Submitted:
03 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Transcriptome Analyses
2.1.1. Platform Used, Filtering and Normalization of Gene Expression Data
2.1.2. Transcriptomic Characterization of Individual Genes
2.1.3. Genomic Fabric Landscape Through Full Characterization of Gene-Pairs
2.1.4. Expression Regulation of Individual
2.1.5. Regulation and Remodeling of Individual Genes and Functional Pathways
2.1.6. Transcriptomic Recovery
2.2. Physiology Studies
2.2.1. Analyses
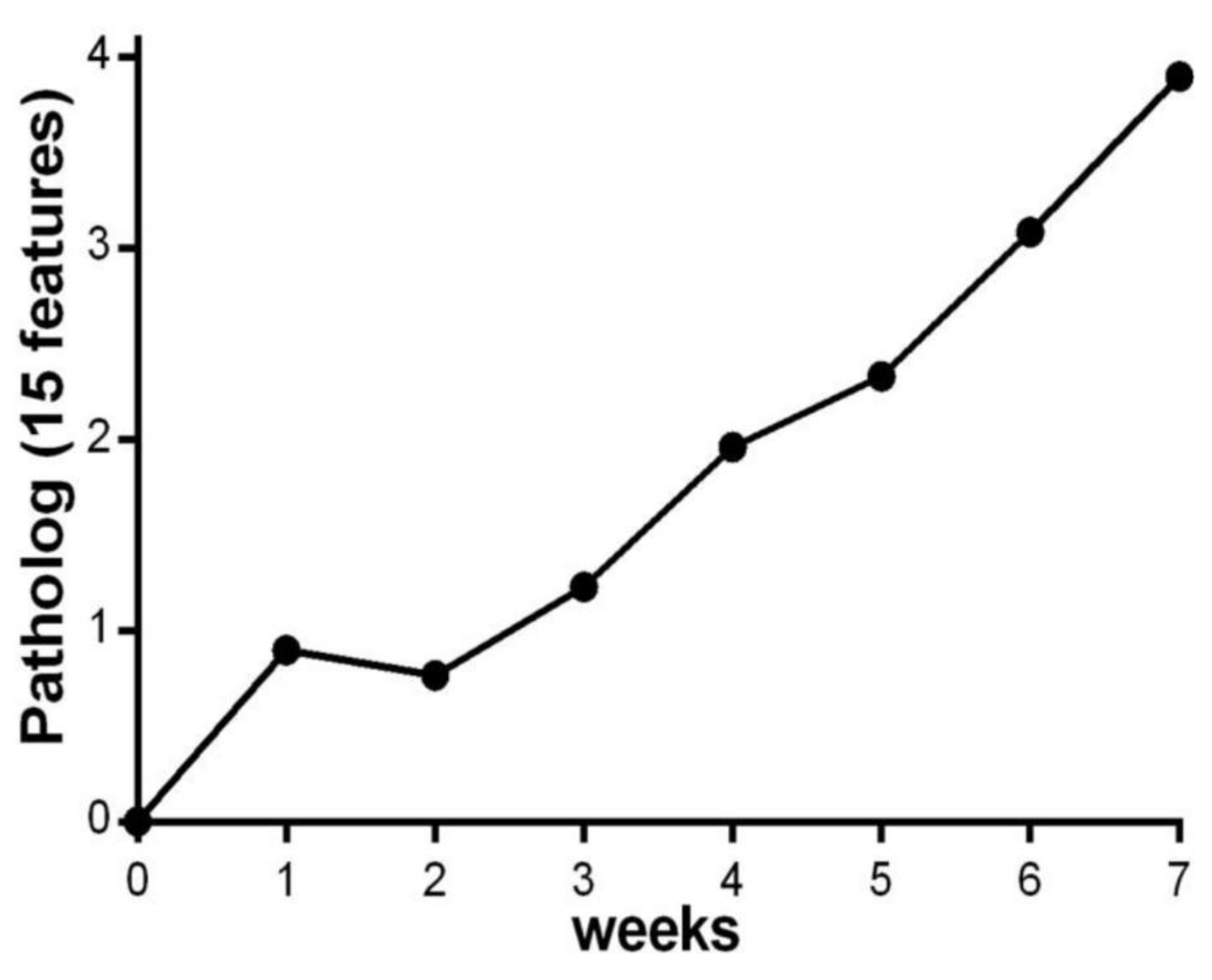
2.2.2. Data Transformation and Pathology Quantification
2.3. Histology and Apoptosis Detection
3. Results
3.1. Sex is an Important Transcriptomic Regulator
3.2. Heart Transcriptome Topology Changes During Development
3.3. Each Heart Chamber Has a Distinct Transcriptomic Topology
3.4. Expression Level and Subcellular Localization of Intercalated Disk Proteins Changes During Estrogen Cycle in Female Heart
3.5. Transcriptomic Consequences of Low Salt Diet
3.5. Transcriptomic, Morphological and Physio Pathological Consequences of Chronic Constant Hypoxia (CCH)
3.6. Transcriptomic, Morphological and Physio Pathological Consequences of Chronic Intermittent Hypoxia (CIH)
4. Animal Models
4.1. Chagas Disease
4.1.1. Experimental Methods
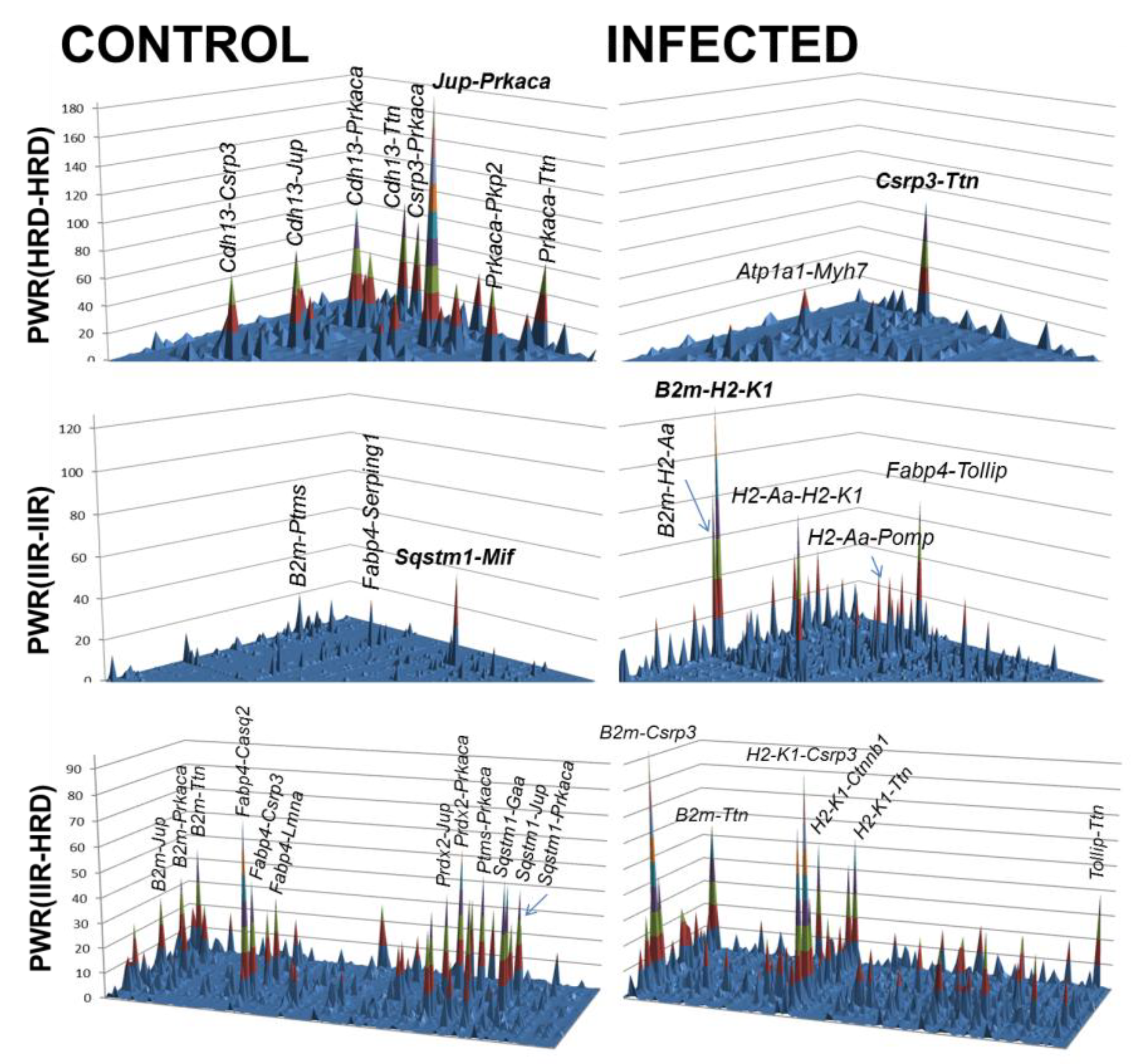
4.1.2. Parasite Infection and Development of Chagas Disease
4.1.3. Cell Therapy for Chagas Disease
4.2. Post-Ischemic Heart Failure
4.2.1. Induction of Ischemic Heart Failure in a Mouse Model
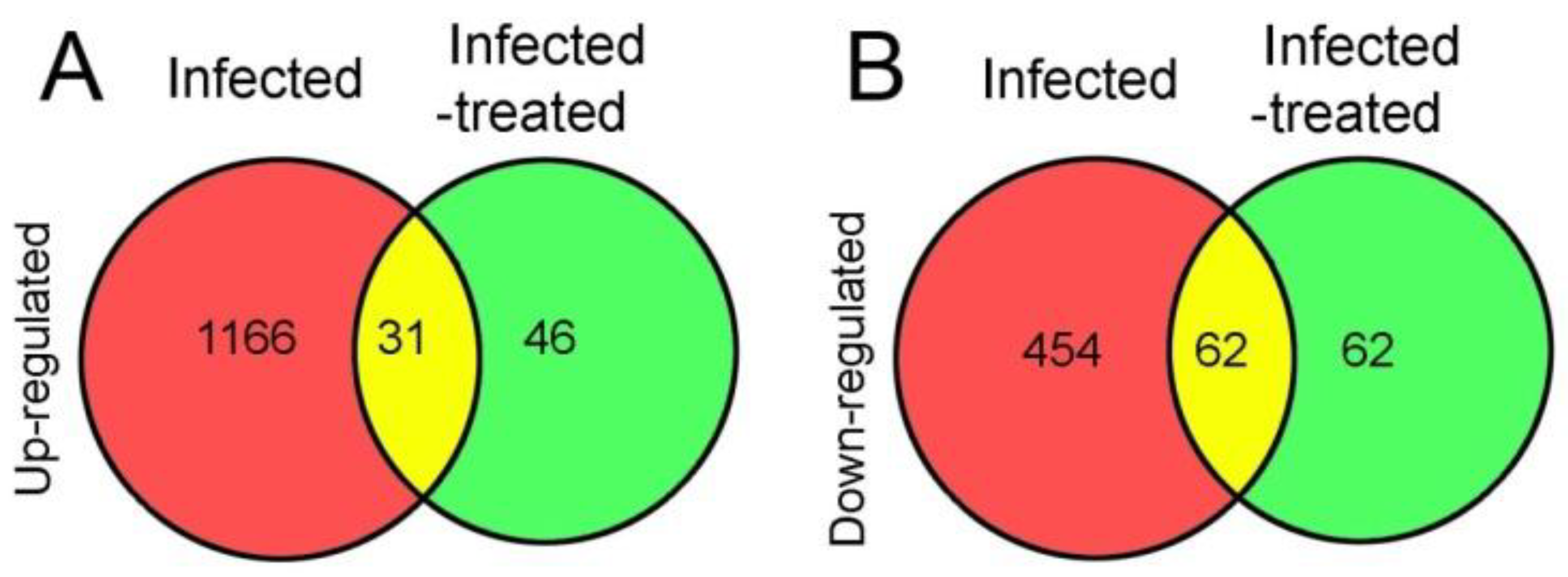
4.2.2. Heart Transcriptomic Changes in Infarcted Mice
4.2.3. Cell Therapy for Post Ischemic Heart Failure
4.3. Pulmonary Hypertension
4.3.1. Induction of Pulmonary Hypertension in Rat Models
4.3.2. Lung Transcriptomic Consequences of Pulmonary Hypertension
4.4. Metabolic Syndrome
4.4.1. Induction of Metabolic Syndrome in a Dog Model
4.4.2. Progression of the Metabolic Syndrome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AECOM | Albert Einstein College of Medicine |
| AVE | Average normalized expression level of a gene |
| CCC | Chronic Chagas Cardiomyopathy |
| CCH | Chronic Constant Hypoxia |
| CHF | Congestive Heart Failure |
| CIH | Chronic Intermittent Hypoxia |
| CRE | Creatinine |
| COORD | percent of statistically synergistically expressed gene pairs + percent of antagonistically expressed pairs minus percent of independently expressed genes within the analyzed pathways |
| COR | pair-wise Pearson correlation coefficient of the (log2) of the normalized expression levels of two genes in the same condition/region |
| GCH | Gene Commanding Height |
| GLU | Blood sugar |
| IACUC | Institutional Animal Care and Use Committee |
| KEGG | Kyoto Encyclopedia of Genes and Genomes |
| MetS | Metabolic Syndrome |
| P | Patholog |
| PH | Pulmonary Hypertension |
| PRE | Pathway Restoration Efficiency |
| PWR | Pair-Wise Relevance |
| REC | Relative Expression Control |
| REV | Relative Expression Variation |
| TD | Transcriptomic Distance |
| TRE | Transcriptomic Recovery Efficiency |
| TSH | thyroid stimulating hormone |
| WIR | Weighted Individual (Gene) Regulation |
| WPR | Weighted Pathway Regulation |
References
- Albu, M.; Sedmera, D.; Stainier, D.Y.R. Recent insights into atrial chamber formation. Semin Cell Dev. Biol. 2025, 175, 103664. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, A.; Adams, J.W.; Vaccarezza, M. The vertebrate heart: an evolutionary perspective. J. Anat. 2017, 231(6), 787–797. [Google Scholar] [CrossRef]
- Kiani, A.K.; Pheby, D.; Henehan, G.; Brown, R.; Sieving, P.; Sykora, P.; et al. INTERNATIONAL BIOETHICS STUDY GROUP. Ethical considerations regarding animal experimentation. J. Prev. Med. Hyg. 2022, 63((2) Suppl 3, E255–E266. [Google Scholar] [CrossRef]
- National Research Council. Guide for the care and use of laboratory animals. 8th Edition. Washington D.C. The National Academies Press. Available online: https://grants.nih.gov/grants/olaw/guide-for-the-care-and-use-of-laboratory-animals.pdf (accessed on 01/31/2026).
- Vo, Q.D. Gene editing therapy as a therapeutic approach for cardiovascular diseases in animal models: A scoping review. PLoS ONE 2025, 20(6), e0325330. [Google Scholar] [CrossRef]
- Zeng, L.; Chi, J.; Zhu, M.; Hao, H.; Long, S.; Liu, Z.; Zhang, C. Rodent Models for Atherosclerosis. Int. J. Mol. Sci. 2025, 27(1), 378. [Google Scholar] [CrossRef]
- Zeng, L.; Chi, J.; Zhu, M.; Hao, H.; Long, S.; Liu, Z.; et al. Rodent Models for Atherosclerosis. Int. J. Mol. Sci. 2025, 27(1), 378. [Google Scholar] [CrossRef]
- Delfos, L.; Huis, C.; Imani, N.; Roeters van Lennep, J.E.; Hooijmans, C.R.; Bot, I. Modulation of IL-1, IL-6 and IL-18 cytokine signaling in animal models of atherosclerosis: a systematic review evaluating animal sex in preclinical research. Eur. J. Pharmacol. 2025, 1008, 178336. [Google Scholar] [CrossRef]
- Wu, X.; Zhou, Q.; Huang, Y.; Jiang, W.; Zhou, J.; Qian, K.; et al. Cellular senescence in age-related cardiovascular disease: past and future. Front Aging. 2026, 6, 1721744. [Google Scholar] [CrossRef] [PubMed]
- Zulkefli, S.B.; Mohamad, F.; Nasir, N.A.A.; Shuid, A.N.; Azme, N. Mechanistic Insights into Therapeutic Strategies for Post- Menopausal Atherosclerosis: Evidence from an Ovariectomized Mouse Model. J. Cardiovasc Transl. Res. 2026, 19(1), 46. [Google Scholar] [CrossRef] [PubMed]
- Enkhmaa, B.; Anuurad, E.; Berglund, L. Mediterranean diet and cardiovascular disease: a step closer to mechanisms using a precision animal model? Transl. Res. 2015, 166(1), 41–3. [Google Scholar] [CrossRef]
- Sun, Y.; Su, Y.; Ding, N.; Yang, Z.; Zhao, J.; Wang, X.; et al. Effect of Yi-Nao-Jie-Yu Prescription on Post-Stroke Depression in Rats using Middle Cerebral Artery Occlusion Combined with Behavioral Restraint. J. Vis. Exp. 2026, (227). [Google Scholar] [CrossRef]
- Chege, B.M.; Mwangi, P.W.; Githinji, C.G.; Bukachi, F. Combined antiretroviral therapy with low- or normal-protein, high-calorie diets appears to induce significant deleterious electrocardiographic changes in a rodent model. Braz. J. Med. Biol. Res. 2026, 59, e14744. [Google Scholar] [CrossRef]
- Zheng, X.; Li, Y.; Wang, P.; Guo, Z.; Liu, Y.; Liu, Q.; et al. Physical exercise mitigates chronic psychological stress-induced vascular inflammation via the BDNF-Kif4-TARM1 axis. Clin. Transl. Med. 2026, 16(4), e70674. [Google Scholar] [CrossRef]
- Mohan, A.S.; Radhakrishnan, S.; Aswany, M. G.; Mohan, N.; John, A.J.; Anie, Y. Cl-amidine attenuates neutrophil extracellular trap-enclosed extracellular vesicle (NET-EV)-mediated thrombosis in diabetic mice. Thromb. Res. 2026, 260, 109658. [Google Scholar] [CrossRef] [PubMed]
- Duke. Mouse and rat microarrays. Available online at: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL9207, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL8928, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi? acc=GPL8938. Accessed 01/31/2026.
- AECOM. Mouse and rat cDNA and oligonucleotide microarray printed by the Microarray Facility of Albert Einstein College of Medicine. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL369 (accessed on 01/31/2026).
- Yale. Mouse microarrays. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL2828 (accessed on 01/31/2026).
- Agilent technology. SurePrint Microarray Hybridization Setup. Available online: https://www.youtube.com/watch?v=AgWbneDtVXU (accessed on 01/31/2026).
- Iacobas, D.A.; Obiomon, E.A.; Iacobas, S. Genomic Fabrics of the Excretory System’s Functional Pathways Remodeled in Clear Cell Renal Cell Carcinoma. Curr. Issues Mol. Biol. 2023, 45, 9471–9499. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, S.; Ede, N.; Iacobas, D.A. The Gene Master Regulators (GMR) Approach Provides Legitimate Targets for Personalized, Time-Sensitive Cancer Gene Therapy. Genes 2019, 10(8), 560. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Iacobas, D.A.; Zhou, D.; Chen, Q.; Lai, J.K.; Gavrialov, O.; Haddad, G.G. Gene expression and phenotypic characterization of mouse heart after chronic constant or intermittent hypoxia. Physiol. Genom. 2005, 22(3), 292–307. [Google Scholar] [CrossRef]
- KEGG. Kyoto Encyclopedia of Genes and Genomes. Available online: https://www.kegg.jp/kegg/pathway.html (accessed on 01/31/2026).
- Nganele, D.M.; Hintze, T.H. Cardiac chemical reflex control of preload in conscious dogs. Am. J. Physiol. 1990, 258 4 Pt 2, H1055–63. [Google Scholar] [CrossRef]
- Iacobas, D.A. Medical Biostatistics, 3rd English ed.; Bucura Mond: Bucarest, Romania, 1997; ISBN 973-97977-3-3. [Google Scholar]
- Gene Expression Omnibus. Sex-dependent gene regulatory networks of the heart rhythm. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE17324 (accessed on March 1st 2026).
- Iacobas, D.A.; Iacobas, S.; Thomas, N.; Spray, D.C. Sex-dependent gene regulatory networks of the heart rhythm. Funct. Integr. Genom. 2010, 10(1), 73–86. [Google Scholar] [CrossRef]
- Cellini, A.; Höfler, D.; Arias-Loza, P.A.; Bandleon, S.; Langsenlehner, T.; Kohlhaas, M.; et al. The α2-isoform of the Na+/K+-ATPase protects against pathological remodeling and β-adrenergic desensitization after myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2021, 321(4), H650–H662. [Google Scholar] [CrossRef]
- Hutschalik, T.; Dasí, A.; Riebel, L.L.; Wiendels, M.; Bakker, F.; Beckers, L.J.A.M.; et al. GJA5 and ATP1A1 perturbations recapitulate inflammation-related beat irregularities in iPSC-based atrial myocardium tissue model. Front Immunol. 2026, 16, 1719392. [Google Scholar] [CrossRef]
- Ying, H.; Fan, H.; Wang, Y.; Jiang, R.; Cai, D.; Cheng, H.; et al. Connexin43 Deficiency Leads to Ventricular Arrhythmias by Reprogramming Proline Metabolism. Adv. Sci. (Weinh) 2026, 13(19), e16090. [Google Scholar] [CrossRef]
- James, A.F.; Choisy, S.C.; Hancox, J.C. Recent advances in understanding sex differences in cardiac repolarization. Prog. Biophys. Mol. Biol. 2007, 94(3), 265–319. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, M.K.; Vos, M.A.; Duker, G.; Demolombe, S.; van Veen, T.A. Gender disparity in cardiac electrophysiology: implications for cardiac safety pharmacology. Pharmacol. Ther. 2010, 127(1), 9–18. [Google Scholar] [CrossRef]
- Burke, J.H.; Ehlert, F.A.; Kruse, J.T.; Parker, M.A.; Goldberger, J.J.; Kadish, A.H. Gender-specific differences in the QT interval and the effect of autonomic tone and menstrual cycle in healthy adults. Am. J. Cardiol. 1997, 79(2), 178–81. [Google Scholar] [CrossRef] [PubMed]
- Omnibus, Gene Expression. Gene expression and phenotypic characterization of mouse heart after chronic constant or intermittent hypoxia. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE2271 (accessed on March 1st 2026).
- Agilent-026655. Whole Mouse Genome Microarray 4x44K v2. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GPL10333 (accessed on Feb. 20/2026).
- Gene Expression Omnibus. Left-right transcriptomic differences in adult male mouse heart ventricles. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE45348 (accessed on Feb. 20/2026).
- Gene Expression Omnibus. Left-right transcriptomic differences in adult male mouse heart atria. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE45339 (accessed on Feb. 20/2026).
- Iacobas, S.; Amuzescu, B.; Iacobas, D.A. Transcriptomic uniqueness and commonality of the ion channels and transporters in the four heart chambers. Sci. Rep. 2021, 11(1), 2743. [Google Scholar] [CrossRef] [PubMed]
- Artemieva, M.M.; Makeeva, A.V.; Adasheva, D.A.; Shein, V.E.; Katrukha, A.G.; Postnikov, A.B.; et al. Left Ventricular and Right Ventricular Hypertrophy Modelling to Study PAPP-A-Mediated IGFBP-4 Cleavage-a Mechanism That Regulates IGF Bioavailability in Adult Rats. Int. J. Mol. Sci. 2026, 27(6), 2761. [Google Scholar] [CrossRef]
- Wing-Ki Chik, B.; Chan, K.L.; Chow, P.C.; Kwong-Man Yu, C.; Yuk-Ting Chan, Y.; Cheung, Y.F. Right Atrial Functional Reserve is Associated With Right Ventricular Diastolic Reserve and Liver Stiffness in Repaired Tetralogy of Fallot. Echocardiography. 2026, 43(3), e70426. [Google Scholar] [CrossRef]
- Zang, H.; Jia, C.; Pan, Y.; Li, X.; Zhang, Q. Left Atrial Volume Index and Left Ventricular Mass Index Determine the Benefits of Spironolactone in Patients With Heart Failure With Preserved Ejection Fraction. J. Am. Heart Assoc. 2026, 15(7), e044115. [Google Scholar] [CrossRef]
- Günthel, M.; Barnett, P.; Christoffels, V.M. Development, Proliferation, and Growth of the Mammalian Heart. Mol. Ther. 2018, 26(7), 1599–1609. [Google Scholar] [CrossRef]
- KEGG. Adrenergic signaling in cardiomyocytes. Available online: https://www.kegg.jp/pathway/mmu04261 (accessed on March 1st 2026).
- KEGG. Calcium signaling pathway. Available online: https://www.kegg.jp/pathway/mmu04020 (accessed on March 1st 2026).
- KEGG. Cardiac muscle contraction. Available online: https://www.kegg.jp/pathway/mmu04260 (accessed on March 1st 2026).
- KEGG. Glycolysis/Gluconeogenesis. Available online: https://www.kegg.jp/pathway/mmu00010 (accessed on March 1st 2026).
- KEGG. Oxidative Phosphorylation. Available online: https://www.kegg.jp/pathway/mmu00190 (accessed on March 1st 2026).
- Thomas, N.M.; Jasmin, J.F.; Lisanti, M.P.; Iacobas, D.A. Sex differences in expression and subcellular localization of heart rhythm determinant proteins. Biochem Biophys. Res. Commun. 2011, 406(1), 117–22. [Google Scholar] [CrossRef]
- Nelson, J.F.; Felicio, L.S.; Randall, P.K.; Sims, C.; Finch, C.E. A longitudinal study of estrous cyclicity in aging C57BL/6J mice: I. Cycle frequency, length and vaginal cytology. Biol. Reprod. 1982, 27(2), 327–39. [Google Scholar] [CrossRef] [PubMed]
- Musil, L.S.; Goodenough, D.A. Biochemical analysis of connexin43 intracellular transport, phosphorylation, and assembly into gap junctional plaques. J. Cell Biol. 1991, 115(5), 1357–74. [Google Scholar] [CrossRef]
- Harvey, B.I.; Yoniles, A.M.; Monsivais, A.; Du, J.; Zadorozny, L.; Yu, Q.; et al. Sex-Specific Differences in LPS-Induced Rapid Myocardial Dysfunction. Int. J. Mol. Sci. 2025, 26(13), 5963. [Google Scholar] [CrossRef]
- Turner, C.G.; DuPont, J.J. The effect of transient sex hormone fluctuations on vascular endothelial function. Am. J. Physiol. Heart Circ. Physiol. 2025, 329(1), H217–H232. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.H.; Khera, A.; Anderson, C.A.M.; Appel, L.J.; DeSilva, D.M.; Gardner, C.; et al. American Heart Association. 2026 Dietary Guidance to Improve Cardiovascular Health: A Scientific Statement From the American Heart Association. Circulation. 2026. [Google Scholar] [CrossRef] [PubMed]
- Gene Expression Omnibus. Transcriptomic Effects of Law Salt Diet on the Mouse Left Ventricle. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE72561 (accessed on 27 January 2026).
- KEGG Pathways. Fatty acid Biosynthesis. Available online: https://www.genome.jp/kegg-bin/show_pathway?mmu00061 (accessed on 11 January 2026).
- KEGG Pathways. Steroid Biosynthesis. Available online: https://www.genome.jp/kegg-bin/show_pathway?mmu00100 (accessed on 11 January 2026).
- Iacobas, D.A.; Allen, H.; Iacobas, S. Low-Salt Diet Regulates the Metabolic and Signal Transduction Genomic Fabrics, and Remodels the Cardiac Normal and Chronic Pathological Pathways. Curr. Issues Mol. Biol. 2024, 46, 2355–2385. [Google Scholar] [CrossRef]
- KEGG pathway. Chagas disease. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu05142 (accessed on 1 March 2026).
- KEGG Pathways. Diabetic cardiomyopathy. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu05415 (accessed on 1 March 2026).
- KEGG Pathways. Dilated cardiomyopathy. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu05414 (accessed on 1 March 2026).
- KEGG Pathways. Hypertrophic cardiomyopathy. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu05410 (accessed on 1 March 2026).
- Iacobas, D.A.; Fan, C.; Iacobas, S.; Haddad, G.G. Integrated transcriptomic response to cardiac chronic hypoxia: translation regulators and response to stress in cell survival. Funct. Integr. Genom. 2008, 8(3), 265–75. [Google Scholar] [CrossRef]
- Penaloza, D.; Arias-Stella, J. The heart and pulmonary circulation at high altitudes: healthy highlanders and chronic mountain sickness. Circulation.;Review 2007, 115(9), 1132–1146. [Google Scholar] [CrossRef]
- Covello, K.L.; Simon, M.C. HIFs, hypoxia, and vascular development Review. Curr. Top. Dev. Biol. 2004, 62, 37–54. [Google Scholar] [CrossRef] [PubMed]
- Macario, A.J. Heat-shock proteins and molecular chaperones: implications for pathogenesis, diagnostics, and therapeutics. Int. J. Clin. Lab Res. 1995, 25(2), 59–70. [Google Scholar] [CrossRef]
- Dursunoglu, D.; Dursunoglu, N. Cardiovascular diseases in obstructive sleep apnea Review. Tuberk. Toraks 2006, 54(4), 382–396. [Google Scholar]
- Park, A.M.; Nagase, H.; Kumar, S.V.; Suzuki, Y.J. Effects of intermittent hypoxia on the heart. Antioxid. Redox Signal.;Review 2007, 9(6), 723–729. [Google Scholar] [CrossRef]
- Jain, V. Clinical perspective of obstructive sleep apnea-induced cardiovascular complications. Antioxid. Redox Signal.;Review 2007, 9(6), 701–710. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, S.; Iacobas, D.A. Effects of chronic intermittent hypoxia on cardiac rhythm transcriptomic networks. In Intermittent Hypoxia and Human Diseases; Xi, L., Serebrovskaya, T.V., Eds.; Springer-Verlag London UK, 2012; pp. 15–27. [Google Scholar]
- Uniacke, J.; Holterman, C.; Lachance, G.; et al. An oxygen-regulated switch in the protein synthesis machinery. Nature 2012, 486, 126–129. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, D.A.; Iacobas, S.; Haddad, G.G. Heart rhythm genomic fabric in hypoxia. Biochem Biophys. Res. Commun. 2010, 391(4), 1769–74. [Google Scholar] [CrossRef]
- KEGG Pathways. Calcium signaling pathway. Available online: https://www.kegg.jp/pathway/mmu04020 (accessed on March 1st 2026).
- KEGG Pathways. WNT signaling pathway. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu04310 (accessed on March 2nd 2026).
- de Lima, E.V.; Bertolini, V.K.D.S.; Cunha, M.D.P.; Miguel, D.C.; de Almeida, E.A.; Gadelha, F.R. Trypanosoma cruzi DTU diversity, benznidazole response, and clinical manifestations of Chagas disease: a seventeen-year systematic review and meta-analysis. Acta Trop. 2026, 108105. [Google Scholar] [CrossRef]
- Agudelo Higuita, N.I.; Henao-Martínez, A.F.; Vieira Batista, M.; Herrera Bernal, C.P.; Rezende Filho, J.; Rassi, A., Jr. State-of-the-Art Review: Chagas Disease-an Enduring Challenge. Clin. Infect. Dis. 2026, 82(2), 187–205. [Google Scholar] [CrossRef] [PubMed]
- Tanowitz, H.B.; Machado, F.S.; Jelicks, L.A.; Shirani, J.; de Carvalho, A.C.C.; Spray, D.C.; et al. Perspectives on Trypanosoma cruzi–Induced Heart Disease (Chagas Disease). Prog. Cardiovasc. Dis. 2009, 51, 524–539. [Google Scholar] [CrossRef]
- Barbosa, J.M.C.; Duarte, R.B.; Veloso, H.H.; Daliry, A.; Salomão, K. Chagas disease and amiodarone: a bibliometric and systematic review from cell to patient. Front Pharmacol. 2026, 17, 1749345. [Google Scholar] [CrossRef] [PubMed]
- Gonzaga, B.M.S.; Ferreira, R.R.; Coelho, L.L.; Carvalho, A.C.C.; Garzoni, L.R.; Araujo-Jorge, T.C. Clinical trials for Chagas disease: etiological and pathophysiological treatment. Front Microbiol. 2023, 14, 1295017. [Google Scholar] [CrossRef]
- Mukherjee, S.; Belbin, T.J.; Spray, D.C.; Iacobas, D.A.; Weiss, L.M.; Kitsis, R.N.; et al. Microarray analysis of changes in gene expression in a murine model of chronic chagasic cardiomyopathy. Parasitol. Res. 2003, 91(3), 187–96. [Google Scholar] [CrossRef]
- Adesse, D.; Iacobas, D.A.; Iacobas, S.; Garzoni, L.R.; Meirelles Mde, N.; Tanowitz, H.B.; et al. Transcriptomic signatures of alterations in a myoblast cell line infected with four distinct strains of Trypanosoma cruzi. Am. J. Trop. Med. Hyg. 2010, 82(5), 846–54. [Google Scholar] [CrossRef]
- Gene Expression Omnibus. Transcriptomic alterations in a myoblast cell line infected with four distinct strains of Trypanosoma cruzi. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE18175 (accessed on March 1st. 2026).
- Soares, M.B.; de Lima, R.S.; Rocha, L.L.; Vasconcelos, J.F.; Rogatto, S.R.; dos Santos, R.R.; et al. Gene expression changes associated with myocarditis and fibrosis in hearts of mice with chronic chagasic cardiomyopathy. J. Infect. Dis. 2010, 202(3), 416–26. [Google Scholar] [CrossRef] [PubMed]
- Gene Expression Omnibus. Gene expression changes associated with myocarditis and fibrosis in hearts of mice with chronic chagasic cardiomyopathy. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE17363 (accessed on March 1st 2026).
- Goldenberg, R.C.; Iacobas, D.A.; Iacobas, S.; Rocha, L.L.; da Silva de Azevedo Fortes, F.; Vairo, L.; et al. Transcriptomic alterations in Trypanosoma cruzi-infected cardiac myocytes. Microbes Infect. 2009, 11(14-15), 1140–9. [Google Scholar] [CrossRef] [PubMed]
- Gene Expression Omnibus. Transcriptomic alterations in Trypanosoma cruzi-infected cardiac myocytes. Available online: https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE17330 (accessed on March 1st 2026).
- Machado, R.R.B.; B Scariot, D.; Bakoshi, A.B.K.; Mbaye, E.H.A.; Almunif, S.; Sharma, S.; et al. Cytosolic Delivery of a Bithiophene Derivative via Polymersomes Kills Trypanosoma cruzi Amastigotes and Modulates the Inflammatory Response. ACS Appl. Nano Mater. 2026, 9(8), 3689–3703. [Google Scholar] [CrossRef]
- da Silva, G.P.; Sousa, L.R.D.; Vieira, P.M.A.; Stefani, R.; do Nascimento, A.M. In Vitro and In Silico Evaluation of the Trypanocidal Activity of a Subfraction Isolated from Mutisia campanulata. ACS Omega 2026, 11(5), 7659–7671. [Google Scholar] [CrossRef]
- Lanera, S.D.C.; Lara, L.D.S.; Orlando, L.M.R.; de Souza, T.P.; de Oliveira, E.C.; Paes, V.B.; et al. Trypanosoma cruzi carbonic anhydrase inhibitors as a potential antiparasitic agent. Int. J. Biol. Macromol. 2026, 351, 150985. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Fine, E.J.; Koba, W.; Zhao, D.; Rossi, M.A.; Tanowitz, H.B.; et al. Micropositron emission tomography in the evaluation of Trypanosoma cruzi-induced heart disease: Comparison with other modalities. Am. J. Trop. Med. Hyg. 2009, 81, 900–5. [Google Scholar] [CrossRef]
- de Souza, A.P.; Tang, B.; Tanowitz, H.B.; Araujo-Jorge, T.C.; Jelicks, E.L. Magnetic resonance imaging in experimental Chagas disease: a brief review of the utility of the method for monitoring right ventricular chamber dilation. Parasitol. Res. 2005, 97, 87–90. [Google Scholar] [CrossRef]
- Jelicks, L.A.; Tanowitz, H.B. Advances in imaging of animal models of Chagas disease. Adv. Parasitol. 2011, 75, 193–208. [Google Scholar] [CrossRef]
- Seah, S.K.; Marsden, P.D.; Voller, A.; Pettitt, L.E. Experimental Trypanosoma cruzi infection in rhesus monkeys--the acute phase. Trans. R Soc. Trop. Med. Hyg. 1974, 68(1), 63–9. [Google Scholar] [CrossRef]
- KEGG. JAK-STAT signaling pathway. Available online: https://www.kegg.jp/pathway/mmu04630 (accessed on March 1st).
- KEGG. Cell cycle. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu04110 (accessed on March 1st).
- Nisimura, L.M.; Coelho, L.L.; de Melo, T.G.; Vieira, P.C.; Victorino, P.H.; Garzonim, L.R.; et al. Trypanosoma cruzi Promotes Transcriptomic Remodeling of the JAK/STAT Signaling and Cell Cycle Pathways in Myoblasts. Front Cell Infect. Microbiol. 2020, 10, 255. [Google Scholar] [CrossRef] [PubMed]
- KEGG. Gap junction. Available online: https://www.kegg.jp/kegg-bin/show_pathway?mmu04540 (accessed on March 1st).
- Adesse, D.; Goldenberg, R.C.; Fortes, F.S.; Jasmin; Iacobas, D.A.; Iacobas, S.; et al. Gap junctions and chagas disease. Adv. Parasitol. 2011, 76, 63–81. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, D.A.; Manzoor, S.; Daniels, D.; Iacobas, S.; Xi, L. Mitochondrial Collapse Responsible for Chagasic and Post-Ischemic Heart Failure Is Reversed by Cell Therapy Under Different Transcriptomic Topologies. Curr. Issues Mol. Biol. 2025, 47, 940. [Google Scholar] [CrossRef]
- Parker, A.M.; Lees, J.G.; Murray, A.J.; Velagic, A.; Lim, S.Y.; De Blasio, M.J.; Ritchie, R.H. Precision Medicine: Therapeutically Targeting Mitochondrial Alterations in Heart Failure. JACC Basic Transl. Sci. 2025, 10, 101345. [Google Scholar] [CrossRef]
- Mongelli, A.; Mengozzi, A.; Geiger, M.; Gorica, E.; Mohammed, S.A.; Paneni, F.; et al. Mitochondrial epigenetics in aging and cardiovascular diseases. Front. Cardiovasc. Med. 2023, 10, 1204483. [Google Scholar] [CrossRef] [PubMed]
- Pietrangelo, D.; Lopa, C.; Litterio, M.; Cotugno, M.; Rubattu, S.; Lombardi, A. Metabolic Disturbances Involved in Cardiovascular Diseases: The Role of Mitochondrial Dysfunction, Altered Bioenergetics and Oxidative Stress. Int. J. Mol. Sci. 2025, 26, 6791. [Google Scholar] [CrossRef]
- Dhalla, N.S.; Ostadal, P.; Tappia, P.S. Involvement of Oxidative Stress in Mitochondrial Abnormalities During the Development of Heart Disease. Biomedicines 2025, 13, 1338. [Google Scholar] [CrossRef]
- Pinto Dias, J.C. The treatment of Chagas disease (South American trypanosomiasis). Ann. Intern Med. 2006, 16;144, 772–4. [Google Scholar] [CrossRef]
- Antonoglou, M.B.; Sánchez Alberti, A.; Redolfi, D.M.; Bivona, A.E.; Noli Truant, S.; Sarratea, M.B.; et al. Protective immunity against Chagas disease induced by a superantigen-based chimeric DNA vaccine delivered by attenuated Salmonella. Front Immunol. 2026, 17, 1788924. [Google Scholar] [CrossRef]
- Sanchez Alberti, A.; Bivona, A.E.; Matos, M.N.; Cerny, N.; Schulze, K.; Weißmann, S.; et al. Mucosal Heterologous Prime/Boost Vaccination Induces Polyfunctional Systemic Immunity, Improving Protection Against Trypanosoma cruzi. Front Immunol. 2020, 11, 128. [Google Scholar] [CrossRef] [PubMed]
- Cunha-Neto, E.; Santoro Rosa, D. Can Vaccination Prevent the Development of Chagas Disease Cardiomyopathy? JACC Basic Transl. Sci. 2026, 11(3), 101513. [Google Scholar] [CrossRef] [PubMed]
- Tejo, A.M.; Campos, S.V. Parasitic diseases in heart transplantation: challenges of Toxoplasmosis and Chagas Disease. JHLT Open 2025, 11, 100450. [Google Scholar] [CrossRef]
- Soares, M.B.; Lima, R.S.; Souza, B.S.; Vasconcelos, J.F.; Rocha, L.L.; Dos Santos, R.R.; et al. Reversion of gene expression alterations in hearts of mice with chronic chagasic cardiomyopathy after transplantation of bone marrow cells. Cell Cycle 2011, 10(9), 1448–55. [Google Scholar] [CrossRef] [PubMed]
- Barbash, I.M.; Chouraqui, P.; Baron, J.; Feinberg, M.S.; Etzion, S.; Tessone, A.; et al. Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation 2003, 108(7), 863–868. [Google Scholar] [CrossRef]
- Campos de Carvalho, A.C.; Goldenberg, R.C.; Jelicks, L.A.; Soares, M.B.; Dos Santos, R.R.; Spray, D.C.; et al. Cell Therapy in Chagas Disease. Interdiscip. Perspect. Infect. Dis. 2009, 2009, 484358. [Google Scholar] [CrossRef]
- Sousa, M.G.; Paulino-Junior, D.; Pascon, J.P.; Pereira-Neto, G.B.; Carareto, R.; Champion, T.; et al. Cardiac function in dogs with chronic Chagas cardiomyopathy undergoing autologous stem cell transplantation into the coronary arteries. Can. Vet. J. 2011, 52(8), 869–74. [Google Scholar]
- Soares, M.B.; Santos, R.R. Current status and perspectives of cell therapy in Chagas disease. Mem. Inst. Oswaldo Cruz 2009, 104 Suppl 1, 325–32. [Google Scholar] [CrossRef]
- Jasmin; Torres, A.L.; Nunes, H.M.; Passipieri, J.A.; Jelicks, L.A.; Gasparetto, E.L.; et al. Optimized labeling of bone marrow mesenchymal cells with superparamagnetic iron oxide nanoparticles and in vivo visualization by magnetic resonance imaging. J. Nanobiotechnology 2011, 9, 4. [Google Scholar] [CrossRef]
- Soares, M.B.; Lima, R.S.; Rocha, L.L.; Takyia, C.M.; Pontes-de-Carvalho, L.; de Carvalho, A.C. Transplanted bone marrow cells repair heart tissue and reduce myocarditis in chronic chagasic mice. Am. J. Pathol. 2004, 164(2), 441–7. [Google Scholar] [CrossRef]
- Iacobas, D.A.; Iacobas, S.; Tanowitz, H.B.; Campos de Carvalho, A.; Spray, D.C. Functional genomic fabrics are remodeled in a mouse model of Chagasic cardiomyopathy and restored following cell therapy. Microbes Infect. 2018, 20(3), 185–195. [Google Scholar] [CrossRef]
- KEGG. Chemokine signaling pathway. Available online: https://www.kegg.jp/pathway/mmu04062 (accessed on March 1st 2026).
- Anversa, P.; Li, P.; Zhang, X.; Olivetti, G.; Capasso, J.M. Ischaemic myocardial injury and ventricular remodelling. Cardiovasc Res. 1993, 27(2), 145–57. [Google Scholar] [CrossRef]
- Sutton, M.G.; Sharpe, N. Left ventricular remodeling after myocardial infarction: pathophysiology and therapy. Circulation 2000, 101, 2981–2988. [Google Scholar] [CrossRef] [PubMed]
- Mehta, J.L.; Li, Y. Inflammation in ischemic heart disease: Response to tissue injury or a pathogenetic villain? Cardiovasc. Res. 1999, 43(2), 291–299. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.W.; Kang, E.J.; Hwang, S.H.; Kim, S.M. Cardiac CT for the Assessment of Ischemic Cardiomyopathy. J. Korean Soc. Radiol. 2026, 87(2), 181–197. [Google Scholar] [CrossRef]
- Lachtermacher, S.; Esporcatte, B.L.; Montalvão, F.; Costa, P.C.; Rodrigues, D.C.; Belem, L.; et al. Cardiac gene expression and systemic cytokine profile are complementary in a murine model of post-ischemic heart failure. Braz. J. Med. Biol. Res. 2010, 43(4), 377–89. [Google Scholar] [CrossRef]
- Frasure-Smith, N.; Lespérance, F.; Talajic, M. Depression and 18-month prognosis after myocardial infarction. Circulation. 1995, 91, 999–1005. [Google Scholar] [CrossRef]
- Patel, P.; Yang, F.; Iacobas, D.A.; Xi, L. Mental disorders after myocardial infarction: potential mediator role for chemokines in heart-brain interaction? J. Geriatr. Cardiol. 2024, 21(9), 913–926. [Google Scholar] [CrossRef]
- Indian, P.A.; Trivedi, M.V.; Gaikwad, A.B. Deciphering the role of mitochondrial cytochrome C oxidase subunit 4 in cardiac health and disease. Life Sci. 2026, 391, 124295. [Google Scholar] [CrossRef] [PubMed]
- Kurian, G.A.; Gino, E.R. Strategic targeting of mitochondria in ischemic heart disease: mechanisms and emerging therapies. Expert Rev. Cardiovasc Ther. 2026, 24(3), 185–205. [Google Scholar] [CrossRef] [PubMed]
- Iacobas, D.A.; Xi, L. Theory and Applications of the (Cardio) Genomic Fabric Approach to Post-Ischemic and Hypoxia-Induced Heart Failure. J. Pers. Med. 2022, 12, 1246. [Google Scholar] [CrossRef] [PubMed]
- Lachtermacher, S.; Esporcatte, B.L.; Fortes Fda, S.; Rocha, N.N.; Montalvão, F.; Costa, P.C.; Belem, L.; Rabischoffisky, A.; Faria Neto, H.C.; Vasconcellos, R.; Iacobas, D.A.; Iacobas, S.; Spray, D.C.; Thomas, N.M.; Goldenberg, R.C.; de Carvalho, A.C. Functional and transcriptomic recovery of infarcted mouse myocardium treated with bone marrow mononuclear cells. Stem Cell Rev. Rep. 2012, 8(1), 251–61. [Google Scholar] [CrossRef]
- Liu, L.; Jiang, Q.; Du, C.; Yang, T.; Zhou, L.; Chen, J.; et al. Ankrd1 regulates endogenous cardiac regeneration in mice by modulating cyclin D1. Eur. J. Pharmacol. 2024, 983, 177005. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wang, X.; Bi, Y.; Zhang, M.; Xiong, W.; Hu, X.; et al. SNX5-Rab11a protects against cardiac hypertrophy through regulating LRP6 membrane translocation. J. Mol. Cell Cardiol. 2024, 194, 46–58. [Google Scholar] [CrossRef]
- Kovacs, G.; Bartolome, S.; Denton, C.P.; Gatzoulis, M.A.; Gu, S.; Khanna, D.; et al. Definition, classification and diagnosis of pulmonary hypertension. Eur. Respir. J. 2024, 64(4), 2401324. [Google Scholar] [CrossRef]
- Cao, Y.; Dong, W.; Shi, Y.; Sun, X.; Su, H.; Xie, D.; et al. Conceptual Reconsideration of the Classification of Pulmonary Hypertension: From 5 Groups to 2 Groups. JACC Asia 2026, S2772-3747(26)00112-2. [Google Scholar] [CrossRef]
- Blanca-Jover, E.; Contreras-Chova, F.; Jerez-Calero, A.; Uberos-Fernandez, J.; Pérez-Lara, L. Congenital Heart Disease and Pulmonary Arterial Hypertension: Current Perspectives. Rev. Cardiovasc Med. 2026, 27(3), 48337. [Google Scholar] [CrossRef]
- Qiu, Y.; Lyu, X.; Zhang, D.; Xu, H.; He, X.; Chen, J.; et al. Gut Microbiota in Pulmonary Arterial Hypertension: Murine Models and Human Microbial Signatures, Pathogenic Mechanisms, and Emerging Therapeutic Avenues. Compr. Physiol. 2026, 16(1), e70094. [Google Scholar] [CrossRef]
- Miller, D.M.; Archer, S.L.; Dunham-Snary, K.J. Preclinical models of mitochondrial dysfunction: mtDNA and nuclear-encoded regulators in diverse pathologies. Front Aging 2025, 6, 1585508. [Google Scholar] [CrossRef]
- Kris, L.P.; Dixon, D.L.; Bihari, S.; Carr, J.M. Revisiting the monocrotaline-treated rat as a model of inflammatory lung disease: COVID-19 and future pandemic threats? Anim. Model Exp. Med. 2025, 8(10), 1785–1793. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Li, J.; Wei, R.; Meng, L.; Kong, X.; Wei, K.; et al. Ethyl pyruvate alleviates pulmonary arterial hypertension via PI3K-Akt signaling. Mol. Cell Biochem. 2025, 480(2), 1045–1054. [Google Scholar] [CrossRef]
- Stam, K.; Clauss, S.; Taverne, Y.J.H.J.; Merkus, D. Chronic Thromboembolic Pulmonary Hypertension–What Have We Learned From Large Animal Models. Front Cardiovasc Med. 2021, 8, 574360. [Google Scholar] [CrossRef] [PubMed]
- Rainer, P.P.; Kass, D.A. Old dog, new tricks: novel cardiac targets and stress regulation by protein kinase G. Cardiovasc Res. 2016, 111(2), 154–62. [Google Scholar] [CrossRef]
- Mathew, R.; Altura, B.T.; Altura, B.M. Strain differences in pulmonary hypertensive response to monocrotaline alkaloid and the beneficial effect of oral magnesium treatment. Magnesium 1989, 8(2), 110–6. [Google Scholar] [PubMed]
- Krstic, A.M.; Jones, T.L.M.; Power, A.S.; Ward, M.L. The Monocrotaline Rat Model of Right Heart Disease Induced by Pulmonary Artery Hypertension. Biomedicines. 2024, 12(9), 1944. [Google Scholar] [CrossRef]
- Ferrarini, G.; Canevari, M.; Azzini, V.; Agostoni, P.; Pezzuto, B.; Vignati, C. Physiological Responses to Acute Hypobaric and Normobaric Hypoxia: Differences in Maximal Exercise and Clinical Impact. High Alt. Med. Biol. 2026, 15578682261442067. [Google Scholar] [CrossRef]
- McGowan, J.; Cook, J. Altitude-Induced Pulmonary Hypertension. In StatPearls [Internet]; StatPearls Publishing: Treasure Island (FL), 2026. [Google Scholar] [PubMed]
- Mathew, R.; Huang, J.; Iacobas, S.; Iacobas, D.A. Pulmonary Hypertension Remodels the Genomic Fabrics of Major Functional Pathways. Genes 2020, 11, 126. [Google Scholar] [CrossRef] [PubMed]
- Mathew, R.; Iacobas, S.; Huang, J.; Iacobas, D.A. Metabolic Deregulation in Pulmonary Hypertension. Curr. Issues Mol. Biol. 2023, 45, 4850–4874. [Google Scholar] [CrossRef]
- KEGG Pathways. Vascular smooth muscle contraction. Available online: https://www.kegg.jp/pathway/rno04270 (accessed on March 21st 2026).
- KEGG Pathway. Citrate Cycle (TCA Cycle)—Rattus Norvegicus (Rat). Available online: https://www.genome.jp/kegg-bin/show_pathway?rno00020 (accessed on 2026).
- KEGG Pathway. Fructose and Manose Metabolism. Available online: https://www.genome.jp/kegg-bin/show_pathway?rno00051 (accessed on March 20th 2026).
- Alberti, K.G.; Zimmet, P.; Shaw, J. IDF Epidemiology Task Force Consensus Group. The metabolic syndrome--a new worldwide definition. Lancet 2005, 366(9491), 1059–1062. [Google Scholar] [CrossRef]
- Lakka, H.M.; Laaksonen, D.E.; Lakka, T.A.; Niskanen, L.K.; Kumpusalo, E.; Tuomilehto, J.; et al. The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 2002, 288(21), 2709–2716. [Google Scholar] [CrossRef]
- Ha, V.; Jayalath, V.H.; Cozma, A.I.; Mirrahimi, A.; de Souza, R.J.; Sievenpiper, J.L. Fructose-containing sugars, blood pressure, and cardiometabolic risk: a critical review. Curr. Hypertens. Rep. 2013, 15(4), 281–97. [Google Scholar] [CrossRef]
- Seneff, S.; Wainwright, G.; Mascitelli, L. Is the metabolic syndrome caused by a high fructose, and relatively low fat, low cholesterol diet? Arch. Med. Sci. 2011, 7(1), 8–20. [Google Scholar] [CrossRef] [PubMed]
- Ferder, L.; Ferder, M.D.; Inserra, F. The role of high fructose corn syrup in metabolic syndrome and hypertension. Curr. Hypertens. Res. 2010, 12, 105–112. [Google Scholar] [CrossRef]
- Dai, S.; McNeill, J.H. Fructose-induced hypertension in rats is concentration- and duration-dependent. J. Pharmacol. Toxicol. Methods 1995, 33(2), 101–107. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Yamamoto, D.; Suzuki, T.; Fujii, M.; Suzuki, N.; Fujishiro, M.; et al. High fat and high fructose diet induced intracranial atherosclerosis and enhanced vasoconstrictor responses in non-human primate. Life Sci. 2006, 80(3), 200–204. [Google Scholar] [CrossRef]
- Stirpe, F.; Della Corte, E.; Bonetti, E.; Abbondanza, A.; Abbati, A.; De Stefano, F. Fructose-induced hyperuricaemia. Lancet. 1970, 2(7686), 1310–1311. [Google Scholar] [CrossRef] [PubMed]
- Lê, K.A.; Ith, M.; Kreis, R.; Faeh, D.; Bortolotti, M.; Tran, C.; et al. Fructose overconsumption causes dyslipidemia and ectopic lipid deposition in healthy subjects with and without a family history of type 2 diabetes. Am. J. Clin. Nutr. 2009, 89(6), 1760–1765. [Google Scholar] [CrossRef]
- Eickelmann, C.; Lieder, H.R.; Sturek, M.; Heusch, G.; Kleinbongard, P. Differences in vasomotor function of mesenteric arteries between Ossabaw minipigs with predisposition to metabolic syndrome and Göttingen minipigs. Am. J. Physiol. Heart Circ. Physiol. 2024, 326(2), H408–H417. [Google Scholar] [CrossRef]
- Kehinde, S.A.; Qaisrani, Z.N.; Pattanayaiying, R.; Lin, W.P.; Lay, B.B.; Phyo, K.Y.; et al. Preclinical Evidence of Curcuma longa Linn. as a Functional Food in the Management of Metabolic Syndrome: A Systematic Review and Meta-Analysis of Rodent Studies. Biomedicines 2025, 13(8), 1911. [Google Scholar] [CrossRef]
- Raju, S.U.; Shanmugapriyan, S.; Chakradhar, T.; Jaikumar, S.; Sayana, S.B.; Muninathan, N. Antioxidant Effects of Aqueous Bidens pilosa in Fructose-Fed Rats. Cureus 2025, 17(11), e97222. [Google Scholar] [CrossRef]
- Niewiadomska, J.; Gajek-Marecka, A.; Gajek, J.; Noszczyk-Nowak, A. Biological Potential of Polyphenols in the Context of Metabolic Syndrome: An Analysis of Studies on Animal Models. Biology 2022, 11(4), 559. [Google Scholar] [CrossRef] [PubMed]
- Tvarijonaviciute, A.; Ceron, J.J.; Holden, S.L.; Cuthbertson, D.J.; Biourge, V.; Morris, P.J.; et al. Obesity-related metabolic dysfunction in dogs: a comparison with human metabolic syndrome. BMC Vet. Res. 2012, 8, 147. [Google Scholar] [CrossRef]
- Baharuddin, B. The Impact of Fructose Consumption on Human Health: Effects on Obesity, Hyperglycemia, Diabetes, Uric Acid, and Oxidative Stress With a Focus on the Liver. Cureus. 2024, 16(9), e70095. [Google Scholar] [CrossRef]
- Tappy, L.; Le, K.A. Metabolic effects of fructose and the world wide increase in obesity. Physiol. Rev. 2010, 90(1), 23–46. [Google Scholar] [CrossRef]
- Kertowidjojo, E.; Iacobas, D.A. Insulin resistance and the metabolic syndrome severity—a mathematical model. Acad. Romanian Sci. Ser. Biol. Sci. 2022, 11(1), 91–103. [Google Scholar] [CrossRef]
- Ferreira, M.D.S.; Maldonado, R.A.; Farani, P.S.G. Chagas Disease in the 21st Century: Global Spread, Ecological Shifts, and Research Frontiers. Biology . 2025, 14(11), 1631. [Google Scholar] [CrossRef]
- Ying, H.; Fan, H.; Wang, Y.; Jiang, R.; Cai, D.; Cheng, H.; et al. Connexin43 Deficiency Leads to Ventricular Arrhythmias by Reprogramming Proline Metabolism. Adv. Sci. (Weinh) 2026, 13(19), e16090. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zanella, F.; Ellis, M.W.; Bradford, W.H.; Gutierrez-Lara, E.J.; Wang, T.M.; et al. Connexin-43 Restoration Alleviates Desmosomal Arrhythmogenic Cardiomyopathy. Circ. Heart Fail. 2026, 19(4), e013801. [Google Scholar] [CrossRef] [PubMed]
- Žigová, L.; Hrubá, O.; Kyselovic, J.; Gažová, A. Connexin 43 in Pathophysiology of Cardiac Diseases: From Molecular Mechanisms to Therapeutic Strategies. Physiol. Res. 2025, 74(6), 909–921. [Google Scholar] [CrossRef]
- Iacobas, D.A.; Iacobas, S.; Li, W.E.; Zoidl, G.; Dermietzel, R.; Spray, D.C. Genes controlling multiple functional pathways are transcriptionally regulated in connexin43 null mouse heart. Physiol. Genom. 2005, 20(3), 211–23. [Google Scholar] [CrossRef]
- Ybanez, T.; Ingles, J.; Du Toit, E.; Peart, J. Cardioprotection Through Mitochondrial Modulation: A Systematic Review of Pharmacological Interventions in Animal Models of I/R Injury. Cardiovasc Drugs Ther. 4 Mar 2026. [CrossRef]
- Golforoush, P.; Yellon, D.M.; Davidson, S.M. Mouse models of atherosclerosis and their suitability for the study of myocardial infarction. Basic Res. Cardiol. 2020, 115(6), 73. [Google Scholar] [CrossRef]
- Bouchet, C.; Guibert, C.; Freund-Michel, V. Le facteur de croissance des nerfs (NGF) dans l’hypertension pulmonaire (HTP) [Nerve growth factor (NGF) in pulmonary hypertension (PH)]. In Rev Mal Respir.; French, 2024; Volume 41, 4, pp. 265–268. [Google Scholar] [CrossRef]
- Ning, S.; Guo, X.; Zhu, Y.; Li, C.; Li, R.; Meng, Y.; et al. The mechanism of NRF2 regulating cell proliferation and mesenchymal transformation in pulmonary hypertension. Int. J. Biol. Macromol. 2024, 275 Pt 1, 133514. [Google Scholar] [CrossRef] [PubMed]
- Mani, S.; Berger, S.I. Genetic Variants Associated with Persistent Pulmonary Hypertension of Newborn: A Systematic Review. Neonatology Epub ahead of print. 2026, 1–12. [Google Scholar] [CrossRef]
- Bousseau, S.; Gu, S.; Lahm, T. Biological and Clinical Implications of Sex Differences in Right Heart Failure Associated With Pulmonary Arterial Hypertension. Arterioscler. Thromb. Vasc. Biol. 2026. [Google Scholar] [CrossRef] [PubMed]
- Verkest, K.R. Is the metabolic syndrome a useful clinical concept in dogs? A review of the evidence. Vet. J. 2014, 199(1), 24–30. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



