Submitted:
03 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
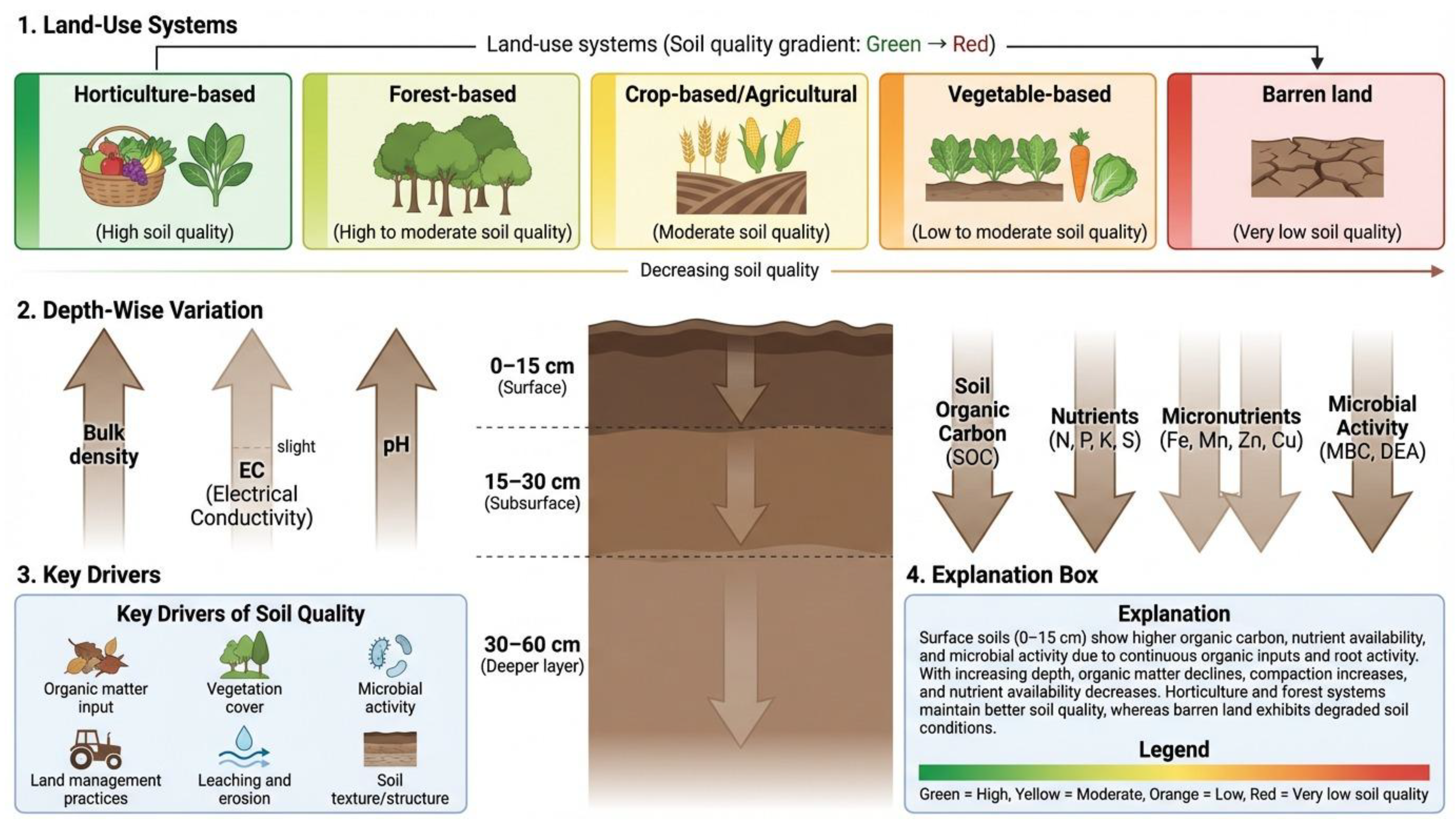
1. Introduction
2. Materials and Methods
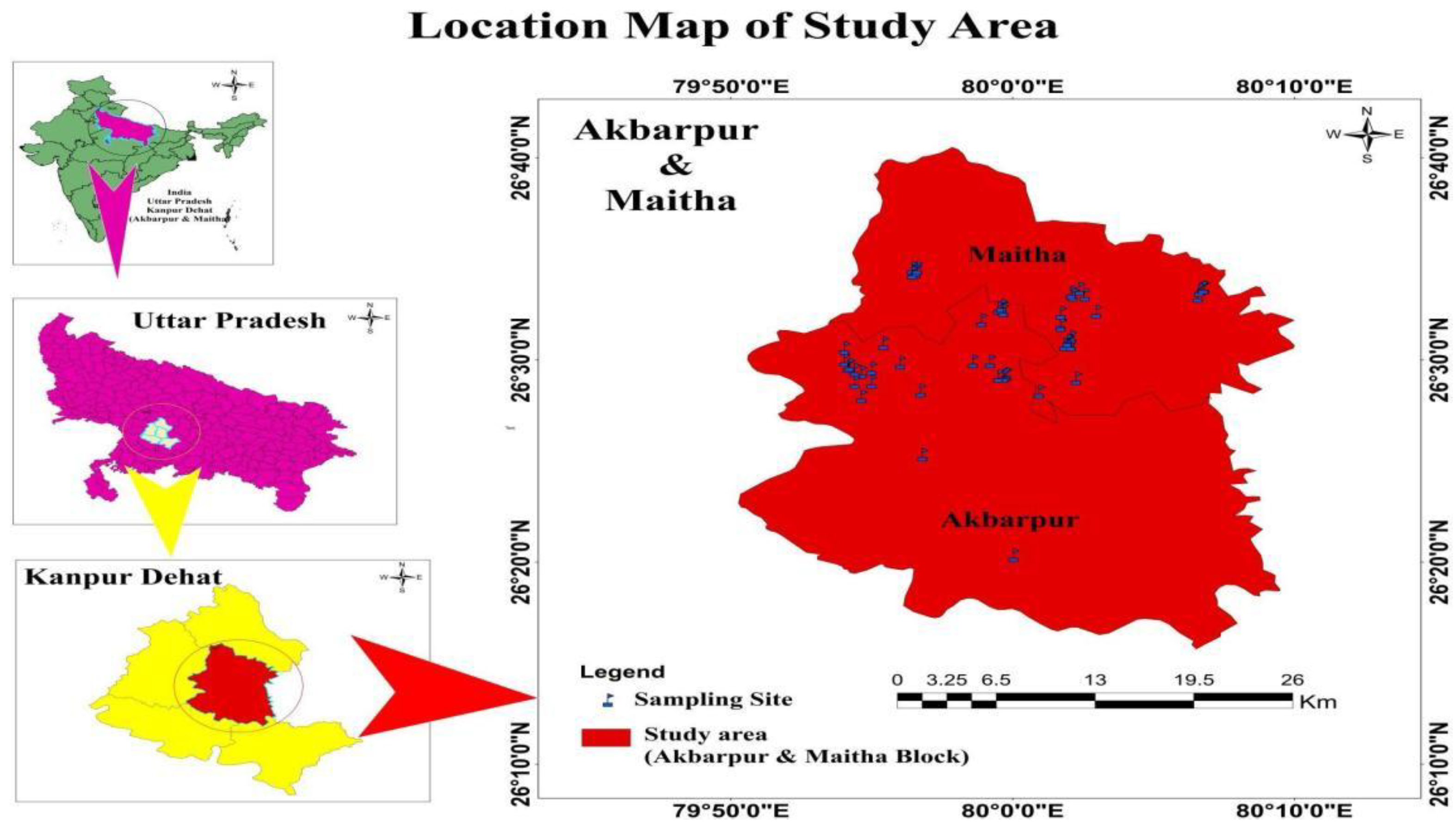
2.1. Region, Climate and Soil
2.2. Soil Sampling and Analysis
2.3. Laboratory Analysis of Soil
2.4. Analysis of Soil Organic Carbon Fraction and Stocks
2.5. Statistical Analysis
3. Result
3.1. Variation in Physico-chemical Properties of Soils across Land-use Types
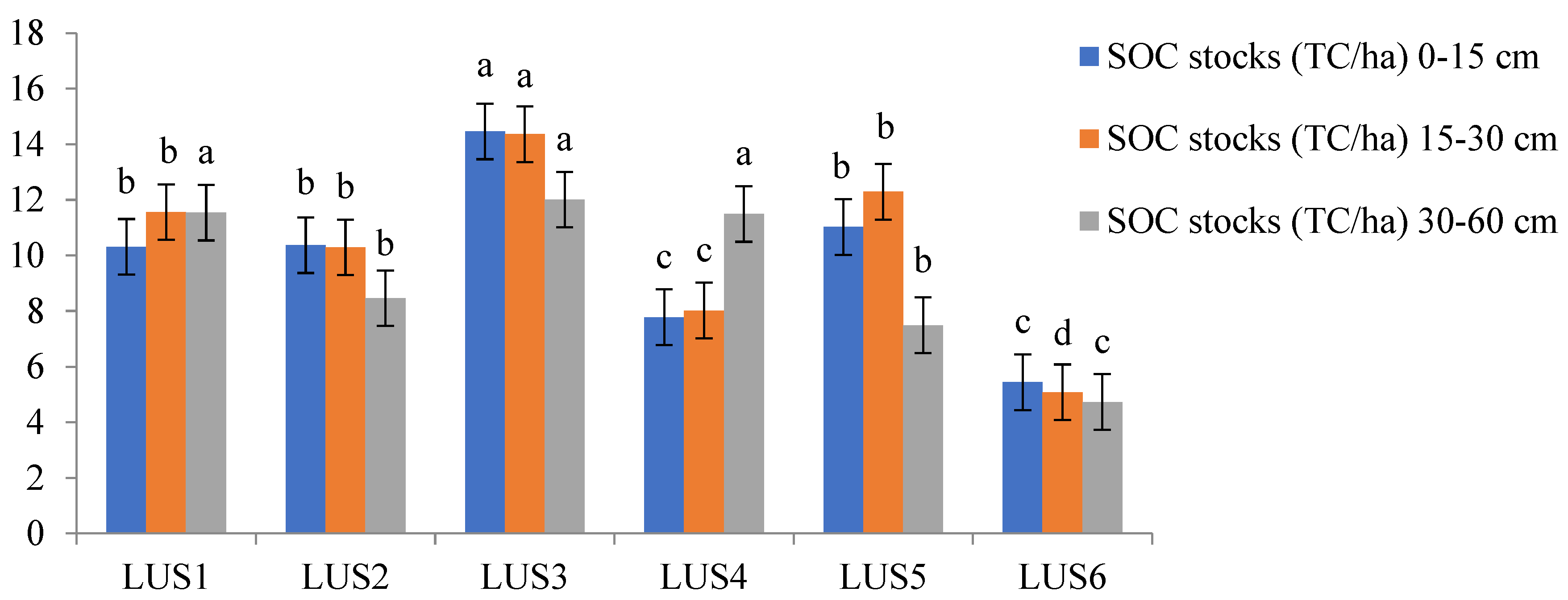
3.2. Soil Organic Carbon Fractions and Stock Dynamics in Various Land-Use Systems
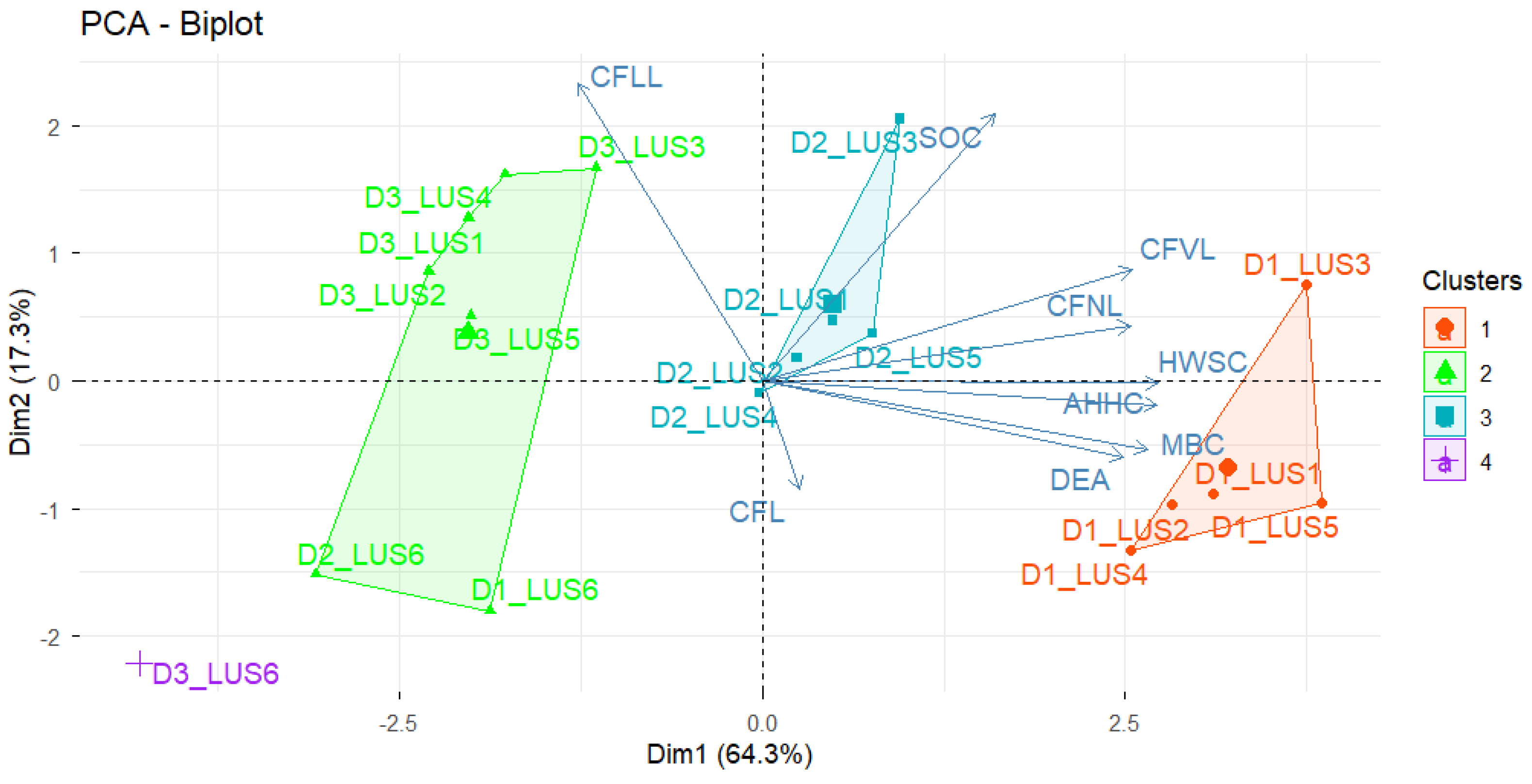
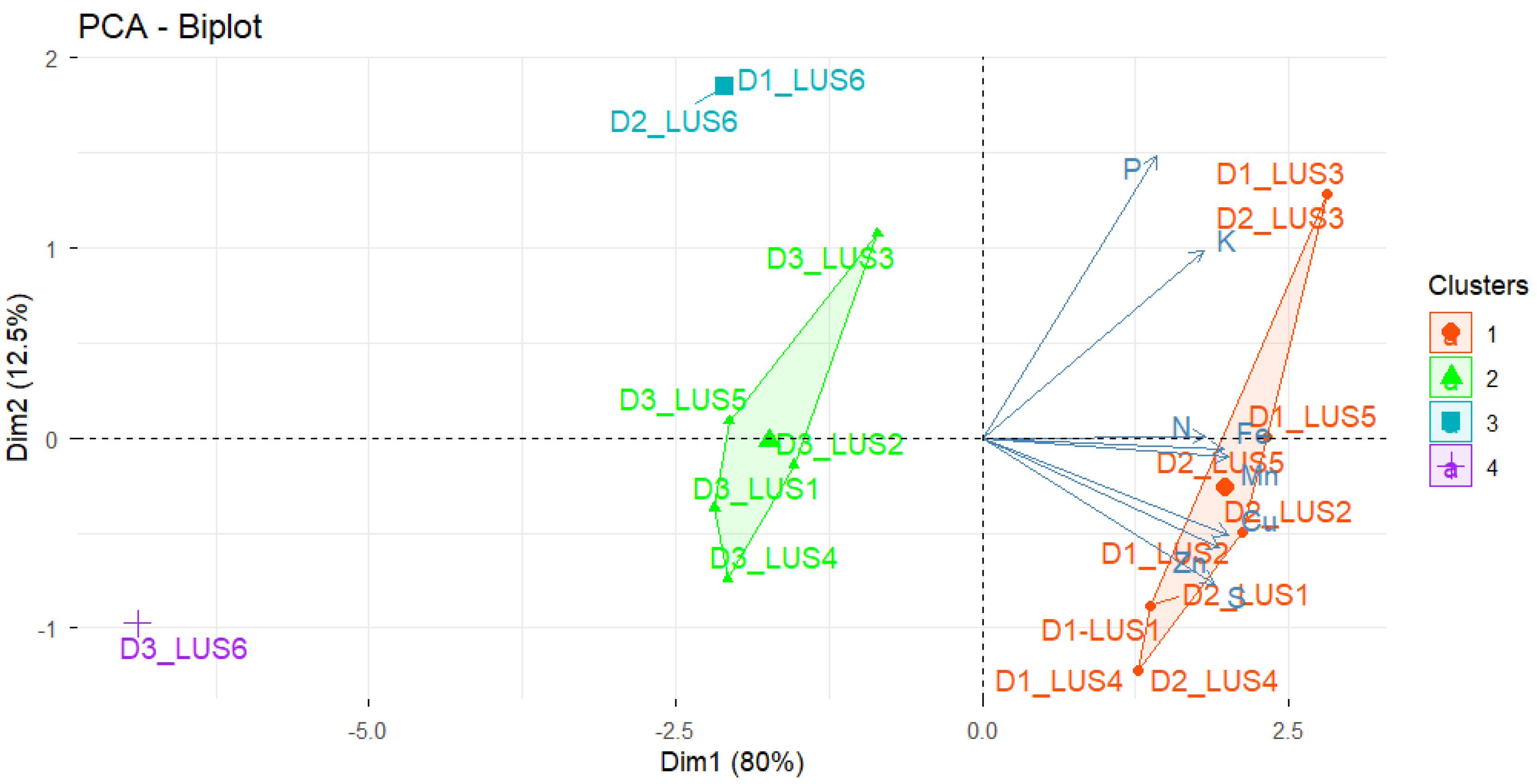
3.3. Principal Component Analysis
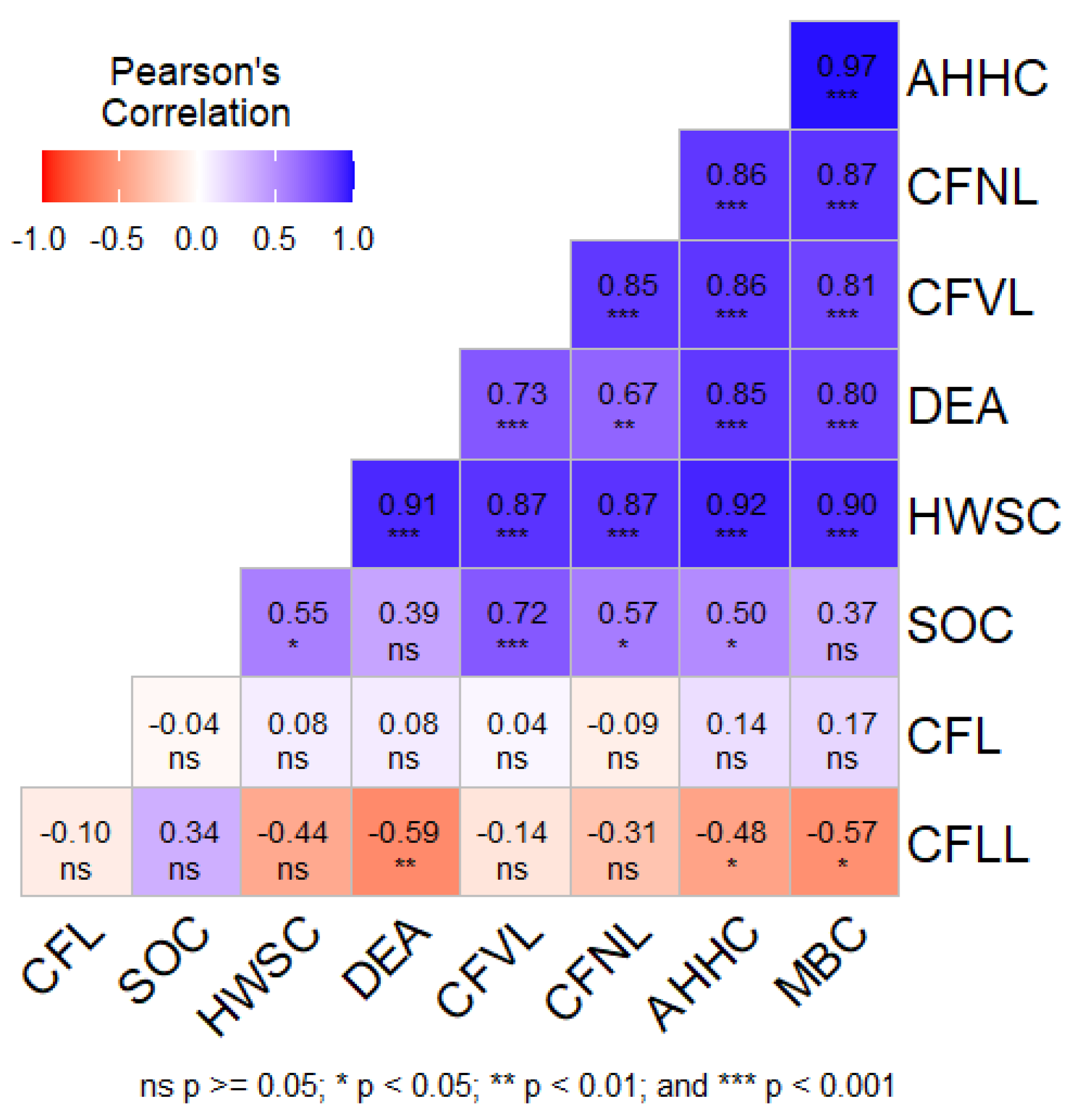
3.4. Pearson’s Correlation
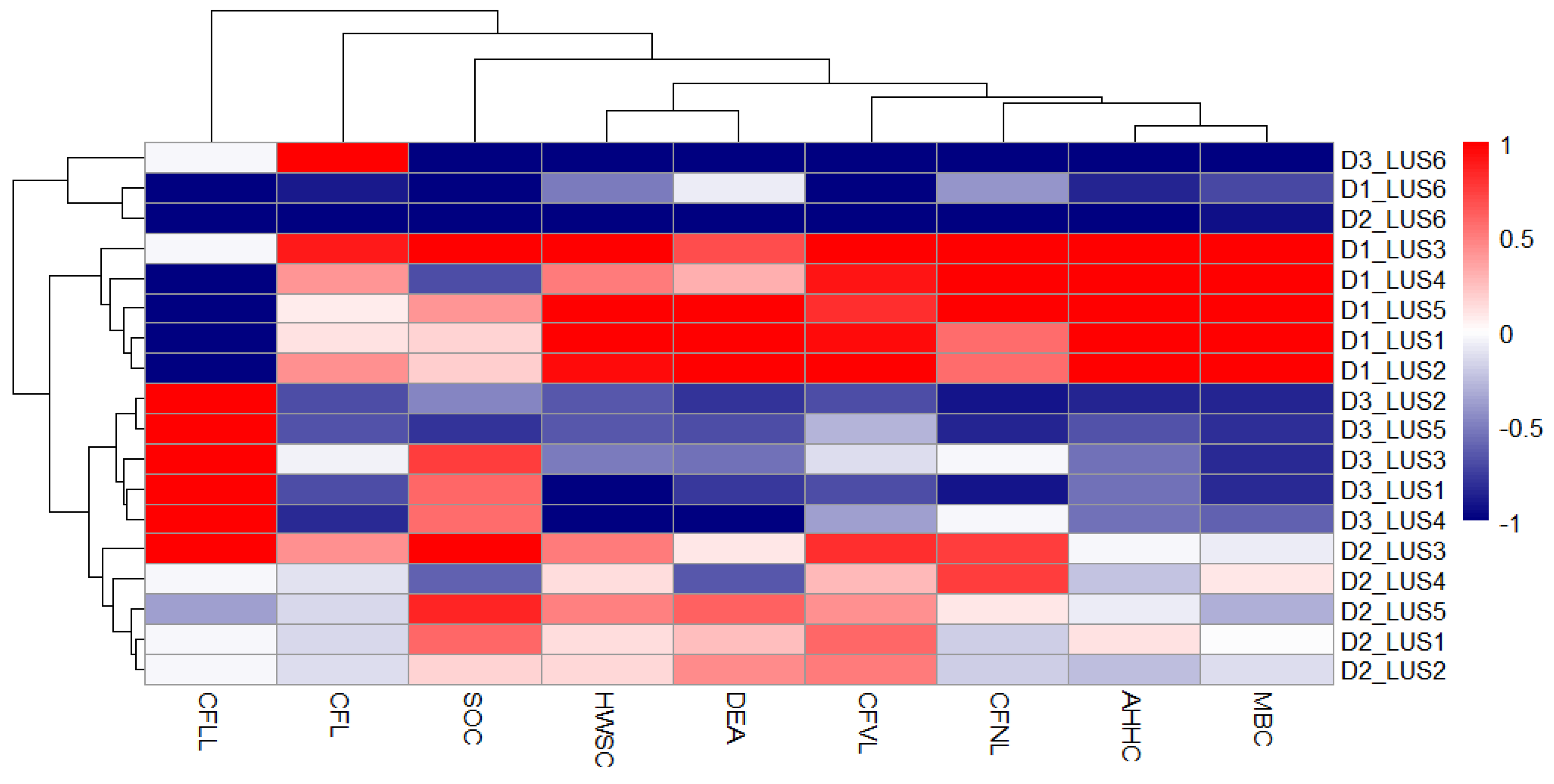
3.5. Heatmap Analysis
3.6. Nutrient Status of Soil under Various LUSs
3.7. Principal Component Analysis
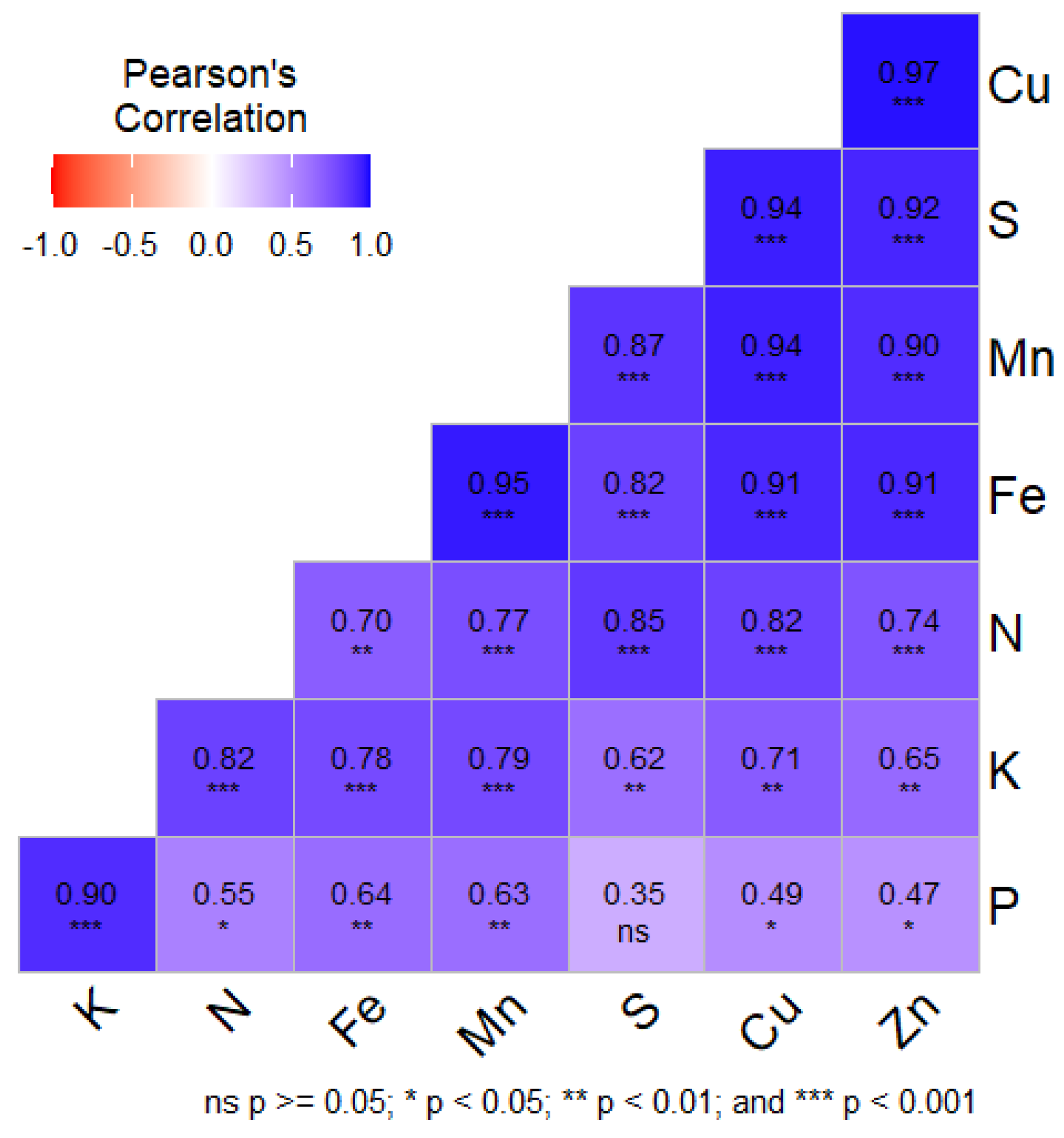
3.8. Pearson’s Correlation
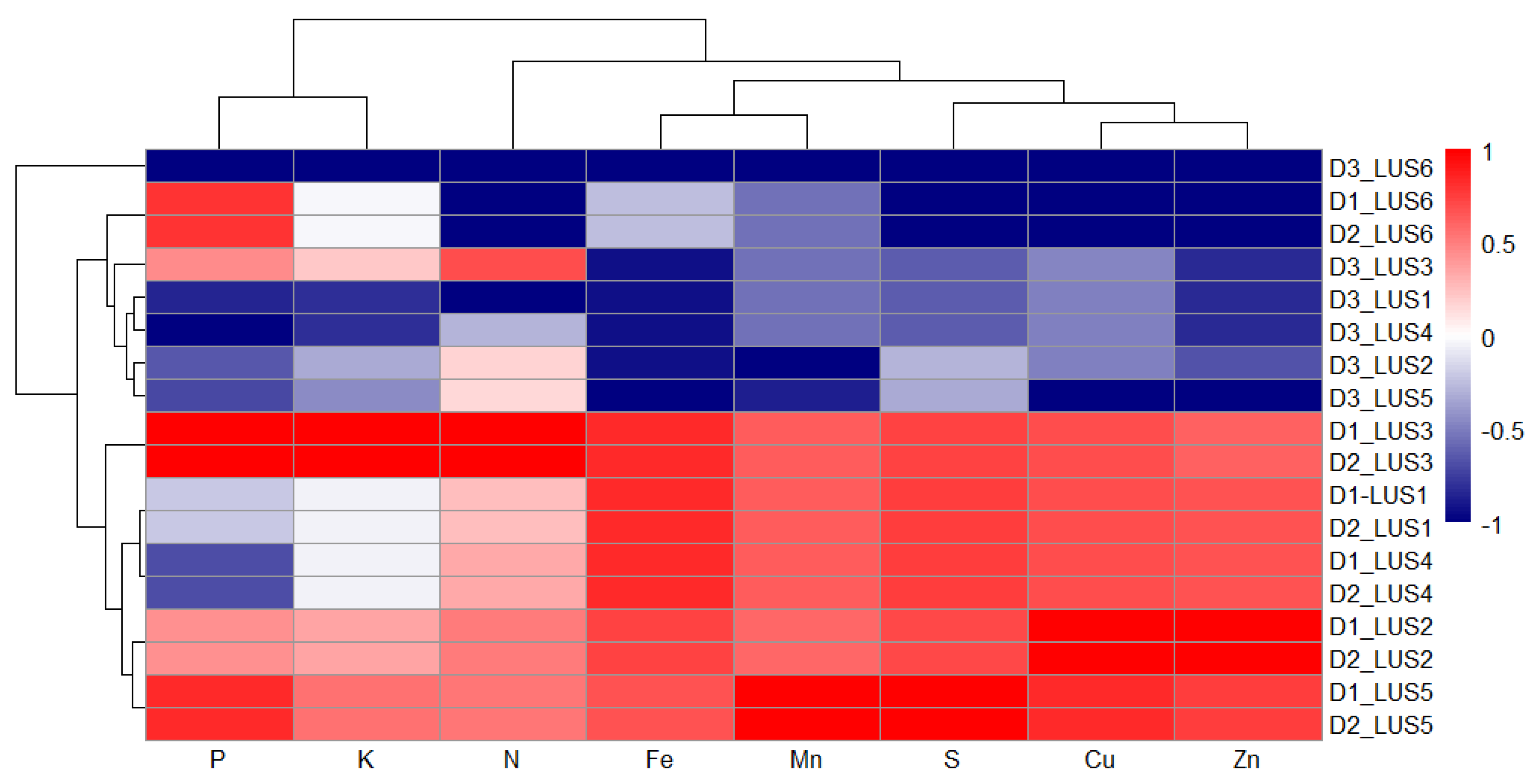
3.9. Heatmap Analysis
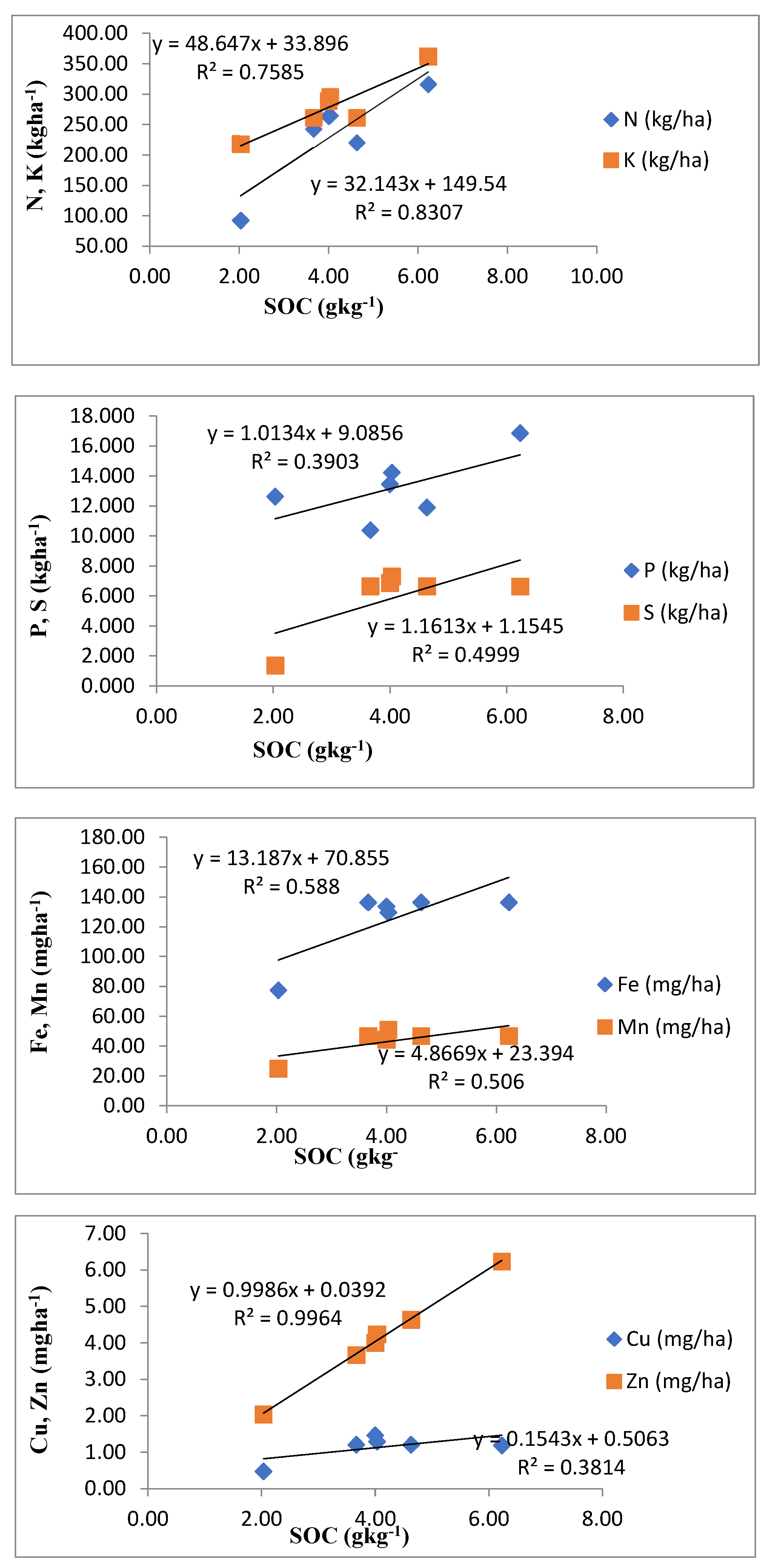
3.10. Analysis of Correlation and Linear Regression
4. Discussion
4.1. Variation in Physico-Chemical Properties of Soils Across Land-Use Types
4.2. Soil Organic Carbon Fractions and Stock Dynamics in Various Land-Use Systems
4.3. Soil Nutrient Dynamics under Different LUSs
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huang, X.; Liu, Y.; Stouffs, R. “Human-earth system dynamics in China’s land use pattern transformation amidst climate fluctuations and human activities”. Sci. Total Environ. 2024, 954, 176013. [Google Scholar] [CrossRef] [PubMed]
- DeFries, R.; Hansen, A.; Turner, B. L.; Reid, R.; Liu, Jianguo. Land use change around protected areas: management to balance human needs and ecological function. Ecol. Appl. 2007, 17(4), 1031–1038. [Google Scholar] [CrossRef]
- Naik, S. K.; Shinde, R.; Mali, S. S.; Sarkar, P. K.; Das, A. Land Degradation: A Global Challenge to Environmental Sustainability and Livelihood Security. In Ecological Solutions to Agricultural Land Degradation; Babu, S., Das, A., Rathore, S.S., Singh, R., Singh, R., Eds.; Springer: Singapore, 2025; pp. 1–27. [Google Scholar] [CrossRef]
- Singh, P.; Sharma, A.; Dhankhar, J. Climate change and soil fertility. In Plant Stress Mitigators Action and Application; Springer Nature: Singapore, 2022; pp. 25–5. [Google Scholar] [CrossRef]
- Thakur, T. K.; Swamy, S. L.; Dutta, J.; Thakur, A.; Mishra, A.; Sarangi; Kumar, R. Assessment of land use dynamics and vulnerability to land degradation in coal-mined landscapes of central India: implications for ecorestoration strategies. Front. Environ. Sci. 2024, 12, 1419041. [Google Scholar] [CrossRef]
- Tessema, M. W.; Abebe, B. G.; Bantider, A. Physical and socioeconomic driving forces of land use and land cover changes: the case of Hawassa City, Ethiopia. Front. Environ. Sci. 2024, 12, 1203529. [Google Scholar] [CrossRef]
- Baisero, D.; Visconti, P.; Pacifici, M.; Cimatti, M.; Rondinini, C. “Projected global loss of mammal habitat due to land-use and climate change”. One Earth 2020, 2(6), 578–585. [Google Scholar] [CrossRef]
- Shuai, W.; Di, S.; Zijiao, Y.; Xiang, G.; Li, C.; Zhang, Z. W. M.; Jin, X. Projecting Future Soil Organic Carbon and Soil Total Nitrogen Stocks Under Climate-Land Use Change Scenarios in Tibet, China. Environ. Sustain. Indic. 2025, 100856. [Google Scholar] [CrossRef]
- Gama, J. T. d. The role of soils in sustainability, climate change, and ecosystem services: Challenges and opportunities. Ecologies 2023, 4(3), 552–567. [Google Scholar] [CrossRef]
- Boruah. Climate Change and CO2 Geosequestration. In CO₂ Geosequestration: Capturing Carbon for a Sustainable Future: Carbon Dioxide Storage in Geological Media; Springer Nature, Switzerland: Cham, 2025; pp. 1–17. [Google Scholar] [CrossRef]
- Rumpel; Amiraslani, F.; Chenu, C.; Cardenas, M. G.; Kaonga, M.; Koutika, L. S.; Wollenberg, E. The 4p1000 initiative: Opportunities, limitations and challenges for implementing soil organic carbon sequestration as a sustainable development strategy. Ambio 2020, 49(1), 350–360. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Song, X.; Liu, Y.; Chai, L. Emerging new global soil governance structure in agrifood systems: Taking the 4 per 1,000 initiative as an example. Front. Sustain. Food Syst. 2023, 7, 1104252. [Google Scholar] [CrossRef]
- Shanmugam, S.; Dalal, R. C.; Joosten, H.; Raison, R. J.; Joo, G. K. SOC stock changes and greenhouse gas emissions following tropical land use conversions to plantation crops on mineral soils, with a special focus on oil palm and rubber plantations. Agriculture 2018, 8(9), 133. [Google Scholar] [CrossRef]
- Lorenz, K.; Lal, R.; Ehlers, K. Soil organic carbon stock as an indicator for monitoring land and soil degradation in relation to U nited N ations’ S ustainable D evelopment G oals. Land Degrad. Dev. 2019, 30(7), 824–838. Available online: https://www.researchgate.net/publication/311318647_Soil_Organic_Carbon_-_An_Appropriate_Indicator_to_Monitor_Trends_of_Land_and_Soil_Degradation_within_the_SDG_Framework. [CrossRef]
- Francaviglia, R.; Almagro, M.; Vicente-Vicente, J. L. Conservation agriculture and soil organic carbon: Principles, processes, practices and policy options. Soil. Syst. 2023, 7(1), 17. [Google Scholar] [CrossRef]
- Maqbool, Z.; Farooq, M. S.; Rafiq, A.; Uzair, M.; Hussain, Q. Utilisation of Climate-Smart Conservation Agriculture Practices for Improved Soil Carbon Sequestration, Greenhouse Gas Mitigation and Sustainable Crop Productivity. Soil. Use Manag. 2025, 41(2), e70103. [Google Scholar] [CrossRef]
- Lal, R.; Follett, R. F.; Stewart, B.A.; Kimble, J. M. Soil carbon sequestration to mitigate climate change and advance food security. Soil. Sci. 2007, 172(12), 943–956. [Google Scholar] [CrossRef]
- Filipović; Mandić, A.; Banožić, M. Perennial Crops and Their Role in Mitigating Climate Change Through Carbon Sequestration. In Agroforestry for Monetising Carbon Credits; Springer Nature, Switzerland: Cham, 2025; pp. 395–432. [Google Scholar] [CrossRef]
- Patel; Kumar, S.; Babu, S.; Singh, R. K.; Rathore, S. S.; Shekhawat, K. Ecosystem Restoration for Achieving the Land Degradation Neutrality. In Ecological Solutions to Agricultural Land Degradation; Springer Nature Singapore, 2025; pp. 283–313. [Google Scholar] [CrossRef]
- Kumar; Kumar, S. Impact of Climate Change on Himalayan Ecosystem and Traditional Crops. In Climate Change Impact on Himalayan Biodiversity; 2025; pp. 393–413. [Google Scholar] [CrossRef]
- Zolotnytska, Y.; Krzyżanowski, J.; Wigier, M.; Krupin, V.; Wojciechowska, A. Food Security Strategy for Mercosur Countries in Response to Climate and Socio-Economic Challenges. Sustainability 2025, 17(16), 7280. [Google Scholar] [CrossRef]
- Goud, R.; Raghavendra, M.; Prasad, P. S.; Hatti, V.; Halli, H. M.; Nayaka, G. V.; Rajpoot, S. K. Sustainable management and restoration of the fertility of damaged soils. Agric. Issues Polic. 2022, 113. [Google Scholar] [CrossRef]
- Abdulmanov, R.; Miftakhov, I.; Ishbulatov, M.; Galeev, E.; Shafeeva, E. Comparison of the effectiveness of GIS-based interpolation methods for estimating the spatial distribution of agrochemical soil properties. Environ. Technol. Innov. 2021, 24, 101970. [Google Scholar] [CrossRef]
- Laskar, A.; Kumar, K.; Roy, P.; Mazumder, A. S.; Das, B. Exploring the Role of Smart Systems in Farm Machinery for Soil Fertility and Crop Productivity. Int. J. Res. Appl. Sci. Eng. Technol. 2024, 12(12), 2063–2075. [Google Scholar] [CrossRef]
- Lieske, J.; Avery-Gomm, S.; Champagne, P.; Fulton, L. GIS, Remote Sensing and Machine Learning: Data Integration to Support the Management of Coastal Island Ecosystems. Environ. Manag. 2025, 1–22. [Google Scholar] [CrossRef]
- Aslam, M.; Fazal, S. Transforming India—An Appraisal of Agricultural Land Use in Uttar Pradesh: A Regional Analysis. J. Land Rural Stud. 2025, 13(1), 46–66. [Google Scholar] [CrossRef]
- Gautam, M. K.; Ghosh, A.; Ashish, L. M.; Singh, R. K.; Singh, U. P.; Maurya, A. Characterization of physico-chemical properties of red soil (Alfisol) in Mirzapur District of Uttar Pradesh. Int. J. Chem. Stud. 2018, 6(3), 1067–1073. [Google Scholar] [CrossRef]
- Nziguheba, G.; Adewopo, J.; Masso, C.; Nabahungu, N. L.; Six, J.; Sseguya, H.; Vanlauwe, B. Assessment of sustainable land use: linking land management practices to sustainable land use indicators. Int. J. Agric. Sustain. 2022, 20(3), 265–288. [Google Scholar] [CrossRef]
- Chopra, S. L.; Kanwar, J. S. Analytical agricultural chemistry; Kalyani Publishers: Ludhiana, India, 1982. [Google Scholar]
- https. Available online: https://books.google.com/books/about/Analytical_Agricultural_Chemistry.html?id=hVABywAACAAJ.
- Walkley; Black, I. A. An examination of the Degtjareff method for determining soil organic matter, and a proposed modification of the chromic acid titration method. Soil. Sci. 1934, 37(1), 29–38. [Google Scholar] [CrossRef]
- Black, A. Operator variation. Methods of Soil Analysis: Part 1 Physical and Mineralogical Properties. Incl. Stat. Meas. Sampl. 1965, 9, 50–53. [Google Scholar] [CrossRef]
- Bouyoucos, G. J. The hydrometer as a new method for the mechanical analysis of soils. Soil. Sci. 23(5), 343–54. [CrossRef]
- Ghani; Dexter, M.; Perrott, K.W. Hot-water extractable carbon in soils: a sensitive measurement for determining impacts of fertilisation, grazing and cultivation. Soil. Biol. Biochem. 2003, 35(9), 1231–1243. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K. A.; Hamilton, J. K.; Rebers, P. A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28(3), 350–356. [Google Scholar] [CrossRef]
- Oades, J. M.; Wagner, G. H. Incorporation of 14C into sugars in a soil incubated with 14C glucose. Geoderma 1970, 4(4), 417–423. [Google Scholar] [CrossRef]
- Subbiah, V.; Asija, G. L. A rapid procedure for the determination of available nitrogen in soils. Curr. Sci. 1956, 25, 259–260. [Google Scholar]
- Olsen, S. R.; Watanabe, F. S.; Cosper, H. R.; Larson, W. E.; Nelson, L. B. Residual phosphorus availability in long-time rotations on calcareous soils. Soil. Sci. 1954, 78(2), 141–152. Available online: https://ui.adsabs.harvard.edu/link_gateway/1954SoilS..78..141O/doi:10.1097/00010694-195408000-00008. [CrossRef]
- Chesnin, L.; Yien, C. H. Turbidimetric determination of available sulfates Soil Science. Soc. Am. 1951, Vol. 15, 149–151. [Google Scholar] [CrossRef]
- Lindsay, W. L.; Norvell, W. Development of a DTPA soil test for zinc, iron, manganese, and copper. Soil. Sci. Soc. Am. J. 1978, 42(3), 421–428. [Google Scholar] [CrossRef]
- Chen, Y.; Du, Z.; Weng, Z.; Sun, K.; Zhang, Y.; Liu, Q.; Van Zwieten, L. Formation of soil organic carbon pool is regulated by the structure of dissolved organic matter and microbial carbon pump efficacy: A decadal study comparing different carbon management strategies. Glob. Change Biol. 2023, 29(18), 5445–5459. [Google Scholar] [CrossRef]
- Chen, S.; Arrouays, D.; Angers, D. A.; Martin, M. P.; Walter, C. Soil carbon stocks under different land uses and the applicability of the soil carbon saturation concept. Soil. Tillage Res. 2019, 188, 53–58. [Google Scholar] [CrossRef]
- Bhattacharyya, T.; Ray, S. K.; Pal, D. K.; Chandran, P.; Mandal, C.; Wani, S. P. Soil carbon stocks in India-issues and priorities. J. Indian Soc. Soil. Sci. 2009, 57(4), 461–468. Available online: http://oar.icrisat.org/id/eprint/448.
- Webster, R. Analysis of variance, inference, multiple comparisons and sampling effects in soil research. Eur. J. Soil. Sci. 2007, 58(1), 74–82. [Google Scholar] [CrossRef]
- Kahsay; Haile, M.; Gebresamuel, G.; Mohammed, M.; Okolo, C. C. Assessing land use type impacts on soil quality: Application of multivariate statistical and expert opinion-followed indicator screening approaches. Catena 2023, 231, 107351. Available online: https://ui.adsabs.harvard.edu/link_gateway/2023Caten.23107351K/doi:10.1016/j.catena.2023.107351. [CrossRef]
- Sharma, D.; Kumar, R.; Singh, B.; Sethi, M. Micronutrients distribution in salt-affected soils of the Punjab in relation to soil properties. Arch. Agron. Soil. Sci. 2009, 55(4), 367–377. [Google Scholar] [CrossRef]
- Gautam, M. K.; Sharma, S.; Kumar, A.; Kumar, S.; Jayant, H.; Sachan, R.; Kumar, M. Assessment of Soil Fertility Status under the Barren Land Soil of the Central Plain Zone of Uttar Pradesh, India. Int. J. Environ. Clim. Change 2023, 13(10), 483–490. [Google Scholar] [CrossRef]
- Meena, R. B.; Meena, S. C.; Meena, R. H.; Alam, N. M.; Meena, R.; Nogiya, M.; Meena, G. L. Impact of land-use systems on fertility parameters and deterioration indices of soil in the sub humid Southern Plains of Rajasthan, India. J. Environ. Biol. 2023, 44(2), 238–249. Available online: https://orcid.org/0000-0003-0624-9424. [CrossRef]
- Uthappa, R.; Devakumar, A. S.; Das, B.; Mahajan, G. R.; Chavan, S. B.; Jinger, D.; Fahad, S. Comparative analysis of soil quality indexing techniques for various tree based land use systems in semi-arid India. Front. For. Glob. Change 2024, 6, 1322660. [Google Scholar] [CrossRef]
- Awoonor, J. K.; Amoako, E. E.; Dogbey, B. F.; Wiredu, I. Quantitative analysis of soil degradation in response to land use change in the Guinea savanna zone of Ghana. Geoderma Reg. 2024, 37, e00779. [Google Scholar] [CrossRef]
- Mandal, M.; Kamp, P.; Singh, M. Effect of long term manuring on carbon sequestration potential and dynamics of soil organic carbon labile pool under tropical rice-rice agro-ecosystem. Commun. Soil. Sci. Plant Anal. 2020, 51(4), 468–480. [Google Scholar] [CrossRef]
- Chauhan, R. P.; Pande, K. R.; Thakur, S. Soil properties affected by land use systems in western Chitwan, Nepal. Int. J. Appl. Sci. Biotechnol. 2014, 2(3), 265–269. [Google Scholar] [CrossRef]
- Kumar, J.; Bhardwaj, D. R.; Thakur, C. L.; Sharma, P.; Sharma, P. Soil organic carbon dynamics under different land use systems in dry temperate ecosystem of north-western Himalayas. Catena 2024, 240, 108022. [Google Scholar] [CrossRef]
- Bhatt, R.; Singh, P.; Sharma, S. Changes in soil organic pool and carbon preservation capacity of macro-and micro-aggregates in response to land-use change in north-western India. J. Soil. Sci. Plant Nutr. 2023, 23(2), 2849–2867. [Google Scholar] [CrossRef]
- Wu; Zhang, M.; Zhai, Z.; Dai, H.; Yang, M.; Zhang, Y.; Liang, T. Soil organic carbon, carbon fractions, and microbial community under various organic amendments. Sci. Rep. 2024, 14(1), 25431. [Google Scholar] [CrossRef]
- Maini; Sharma, V.; Sharma, S. Assessment of soil carbon and biochemical indicators of soil quality under rainfed land use systems in North Eastern region of Punjab, India. Carbon Manag. 2020, 11(2), 169–182. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, P.; Singh, Y. Soil Enzymatic Activity, Bacterial Diversity and Organic Carbon Pool in Response to Residue Management and Intensive Tillage in Rice-Wheat Cropping. J. Soil. Sci. Plant Nutr. 2025, 1–23. Available online: https://ui.adsabs.harvard.edu/link_gateway/2025JSSPN..25.3858S/doi:10.1007/s42729-025-02371-6. [CrossRef]
- Das; Mishra, G.; Lakra, P. C.; Kumar, S.; Mishra, S. N. Impact of land uses on soil organic carbon dynamics in the Indian Himalayan Region. In Soil Carbon Dynamics in Indian Himalayan Region; 2023; pp. 55–75. [Google Scholar] [CrossRef]
- Meena, R. B.; Meena, S. C.; Rathore, A. C.; Meena, D. C.; Meena, R. L.; Alam, N. M.; Meena, M. D. Soil carbon sequestration potential of different land use systems: evidence from sub-humid southern plains and Aravalli hills of Rajasthan, India. Environ. Monit. Assess. 2024, 196(11), 1080. [Google Scholar] [CrossRef]
- Patel; Modak, K.; Mishra, G.; Barman, T.; Giri, K.; Francaviglia, R. Monitoring of soil organic carbon pools using space-for-time substitution approach. In Forests for Inclusive and Sustainable Economic Growth; 2025; pp. 25–36. [Google Scholar] [CrossRef]
- Munny, N. N.; Khan, M. Z.; Hanif, M. D. Vertical distribution of soil properties and organic carbon under different land use systems in saline soils of Bangladesh. Environ. Chall. 2021, 4, 100097. [Google Scholar] [CrossRef]
- Geleta, W.; Fufa, F.; Bekele, A. Evaluating Selected Soil Properties under Different Land Use Types and Soil Depth at Arjo-Dhidhessa. J. Sci. Technol. Arts Res. 2025, 14(1), 32–48. [Google Scholar] [CrossRef]
- Rathore, C.; Singh, C.; Jayaprakash, J.; Gupta, A. K.; Doharey, V. K.; Jinger, D.; Madhu, M. Impact of conservation practices on soil quality and ecosystem services under diverse horticulture land use system. Front. For. Glob. Change 2023, 6, 1289325. [Google Scholar] [CrossRef]
- Ma, Y.; Chen, J.; Li, Z.; Zhou, J.; Zhang, Y.; Sun, S.; Liu, J. The effect of soil physical structure in soil carbon and nitrogen distribution under different land use types in typical forest grassland transition zone on Loess Plateau, China. Soil. Use Manag. 2025, 41(1), e70061. [Google Scholar] [CrossRef]
- Hammad, H. M.; Abbas, F.; Saeed, S.; Fahad, S.; Cerdà, A.; Farhad, W.; Bakhat, H. F. Offsetting land degradation through nitrogen and water management during maize cultivation under arid conditions. Land Degrad. Dev. 2018, 29(5), 1366–1375. [Google Scholar] [CrossRef]
- Mir, Y. H.; Ganie, M. A.; Baba, Z. A.; Mir, S.; Bhat, M. A.; Bhat, J. A.; Kuriqi, A. Assessing the impact of land use change on carbon and soil quality in Kashmir Himalayas. Turk. J. Agric. For. 2024, 48(5). [Google Scholar] [CrossRef]
- Meena, R. B.; Meena, S. C.; Meena, R. H.; Alam, N. M.; Meena, R.; Nogiya, M.; Meena, G. L. Impact of land-use systems on fertility parameters and deterioration indices of soil in the sub humid Southern Plains of Rajasthan, India. J. Environ. Biol. 2023, 44(2), 238–249. Available online: https://orcid.org/0000-0003-0624-9424. [CrossRef]
- Xu, X.; Guo, L.; Wang, S.; Wang, X.; Ren, M.; Zhao, P.; Lin, A. Effective strategies for reclamation of saline-alkali soil and response mechanisms of the soil-plant system. Sci. Total Environ. 2023, 905, 167179. [Google Scholar] [CrossRef]
- Sharma, B. D.; Kumar, R.; Singh, B.; Sethi, M. Micronutrients distribution in salt-affected soils of the Punjab in relation to soil properties. Arch. Agron. Soil. Sci. 2019, 55(4), 367–377. [Google Scholar] [CrossRef]
- Gowthamchand, N. J.; Dhaliwal, S. S.; Sharma, V.; Shukla, A. K.; Behera, S. K.; Kaur, M. Variation of trace metal minerals among different soil orders under different land use systems falling in Malwa region of Punjab in North-western India. J. Trace Elem. Miner. 2024, 9, 100162. [Google Scholar] [CrossRef]
- Manandhar, R.; Odeh, I.O. Interrelationships of land use/cover change and topography with soil acidity and salinity as indicators of land degradation. Land 2014, 3(1), 282–299. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, L.; Fu, W.; Xu, C.; Zhang, H.; Xu, X.; Ma, H.; Wang, J.; Zhang, Y. Soil acidification can be improved under different long-term fertilization regimes in a sweetpotato–wheat rotation system. Plants 2024, 13(13), 1740. [Google Scholar] [CrossRef]
- Liu, B.; Pan, L.; Qi, Y.; Guan, X.; Li, J. Land use and land cover change in the Yellow River Basin from 1980 to 2015 and its impact on the ecosystem services. Land 2021, 10(10), 1080. [Google Scholar] [CrossRef]
- Zhu, W.; Gu, S.; Jiang, R.; Zhang, X.; Hatano, R. Saline–alkali soil reclamation contributes to soil health improvement in China. Agriculture 2024, 14(8), 1210. [Google Scholar] [CrossRef]
- Jabbarov, Z.; Abdrakhmanov, T.; Abdullaev, S.; Makhammadiev, S.; Nomozov, U.; Rakhmatullaeva, G.; Kovácik, P. Evaluation of carbonate accumulation, inorganic carbon content, and soil property changes in newly developed soils of degraded landscapes. J. Degraded Min. Lands Manag. 2025, 12(4), 7993–8004. [Google Scholar] [CrossRef]
- Zhao, F.; Dong, G.; Shi, Z.; Chen, J.; Jiang, S.; Xu, Z.; Lafortezza, R.; Shao, C. Global Warming Potential Induced by Albedo and Greenhouse Gases Across Different Land Uses of the Saline-Alkaline Agropastoral Ecotone in the Songnen Plain. Agronomy 2026, 16(7), 705. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, J.; Zhang, Z. Remote Sensing Inversion and Spatiotemporal Dynamics of Multi-Depth Soil Salinity in a Typical Arid Wetland: A Case Study of Ebinur Wetland Reserve, Xinjiang. Remote Sens. 2025, 17(24), 3958. [Google Scholar] [CrossRef]
- Xu, D.; Ding, Y.; Yan, Y.; Qian, J.; Zhao, Q.; Xia, A. Soil Inorganic Carbon Content and Its Environmental Controls in the Weibei Loess Region: A Random Forest-Based Spatial Analysis. Land 2025, 14(8), 1609. [Google Scholar] [CrossRef]
- Wang, K.; Li, X.; Lyu, X.; Dang, D.; Dou, H.; Li, M.; Liu, S.; Cao, W. Optimizing the land use and land cover pattern to increase its contribution to carbon neutrality. Remote Sens. 2022, 14(19), 4751. [Google Scholar] [CrossRef]
- Nandwa, S.M. Soil organic carbon (SOC) management for sustainable productivity of cropping and agro-forestry systems in Eastern and Southern Africa. Nutr. Cycl. Agroecosystems 2001, 61(1), 143–158. [Google Scholar] [CrossRef]
- Mihelič, R.; Pečnik, J.; Glavan, M.; Pintar, M. Impact of sustainable land management practices on soil properties: Example of organic and integrated agricultural management. Land 2020, 10(1), 8. [Google Scholar] [CrossRef]
- Liu, Y.; Sui, X.; Hua, H.; Liu, X.; Chang, Q.; Xu, R.; Li, M.; Mu, L. Soil aggregate stability and organic carbon content among different forest types in temperate ecosystems in northeastern China. Forests 2024, 15(2), 279. [Google Scholar] [CrossRef]
- Sahoo, U. K.; Singh, S. L.; Gogoi, A.; Kenye, A.; Sahoo, S. S. Active and passive soil organic carbon pools as affected by different land use types in Mizoram, Northeast India. PLoS ONE 2019, 14(7), e0219969. [Google Scholar] [CrossRef]
- Shahpiri, A.; Kooch, Y.; Hojjati, S. M. Deterioration rate of below-ground organic matter fractions depends on the degradation intensity of above-ground vegetation cover. Sci. Total Environ. 2025, 1002, 180652. [Google Scholar] [CrossRef]
- Zhao, Y.; Xu, Y.; Cha, X.; Zhang, P.; Li, Y.; Cai, A.; Zhou, Z.; Yang, G.; Han, X.; Ren, C. A Global Meta-Analysis of Land Use Change on Soil Mineral-Associated and Particulate Organic Carbon. Glob. Change Biol. 2025, 31(3), e70111. [Google Scholar] [CrossRef]
- Song, Y. H.; Xu, F. B.; Wang, M. H.; Xie, Y. B.; Tian, L. M.; Lv, C. X.; Zhang, D. Plant Species Diversity Improves Soil Physicochemical Traits and Modulates Soil Microbial Community Structure, with a Pronounced Enhancement of Fungal Diversity in Urban Forests. Plants 2025, 15(1), 79. [Google Scholar] [CrossRef]
- Wang, M.; Cui, J.; Liu, H.; Xu, X. Characterization of Soil Microbial Biomass Carbon and Nitrogen in Four Forest Types of Shushan Urban Forest Park. Forests 2023, 14(7), 1498. [Google Scholar] [CrossRef]
- Wang, L.; Quzong, C.; Skal, S. G.; Mu, C.; Zhao, Y.; Fang, B.; Dorji, T. Depth-related patterns and physicochemical drivers of soil microbial communities in the alpine desert of Ngari, Xizang. Microorganisms 2025, 13(12), 2775. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Yu, S.; Wang, Y.; Song, H.; Wang, H. Enhancing soil properties and organic matter transformation in low-fertility reclaimed soil through reed-based exogenous carbon: Exploring the impact of rebuilt microbial community and enzyme activity. J. Environ. Sci. Health Part B 2026, 61(4), 185–202. [Google Scholar] [CrossRef] [PubMed]
- Tomar, S.; Bhardwaj, D. R.; Kumar, J.; Sharma, P.; Dogra, R. Soil carbon and nitrogen dynamics in agroforestry land use systems of north-western Himalayas: implications for environmental sustainability. Agrofor. Syst. 2025, 99(5), 96. [Google Scholar] [CrossRef]
- Meena, S.; Manjaiah, K.; Sharma, V.; Bana, R.; Yadav, S.; Meena, A.; Saini, R. Effects of land use systems on soil physical, chemical and biological properties in north-eastern region of India. Indian J. Agric. Sci. 2025, 95(11), 1379–1386. [Google Scholar] [CrossRef]
- Hu, Y.; Kong, M. Differential Responses of Soil Phosphorus Availability to Variations in Repeated Drying–Rewetting Cycles Under Different Land-Use Types in the Semi-Arid Loess Plateau of China. Agriculture 2026, 16(3), 376. [Google Scholar] [CrossRef]
- Liang, H.; Wang, C.; Lu, X.; Sai, C.; Liang, Y. Dynamic Changes in Soil Phosphorus Accumulation and Bioavailability in Phosphorus-Contaminated Protected Fields. Int. J. Environ. Res. Public Health 2022, 19(19), 12262. [Google Scholar] [CrossRef] [PubMed]
- Barłóg, P.; Hlisnikovský, L.; Łukowiak, R.; Kunzová, E. Soil Phosphorus and Potassium Fractions in Response to the Long-Term Application of Pig Slurry and NPK Mineral Fertilizers. Agronomy 2025, 15(5), 1183. [Google Scholar] [CrossRef]
- Getnet, K.; Miju, C.; Bitew, A.; Atinafu, M. The effects of land use types and soil depths on soil micronutrients, soil pH, and organic carbon in Southern Ethiopia. Appl. Environ. Soil. Sci. 2025, 2025(1), 5575783. [Google Scholar] [CrossRef]
- Dhaliwal, S. S.; Sharma, V.; Shukla, A. K.; Kaur, J.; Gupta, R. K.; Verma, V.; Singh, P. Interactive effect of land use systems on depth-wise soil properties and micronutrients minerals in North-Western, India. Heliyon 2023, 9(2). [Google Scholar] [CrossRef] [PubMed]
- Omokaro, G. O.; Ojie, C. O.; Otas, E. S.; Onakpoma, P. A. Macro and Micronutrient Status Under Different Land Use Types in the University of Benin. Int. J. Precis. Farming 2023, 1, 42–49. [Google Scholar] [CrossRef]
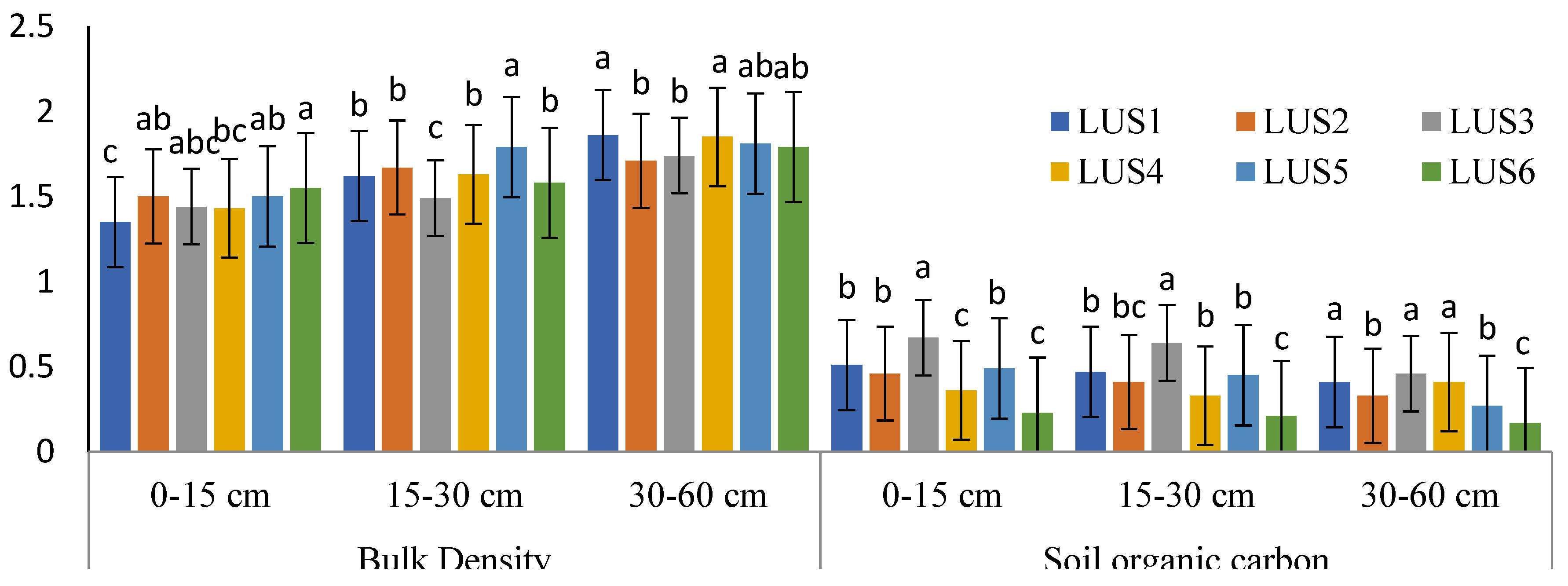
| LUS | Depth | pH | EC (Mgm-1) | Sand % | Silt % | Clay % |
|---|---|---|---|---|---|---|
| LUS1 | D1 | 7.43d±0.08 | 0.15b±0.05 | 39.60a±1.12 | 40.63a±0.68 | 19.77a±0.52 |
| D2 | 7.51c±0.12 | 0.42b±0.07 | 34.43a±1.67 | 46.88b±1.07 | 18.68bc±0.86 | |
| D3 | 7.55c±0.07 | 0.55b±0.12 | 26.73a±1.11 | 52.69a±0.64 | 20.58a±0.58 | |
| LUS2 | D1 | 7.71c±0.20 | 0.31b±0.06 | 42.45a±2.28 | 40.93a±0.68 | 16.20b±1.60 |
| D2 | 7.86bc±0.27 | 0.34b±0.10 | 36.63a±1.48 | 46.35b±1.07 | 17.02c±1.12 | |
| D3 | 7.94b±0.47 | 0.56b±0.12 | 27.65a±1.12 | 52.36a±1.34 | 20.00a±0.39 | |
| LUS3 | D1 | 7.77bc±0.12 | 0.11b±0.02 | 38.38a±0.89 | 40.49a±0.57 | 21.13a±0.86 |
| D2 | 7.90bc±0.12 | 0.18b±0.05 | 34.65a±1.48 | 45.75b±1.01 | 19.60b±0.82 | |
| D3 | 7.81bc±0.09 | 0.20c±0.05 | 26.73a±1.20 | 51.42a±1.22 | 21.85a±0.53 | |
| LUS4 | D1 | 7.82b±0.07 | 0.19b±0.03 | 37.11a±1.11 | 40.71a±0.88 | 22.18a±0.86 |
| D2 | 8.02b±0.15 | 0.25b±0.04 | 37.11a±1.11 | 45.13b±0.85 | 19.11bc±0.62 | |
| D3 | 7.76bc±0.12 | 0.41bc±0.07 | 27.30a±1.07 | 49.34a±1.27 | 23.36a±0.39 | |
| LUS5 | D1 | 7.61bc±0.06 | 0.13b±0.04 | 42.80a±1.30 | 40.39a±1.39 | 16.81b±1.37 |
| D2 | 7.73c±0.15 | 0.15b±0.04 | 36.33a±1.48 | 46.05b±1.07 | 17.62bc±1.12 | |
| D3 | 7.83bc±0.10 | 0.23c±0.02 | 27.35a±1.12 | 52.05a±1.34 | 20.60a±0.39 | |
| LUS6 | D1 | 8.58a±0.13 | 3.63a±0.75 | 34.23a±18.86 | 46.06a±17.15 | 19.71a±1.57 |
| D2 | 9.13a±0.12 | 3.68a±0.83 | 22.51b±6.13 | 52.05a±9.38 | 25.44a±3.73 | |
| D3 | 10.32a±0.10 | 4.34a±0.52 | 27.72a±9.99 | 49.10a±9.60 | 23.17a±9.62 |
| Depth | LUS | Carbon Fraction (gmkg-1) | Hot Water-Soluble Carbon (HWSC) | Acid Hydrolysable Humic Carbon (AHHC) | Microbial Biomass Carbon (MBC) (µgg-1) |
Dehydrogenase Activity (DEA) (µg TPFg-1day-1) |
Soil organic carbon stocks (MgCha¹) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Very labile | Labile | Less labile | Non labile | Mgkg-1 | ||||||
| D | LUS1 | 3.09b | 1.76c | 1.93b | 2.02b | 142.23b | 589.21b | 164.76b | 30.08a | 10.31b |
| LUS2 | 3.10ab | 1.89b | 1.89b | 2.02b | 137.46c | 561.12c | 172.51b | 26.76b | 10.37b | |
| LUS3 | 3.18a | 2.07a | 2.16a | 2.18a | 149.72a | 604.74a | 182.91a | 24.54bc | 14.46a | |
| LUS4 | 3.07b | 1.88b | 1.89b | 2.18a | 129.60d | 551.96c | 184.83a | 22.60cd | 7.78bc | |
| LUS5 | 3.04b | 1.75c | 1.87b | 2.15a | 149.57a | 579.95b | 182.18a | 30.54a | 11.02b | |
| LUS6 | 2.13c | 1.37d | 1.90b | 1.84c | 112.27e | 201.21d | 108.58c | 20.80d | 5.44c | |
| D2 | LUS1 | 2.96ab | 1.66b | 2.16b | 1.88b | 123.03b | 359.44a | 130.75a | 22.30ab | 11.56b |
| LUS2 | 2.93abc | 1.67b | 2.16b | 1.88b | 123.40b | 299.84c | 127.06ab | 23.36ab | 10.29b | |
| LUS3 | 3.04a | 1.89a | 2.47a | 2.05a | 129.60a | 335.86b | 128.95ab | 21.56b | 14.36a | |
| LUS4 | 2.84c | 1.68b | 2.16b | 2.05a | 123.03b | 301.48c | 134.15a | 17.96c | 8.02c | |
| LUS5 | 2.90bc | 1.66b | 2.08c | 1.93b | 129.40a | 330.84b | 121.52b | 24.10a | 12.29b | |
| LUS6 | 2.17d | 1.20c | 1.93d | 1.71c | 100.73c | 134.22d | 101.95c | 16.20c | 5.08d | |
| D3 | LUS1 | 2.50b | 1.45a | 2.41a | 1.75b | 101.83c | 250.38a | 105.00b | 17.38a | 11.54a |
| LUS2 | 2.50b | 1.45a | 2.48a | 1.75b | 109.72b | 200.00c | 104.16b | 17.26a | 8.46b | |
| LUS3 | 2.70a | 1.70a | 2.49a | 1.91a | 112.28a | 249.08a | 104.72b | 18.42a | 12.01a | |
| LUS4 | 2.61ab | 1.39a | 2.42a | 1.91a | 101.81c | 251.55a | 111.75a | 13.80b | 11.49a | |
| LUS5 | 2.64a | 1.46a | 2.41a | 1.76b | 109.73b | 232.11b | 105.66b | 17.72a | 7.49b | |
| LUS6 | 2.03c | 3.02a | 2.16b | 1.49c | 90.87d | 103.30d | 91.99c | 14.82b | 4.73c | |
| LUS | Depth | N (Kgha-1) | P (Kgha-1) | K (Kgha-1) | S (mg kg-1) | Fe (mg kg-1) | Mn (mg kg-1) | Cu (mg kg-1) | Zn (mg kg-1) |
|---|---|---|---|---|---|---|---|---|---|
| LUS1 | D1 | 252.50c±24.94 | 12.56c±1.31 | 277.72d±8.00 | 78.15ab±5.38 | 164.12a±5.35 | 52.42b±6.58 | 2.61a±0.59 | 1.45b±0.15 |
| D2 | 252.50c±25.11 | 12.56c±1.28 | 277.72d±8.41 | 78.15ab±7.36 | 164.12a±5.31 | 52.42b±7.43 | 2.61a±0.53 | 1.45b±0.15 | |
| D3 | 156.00d±13.68 | 10.57b±1.51 | 229.06c±11.79 | 43.11b±3.48 | 80.65a±8.96 | 35.31a±3.25 | 1.80a±0.15 | 0.72a±0.16 | |
| LUS2 | D1 | 272.67b±3.93 | 14.58b±0.31 | 303.70c±10.35 | 77.32b±6.65 | 159.85a±27.21 | 51.85b±5.43 | 2.85a±0.30 | 1.80a±0.23 |
| D2 | 272.67b±5.60 | 14.58b±0.29 | 303.70c±10.27 | 77.32b±6.38 | 159.85a±27.22 | 51.85b±5.43 | 2.85a±0.30 | 1.80a±0.23 | |
| D3 | 245.84b±4.02 | 11.17b±0.32 | 259.55b±2.34 | 51.70a±14.25 | 81.09a±19.12 | 29.20a±6.74 | 1.79a±0.37 | 0.80a±0.21 | |
| LUS3 | D1 | 330.45a±7.87 | 17.98a±0.07 | 395.22a±3.81 | 77.97ab±5.48 | 164.11a±5.38 | 52.37b±6.56 | 2.61a±0.57 | 1.42b±0.14 |
| D2 | 330.45a±19.99 | 17.98a±0.10 | 395.22a±31.73 | 77.97ab±5.14 | 164.11a±5.37 | 52.37b±6.56 | 2.61a±0.57 | 1.42b±0.14 | |
| D3 | 287.40a±2.02 | 14.63a±1.47 | 295.28a±7.52 | 43.04b±3.56 | 80.78a±9.13 | 35.38a±3.18 | 1.81a±0.16 | 0.72a±0.17 | |
| LUS4 | D1 | 258.62bc±9.37 | 11.10d±0.53 | 277.72d±8.00 | 78.15ab±5.38 | 164.12a±5.35 | 52.42b±6.58 | 2.61a±0.59 | 1.45b±0.15 |
| D2 | 258.62bc±9.00 | 11.10d±0.40 | 277.72d±13.46 | 78.15ab±5.31 | 164.12a±5.77 | 52.42b±6.58 | 2.61a±0.59 | 1.45b±0.15 | |
| D3 | 210.78c±16.84 | 8.96c±0.38 | 229.06c±11.79 | 43.11b±3.48 | 80.65a±8.96 | 35.31a±3.25 | 1.80a±0.15 | 0.72a±0.16 | |
| LUS5 | D1 | 275.05b±3.66 | 15.83b±0.37 | 317.55b±2.33 | 84.33a±2.69 | 156.97a±13.66 | 60.89a±5.43 | 2.70a±0.73 | 1.49b±0.14 |
| D2 | 275.05b±3.98 | 15.83b±0.38 | 317.55b±2.80 | 84.33a±2.60 | 156.97a±13.85 | 60.89a±5.43 | 2.70a±0.73 | 1.49b±0.14 | |
| D3 | 245.06b±2.88 | 11.01b±0.51 | 252.74b±1.64 | 50.71b±5.01 | 74.86a±14.78 | 30.72a±8.60 | 1.28b±0.31 | 0.59a±0.18 | |
| LUS6 | D1 | 122.79d±11.41 | 15.74b±2.09 | 279.86d±17.67 | 15.15c±1.26 | 113.24b±0.88 | 35.48c±0.75 | 1.32b±0.35 | 0.60c±0.09 |
| D2 | 122.79d±7.28 | 15.74b±1.97 | 279.86d±17.75 | 15.15c±1.23 | 113.24b±0.87 | 35.48c±0.75 | 1.32b±0.35 | 0.60c±0.09 | |
| D3 | 30.55e±3.64 | 6.40d±0.76 | 94.32d±10.44 | 10.96c±0.73 | 5.59b±0.62 | 3.43b±0.66 | 0.53c±0.13 | 0.23b±0.04 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




