 99.3 | 73.6 | 99.8 | 99.5 | 98.9 | 99.9 | 99.8 | 99.3 | 96.3 |
| 2010 | 99.1 | 90.0 | 99.1 | 98.7 | 98.1 | 99.0 | 98.6 | 98.5 | 95.9 |
| 2011 | 99.6 | 93.3 | 99.8 | 99.7 | 98.8 | 99.8 | 99.7 | 99.4 | 97.3 |
| 2012 | 99.7 | 95.7 | 100.0 | 99.9 | 98.9 | 99.9 | 99.9 | 99.6 | 96.4 |
| 2013 | 99.1 | 97.2 | 99.6 | 99.4 | 97.6 | 99.6 | 99.5 | 99.0 | 95.1 |
| 2014 | 98.2 | 96.4 | 99.0 | 98.9 | 97.4 | 99.0 | 98.9 | 98.3 | 93.9 |
| 2015 | 98.6 | 97.2 | 99.0 | 99.3 | 98.2 | 99.0 | 99.2 | 99.0 | 95.1 |
| 2016 | 97.7 | 97.0 | 98.7 | 98.6 | 97.6 | 98.6 | 98.5 | 97.7 | 94.6 |
| 2017 | 97.5 | 96.3 | 98.3 | 98.2 | 97.8 | 97.7 | 97.9 | 97.5 | 94.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




