Submitted:
01 May 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Rethinking Atopy: From Classical IgE-Mediated Allergy to a Multilayered Immune Concept
3. IgE-Dependent Cutaneous Immune Network: FcεRI-Expressing Dendritic Cells, Mast Cells, and Basophils
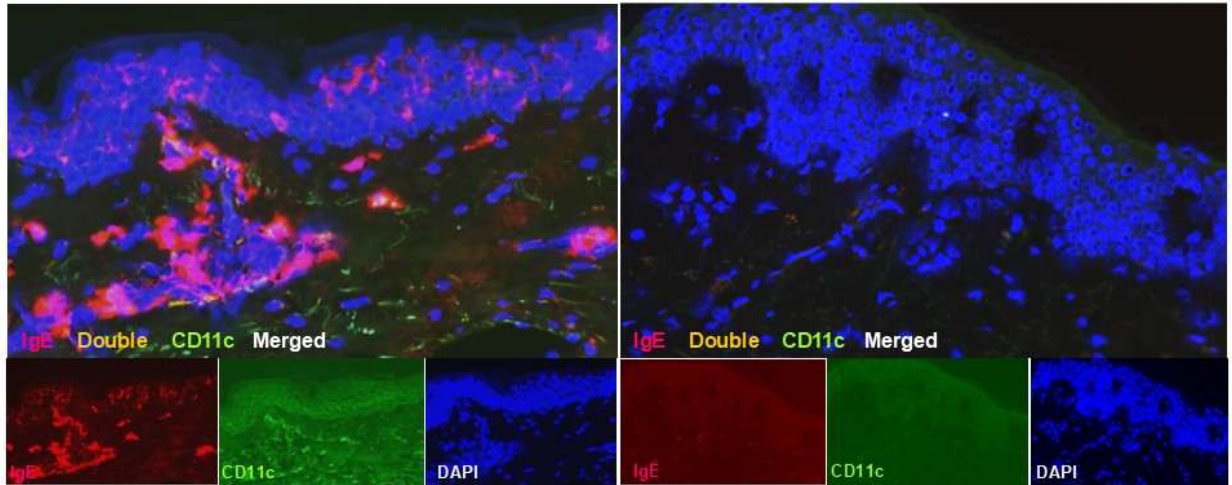
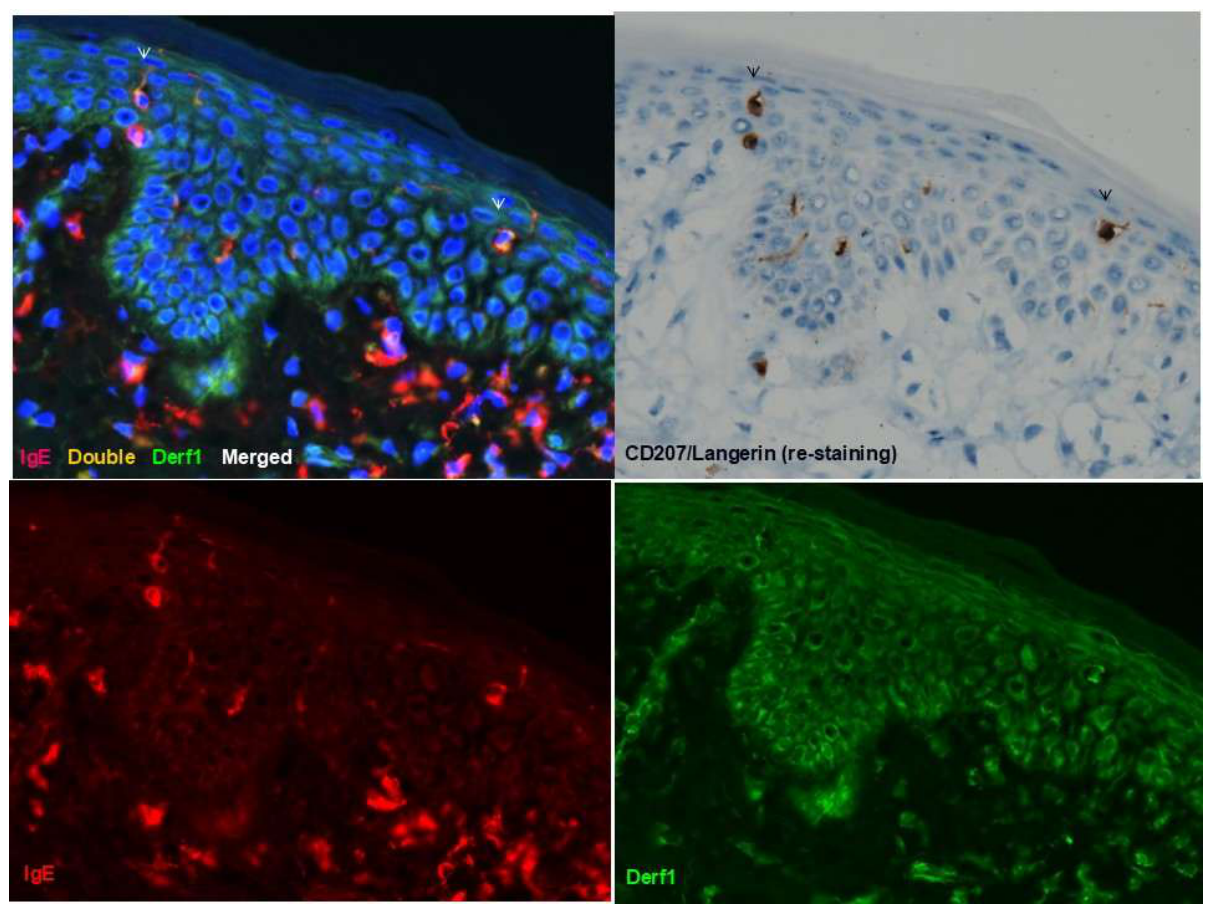
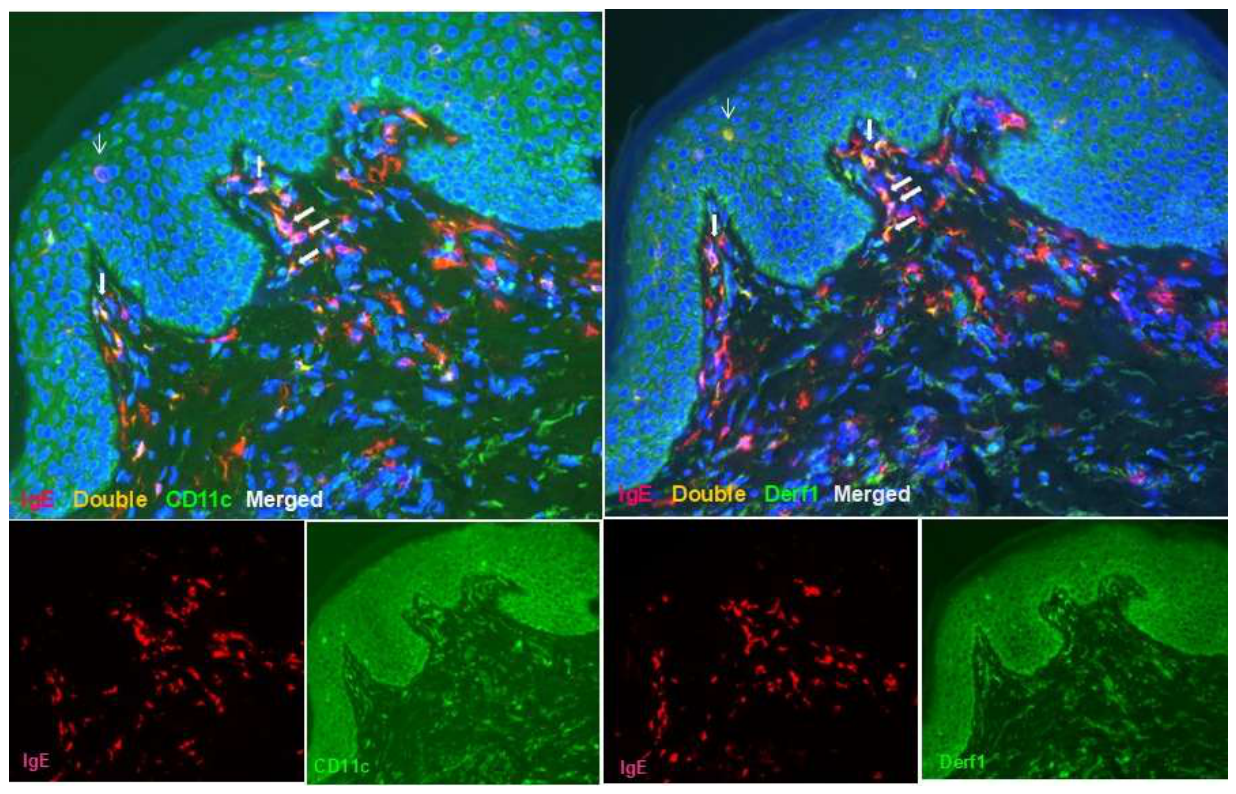
3.1. Langerhans Cells: Silent Antigen Collectors and Central Players in AD Pathogenesis
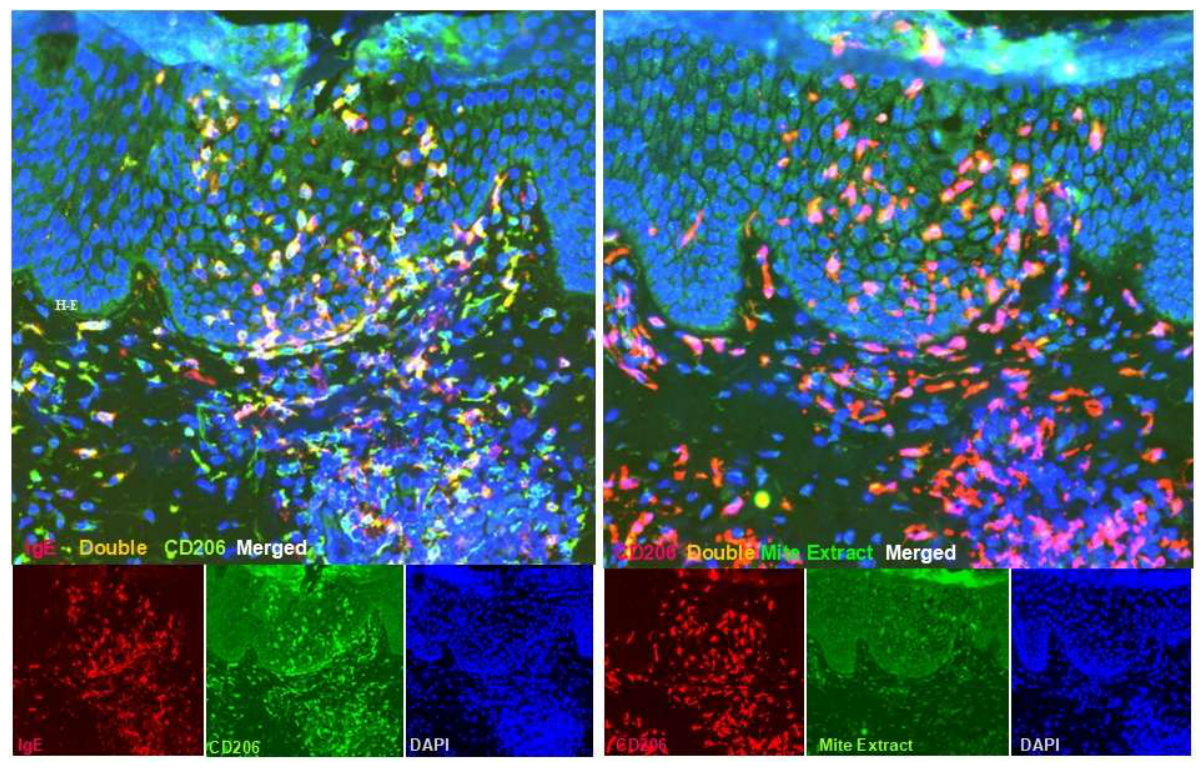
3.2. Inflammatory Dermal Dendritic Cells: Key Drivers of Th2-Skewing in the Dermis
3.3. Inflammatory Dendritic Epidermal Cells: IgE-Dependent Amplifiers Linking Delayed-Type Hypersensitivity to Spongiosis in Atopic Dermatitis
3.4. Mast Cells and Basophils: IgE-Dependent Innate–Adaptive Amplifiers
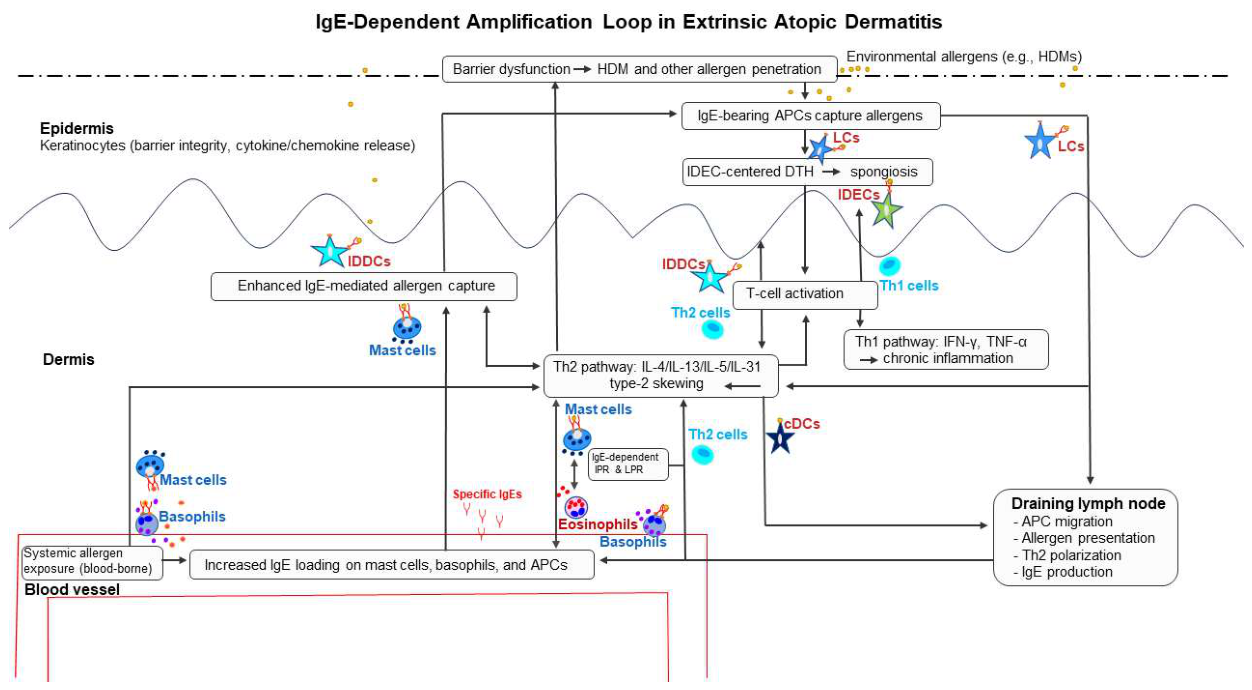
4. The IgE-Dependent Amplification Loop: An Integrated Pathomechanistic Model
4.1. Architecture of the IgE-Dependent Amplification Loop
4.2. Integrating the IgE-Dependent Amplification Loop Into the Broader Pathophysiology of AD
5. Therapeutic Implications and Future Directions Derived from the IgE-Dependent Amplification Loop
5.1. IL-4/IL-13 Blockade as Modulators of the IgE-Dependent Amplification Loop
5.1.1. Dupilumab-Induced Immune Remodeling
5.1.2. Why Dupilumab Outperforms IL-13–Only Blockade
5.1.3. Dupilumab in Combination with Adjunctive Therapies
5.2. Positioning the IgE-Dependent Amplification Loop in the Era of Next-Generation Targeted Therapies
- IL-4Rα blockade partially suppresses but does not eliminate the IgE-dependent amplification loop.
- Therapies that fail to reduce IgE or FcεRI signaling may show limited efficacy in IgE-high extrinsic AD.
- Combination approaches targeting both type 2 cytokines and IgE pathways may offer superior disease control.
- The IgE-dependent amplification loop should be recognized as a therapeutic axis distinct from type 2 cytokine signaling.
- dual IL-4/IL-13–IgE pathway inhibition,
- FcεRI-targeted therapies,
- CD23-modulating approaches, and
- precision-medicine frameworks identifying IgE-high or FcεRI-high endotypes.
6. Conclusion and Overall Synthesis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Bieber, T.; Paller, A. S.; Kabashima, K.; Feely, M.; Rueda, M. J.; Ross Terres, J. A.; Wollenberg, A. Atopic dermatitis: pathomechanisms and lessons learned from novel systemic therapeutic options. J. Eur. Acad. Dermatol. Venereol. 2022, 36, 1432–1449. [Google Scholar] [CrossRef]
- Guttman-Yassky, E.; Renert-Yuval, Y.; Brunner, P. Atopic dermatitis. Lancet 2025, 405(10478), 583–596. [Google Scholar] [CrossRef]
- Hill, L.W.; Sulzberger, M.B. Evolution of atopic dermatitis. Arch. Derm. Syphilol. 1935, 32, 451–463. [Google Scholar] [CrossRef]
- Bruijnzeel-Koomen, C.; van Wichen, D.F.; Toonstra, J.; Berrens, L.; Bruynzeel, P.L.B. The presence of IgE molecules on epidermal Langerhans cells in patients with atopic dermatitis. Arch. Derm. Res. 1986, 278, 199–205. [Google Scholar] [CrossRef]
- Bieber, T. Fc epsilon RI-expressing antigen-presenting cells: New players in the atopic game. Immunol. Today 1997, 18, 311–313. [Google Scholar] [CrossRef] [PubMed]
- Leung, D.Y. Pathogenesis of atopic dermatitis. J. Allergy Clin. Immunol. 1999, 104, 99–108. [Google Scholar] [CrossRef]
- Palmer, C. N. A.; Irvine, A. D.; Terron-Kwiatkowski, A.; Zhao, Y.; Liao, H.; Lee, S. P.; Goudie, D. R.; Sandilands, A.; Campbell, L. E.; Smith, F. J. D.; et al. Common loss-of-function variants of the epidermal barrier protein filaggrin are a major predisposing factor for atopic dermatitis. Nat. Genet. 2006, 38, 441–446. [Google Scholar] [CrossRef]
- Beck, L.A.; Thaçi, D.; Hamilton, J.D.; Graham, N.M.; Bieber, T.; Rocklin, R.; Ming, J.E.; Ren, H.; Kao, R.; Simpson, E.; et al. Dupilumab treatment in adults with moderate-to-severe atopic dermatitis. N. Engl. J. Med. 2014, 371, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Thomsen, S.F.; Lacour, J.-P.; Jaumont, X.; Lazarewicz, S. Targeting immunoglobulin E in atopic dermatitis: A review of the existing evidence. World Allergy Organ. J. 2021, 14, 100519. [Google Scholar] [CrossRef] [PubMed]
- Honda, T.; Kabashima, K. Reconciling innate and acquired immunity in atopic dermatitis. J. Allergy Clin. Immunol. 2020, 145, 1136–1137. [Google Scholar] [CrossRef] [PubMed]
- Davidson, W.F.; Leung, D.Y.M.; Beck, L.A.; Berin, C.M.; Boguniewicz, M.; Busse, W.W.; Chatila, T.A.; Geha, R.S.; Gern, J.E.; Guttman-Yassky, E.; et al. Report from the National Institute of Allergy and Infectious Diseases Workshop on “Atopic Dermatitis and the Atopic March: Mechanisms and Interventions”. J. Allergy Clin. Immunol. 2019, 143, 894–913. [Google Scholar] [CrossRef]
- Furue, M.; Chiba, T.; Tsuji, G.; Ulzii, D.; Kido-Nakahara, M.; Nakahara, T.; Kadono, T. Atopic dermatitis: immune deviation, barrier dysfunction, IgE autoreactivity and new therapies. Allergol. Int. 2017, 66, 398–403. [Google Scholar] [CrossRef]
- Suárez-Fariñas, M.; Dhingra, N.; Gittler, J.; Shemer, A.; Cardinale, I.; de Guzman Strong, C.; Krueger, J. G.; Guttman-Yassky, E. Intrinsic atopic dermatitis shows similar TH2 and higher TH17 immune activation compared with extrinsic atopic dermatitis. J. Allergy Clin. Immunol. 2013, 132, 361–370. [Google Scholar] [CrossRef]
- Tanei, R.; Hasegawa, Y. Immunological Pathomechanisms of Spongiotic Dermatitis in Skin Lesions of Atopic Dermatitis. Int. J. Mol. Sci. 2022, 23, 6682. [Google Scholar] [CrossRef] [PubMed]
- Tanei, R.; Hasegawa, Y. Immunohistopathological Analysis of Immunoglobulin E-Positive Epidermal Dendritic Cells with House Dust Mite Antigens in Naturally Occurring Skin Lesions of Adult and Elderly Patients with Atopic Dermatitis. Dermatopathology 2021, 8, 426–441. [Google Scholar] [CrossRef]
- Knol, E. F.; van Neerven, R. J. J. IgE versus IgG and IgA: Differential roles of allergen specific antibodies in sensitization, tolerization, and treatment of Allergies. Immunol. Rev. 2024, 328, 314–333. [Google Scholar] [CrossRef] [PubMed]
- Coca, A. F.; Grove, E. F. Studies in Hypersensitiveness: XIII. A Study of the Atopic Reagins. J. Immunol. 1925, 10, 445–464. [Google Scholar] [CrossRef]
- Ishizaka, K.; Ishizaka, T.; Hornbrook, M. M. Physicochemical properties of reaginic antibody. V. Correlation of reaginic activity with gamma-E-globulin antibody. J. Immunol. 1966, 97, 840–853. [Google Scholar] [CrossRef] [PubMed]
- Hanifin, J.M.; Rajka, G. Diagnostic features of atopic dermatitis. Acta Derm. 1980, 92, 44–47. [Google Scholar] [CrossRef]
- Okada, M.; Terui, T.; Honda, M.; Tanaka, M.; Chikama, R.; Tabata, N.; Takahashi, K.; Tagami, H. Cutaneous late phase reaction in adult atopic dermatitis patients with high serum IgE antibody to Dermatophagoides farinae: Correlation with IL-5 production by allergen-stimulated peripheral blood mononuclear cells. J. Derm. Sci. 2002, 29, 73–84. [Google Scholar] [CrossRef]
- Murota, H.; Katayama, I. Exacerbating factors of itch in atopic dermatitis. Allergol. Int. 2017, 66, 8–13. [Google Scholar] [CrossRef]
- Nattkemper, L. A.; Tey, H. L.; Valdes-Rodriguez, R.; Lee, H.; Mollanazar, N. K.; Albornoz, C.; Sanders, K. M.; Yosipovitch, G. The Genetics of Chronic Itch: Gene Expression in the Skin of Patients with Atopic Dermatitis and Psoriasis with Severe Itch. J. Invest. Dermatol. 2018, 138, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Tominaga, M.; Takamori, K. Peripheral itch sensitization in atopic dermatitis. Allergol. Int. 2022, 71, 265–277. [Google Scholar] [CrossRef] [PubMed]
- De Benedetto, A.; Agnihothri, R.; McGirt, L.Y.; Bankova, L.G.; Beck, L.A. Atopic dermatitis: a disease caused by innate immune defects? J. Invest. Dermatol. 2009, 129, 14–30. [Google Scholar] [CrossRef]
- Kong, H.H.; Oh, J.; Deming, C.; Conlan, S.; Grice, E.A.; Beatson, M.A.; Nomicos, E.; Polley, E.C.; Komarow, H.D.; NISC Comparative Sequence Program. Temporal shifts in the skin microbiome associated with disease flares and treatment in children with atopic dermatitis. Genome Res. 2012, 22, 850–859. [Google Scholar] [CrossRef]
- Kabashima, K. New concept of the pathogenesis of atopic dermatitis: interplay among the barrier, allergy, and pruritus as a trinity. J. Dermatol. Sci. 2013, 70, 3–11. [Google Scholar] [CrossRef]
- Torres, T.; Mendes-Bastos, P.; Cruz, M.J.; Duarte, B.; Filipe, P.; Lopes, M.J.P.; Gonçalo, M. Interleukin-4 and Atopic Dermatitis: Why Does it Matter? A Narrative Review. Dermatol. Ther. (Heidelb) 2025, 15, 579–597. [Google Scholar] [CrossRef]
- Liu, Fu-Tong; Goodarzi, F.-T.; Chen, H.H.-Y. IgE, Mast Cells, and Eosinophils in Atopic Dermatitis. Clin. Rev. Allerg. Immunol. 2011, 41, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Miyake, K.; Shibata, S.; Yoshikawa, S.; Karasuyama, H. Basophils and their effector molecules in allergic disorders. Allergy 2021, 76, 1693–1706. [Google Scholar] [CrossRef]
- Liu, C.; Zhang, X.; Xiang, Y.; Qu, X.; Liu, H.; Liu, C.; Tan, M.; Jiang, J.; Qin, X. Role of epithelial chemokines in the pathogenesis of airway inflammation in asthma (Review). Mol. Med. Rep. 2018, 17, 6935–6941. [Google Scholar] [CrossRef]
- Charlesworth, E.N.; Hood, A. F.; Soter, N. A.; Kagey-Sobotka, A.; Norman, P. S.; Lichtenstein, L. M. Cutaneous Late-Phase Response to Allergen. Mediator Release and Inflammatory Cell Infiltration. J. Clin. Invest. 1989, 83, 1519–1526. [Google Scholar] [CrossRef]
- Tanei, R.; Hasegawa, Y. Immunohistopathological Analysis of Spongiosis Formation in Atopic Dermatitis Compared with Other Skin Diseases. Dermatopathology 2025, 12, 23. [Google Scholar] [CrossRef]
- Pan, Y.; Hochgerner, M.; Cichoń, M. A.; Benezeder, T.; Bieber, T.; Wolf, P. Langerhans cells: Central players in the pathophysiology of atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2025, 39, 278–289. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T.; Dannenberg, B.; Prinz, J.C.; Rieber, E.P.; Stolz, W.; Braun-Falco, O.; Ring, J. Occurrence of IgE-bearing epidermal Langerhans cells in atopic eczema: A study of the time course of the lesions and with regard to the IgE serum level. J. Investig. Dermatol. 1989, 93, 215–219. [Google Scholar] [CrossRef]
- Mudde, G.C.; Van Reijsen, F.C.; Boland, G.J.; de Gast, G.C.; Bruijnzeel, P.L.; Bruijnzeel-Koomenet, C.A. Allergen presentation by epidermal Langerhans’ cells from patients with atopic dermatitis is mediated by IgE. Immunology 1990, 69, 335–341. [Google Scholar]
- Novak, N.; Bieber, T. Antigen-Presenting Cells. In Atopic Dermatitis, 2nd ed.; Bieber, T., Leung, D.Y., Eds.; Informa Healthcare: London, UK, 2009; pp. 149–156. [Google Scholar]
- Maeda, K.; Yamamoto, K.; Tanaka, Y.; Anan, A.; Yoshida, H. House dust mite (HDM) antigen in naturally occurring lesions of atopic dermatitis (AD): The relationship between HDM antigen in the skin and HDM antigen-specific IgE antibody. J. Dermatol. Sci. 1992, 3, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Mori, T.; Ishida, K.; Mukumoto, S.; Yamada, Y.; Imokawa, G.; Kabashima, K.; Kobayashi, M.; Bito, T.; Nakamura, M.; Ogasawara, K.; et al. Comparison of skin barrier function and sensory nerve electric current perception threshold between IgE-high extrinsic and IgE-normal intrinsic types of atopic dermatitis. Br. J. Dermatol. 2010, 162, 83–90. [Google Scholar] [CrossRef]
- T Tanaka, T.; Tanaka, Y.; Kazumori, S.; Ohno, M.; Akahoshi, Y.; Kanda, G.; Anan, S.; Yoshida, H. A Case of Contact Urticaria from House Dust Mite Extract (Dermatophagoidesfarinae, DF). Skin. Res. (in Japanese). 1986, 28, 344–347. [Google Scholar]
- Darsow, U.; Vieluf, D.; Ring, J. Atopy patch test with different vehicles and allergen concentrations: an approach to standardization. J. Allergy Clin. Immunol. 1995, 95, 677–684. [Google Scholar] [CrossRef]
- Teplitsky, V.; Mumcuoglu, K. Y.; Babai, I.; Dalal, I.; Cohen, R.; Tanay, A. House dust mites on skin, clothes, and bedding of atopic dermatitis patients. Int. J. Dermatol. 2008, 47, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Yasueda, H.; Saito, A.; Nishioka, K.; Kutsuwada, K.; Akiyama, K. Measurement of Dermatophagoides mite allergens on bedding and human skin surfaces. Clin. Exp. Allergy 2003, 33, 1654–1658. [Google Scholar] [CrossRef] [PubMed]
- Tanei, R.; Hasegawa, Y. Double-positive Immunoglobulin E+ and Dermatophagoides farinae antigen+ dendritic cells are observed in Skin Lesions of Older Adults with Atopic Dermatitis: An Immunohistological Study. Dermatol. Clin. Res. 2017, 3, 134–150. [Google Scholar]
- Ohki, O.; Yokozeki, H.; Katayama, I.; Umeda, T.; Azuma, M.; Okumura, K.; Nishioka, K. Functional CD86 (B7-2/B70) is predominantly expressed on Langerhans cells in atopic dermatitis. Br. J. Dermatol. 1997, 136, 838–845. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Soma, Y.; Ra, C.; Kawa, Y.; Kubota, Y.; Mizoguchi, M. Enhanced expression of the high-affinity receptor for IgE (Fc(epsilon)RI) associated with decreased numbers of Langerhans cells in the lesional epidermis of atopic dermatitis. J. Dermatol. Sci. 2001, 27, 156–161. [Google Scholar] [CrossRef]
- Tamminga, S.M.; Van Der Wal, M.M.; Saager, E. S.; Van Der Gang, L.F.; Boesjes, C. M.; Hendriks, A.; Pannekoek, Y.; De Bruin, M. S.; Van Wijk, F.; Van Sorgem, N.M. Single-cell sequencing of human Langerhans cells identifies altered gene expression profiles in patients with atopic dermatitis. Immunohorizons 2025, 24, 9. [Google Scholar] [CrossRef]
- Guttman-Yassky, E.; Lowes, M.A.; Fuentes-Duculan, J.; Whynot, J.; Novitskaya, I.; Cardinale, I.; Haider, A.; Khatcherian, A.; Carucci, J.A.; Bergman, R.; et al. Major differences in inflammatory dendritic cells and their products distinguish atopic dermatitis from psoriasis. J. Allergy Clin. Immunol. 2007, 119, 1210–1217. [Google Scholar] [CrossRef]
- Zaba, L. C.; Krueger, J.G.; Lowes, A. Resident and "inflammatory" dendritic cells in human skin. J. Invest. Dermatol. 2009, 129, 302–308. [Google Scholar] [CrossRef]
- Hespel, C.; Moser, M. Role of inflammatory dendritic cells in innate and adaptive immunity. Eur. J. Immunol. 2012, 42, 2535–2543. [Google Scholar] [CrossRef] [PubMed]
- Novak, N.; Valenta, R.; Bohle, B.; Laffer, S.; Haberstok, J.; Kraft, S.; Bieber, T. FceRI engagement of Langerhans cell–like dendritic cells and inflammatory dendritic epidermal cell–like dendritic cells induces chemotactic signals and different T-cell phenotypes in vitro. J. Allergy Clin. Immunol. 2004, 113, 949–957. [Google Scholar]
- Tanei, R.; Hasegawa, Y.; Sawabe, M. Abundant immunoglobulin E-positive cells in skin lesions support an allergic etiology of atopic dermatitis in the elderly. J. Eur. Acad. Dermatol. Venereol. 2013, 27, 952–960. [Google Scholar] [CrossRef]
- Werfel, T. The role of leukocytes, keratinocytes, and allergen-specific IgE in the development of atopic dermatitis. J. Investig. Dermatol. 2009, 129, 1878–1891. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Wang, Y.; Duramad, O.; Hori, T.; Delespesse, G. J.; Watanabe, N.; Qin, F. X.; Yao, Z.; Cao, W.; Liu, Y. TSLP-activated dendritic cells induce an inflammatory T helper type 2 cell response through OX40 ligand. J. Exp. Med. 2005, 202, 1213–1223. [Google Scholar] [CrossRef]
- Bieber, T. The pro- and anti-inflammatory properties of human antigen-presenting cells expressing the high affinity receptor for IgE (Fc epsilon RI). Immunobiology 2007, 212, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Maurer, D.; Fiebiger, S.; Ebner, C.; et al. Peripheral blood dendritic cells express fc epsilon RI as a complex composed of fc epsilon RI alpha-and fc epsilon RI gamma-chains and can use this receptor for IgE-mediated allergen presentation. J. Immunol. 1996, 157, 607–616. [Google Scholar] [CrossRef]
- Brunner, P. M.; Guttman-Yassky, E.; Leung, D. Y. M. The immunology of atopic dermatitis and its reversibility with broad-spectrum and targeted therapies. J. Allergy Clin. Immunol. 2017, 139, S65–S76. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Wagner, M.; Günther, S.; Towarowski, A.; Tuma, E.; Moderer, M.; Rothenfusser, S.; Wetzel, S.; Endres, S.; Hartmann, G. Plasmacytoid dendritic cells: a new cutaneous dendritic cell subset with distinct role in inflammatory skin diseases. J. Invest. Dermatol. 2002, 119, 1096–1102. [Google Scholar] [CrossRef]
- Baran, W.; Oehrl, S.; Ahmad, F.; Döbel, T.; Alt, C.; Buske-Kirschbaum, A.; Schmitz, M.; Schäkel, K. Phenotype, Function, and Mobilization of 6-Sulfo LacNAc-Expressing Monocytes in Atopic Dermatitis. Front. Immunol. 2018, 9, 1352. [Google Scholar] [CrossRef]
- Wollenberg; Kraft, A.; Hanau, S.; Bieber, D.T. Immunomorphological and ultrastructural characterization of Langerhans cells and a novel, inflammatory dendritic epidermal cell (IDEC) population in lesional skin of atopic eczema. J. Invest. Dermatol. 1996, 106, 446–453. [Google Scholar] [CrossRef]
- Wollenberg, A. Inflammatory dendritic epidermal cells. In Handbook of Atopic Eczema, 2nd ed.; Ring, J., Przybilla, B., Ruzicka, T., Eds.; Springer: Berlin, Germany, 2006; pp. 288–295. [Google Scholar]
- Wang, A.; Fogel, A.L.; Murphy, M.J.; Panse, G.; McGeary, M.K.; McNiff, J.M.; Bosenberg, M.; Vesely, M.D.; Cohen, J.M.; Ko, C.J.; et al. Cytokine RNA In Situ Hybridization Permits Individualized Molecular Phenotyping in Biopsies of Psoriasis and Atopic Dermatitis. JID Innov. 2021, 1, 100021. [Google Scholar] [CrossRef]
- Landheer, J.; Giovannone, B.; Mattson, J.D.; Tjabringa, S.; Bruijnzeel-Koomen, C.A.F.M.; McClanahan, T.; Malefyt, R.D.W.; Knol, E.; Hijnen, D. Epicutaneous application of house dust mite induces thymic stromal lymphopoietin in nonlesional skin of patients with atopic dermatitis. J. Allergy Clin. Immunol. 2013, 132, 1252–1254. [Google Scholar] [CrossRef]
- Katoh, N.; Hirano, S.; Suehiro, M.; Masuda, K.; Kishimoto, S. The characteristics of patients with atopic dermatitis demonstrating a positive reaction in a scratch test after 48 hours against house dust mite antigen. J. Dermatol. 2004, 31, 720–726. [Google Scholar] [CrossRef]
- He, H.; Suryawanshi, H.; Morozov, P.; Gay-Mimbrera, J.; Del Duca, E.; Kim, H.J.; Kameyama, N.; Estrada, Y.; Der, E.; Krueger, J.G.; et al. Single-cell transcriptome analysis of human skin identifies novel fibroblast subpopulation and enrichment of immune subsets in atopic dermatitis. J. Allergy Clin. Immunol. 2020, 145, 1615–1628. [Google Scholar] [CrossRef] [PubMed]
- Charlesworth, E. N.; Hood, A. F.; Soter, N. A.; Kagey-Sobotka, A.; Norman, P. S.; Lichtenstein, L. M. Cutaneous late-phase response to allergen. Mediator release and inflammatory cell infiltration. J. Clin. Invest. 1989, 83, 1519–1526. [Google Scholar] [CrossRef]
- Nakajima, T.; Inagaki, N.; Tanaka, H.; Tanaka, A.; Yoshikawa, M.; Tamari, M.; Hasegawa, K.; Matsumoto, K.; Tachimoto, H.; Ebisawa, M.; et al. Marked increase in CC chemokine gene expression in both human and mouse mast cell transcriptomes following Fcepsilon receptor I cross-linking: an interspecies comparison. Blood 2002, 100, 3861–3868. [Google Scholar] [CrossRef]
- Mitchell, E.B.; Crow, J.; Chapman, M.D.; Jouhal, S.S.; Pope, F.M.; Platts-Mills, T.A. Basophils in allergen-induced patch test sites in atopic dermatitis. Lancet 1982, 16, 127–130. [Google Scholar] [CrossRef]
- Ito, Y.; et al. Basophil recruitment and activation in inflammatory skin diseases. Allergy 2011, 66, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Santos, A. F.; Shreffler, W. G. Road map for the clinical application of the basophil activation test in food allergy. Clin. Exp. Allergy 2017, 47, 1115–1124. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, Y.; Imamura, S.; Fukumoto, A.; Lee, K.; Nishitani, N.; Oda, Y.; Washio, K.; Fukunaga, A. Case of oral mite anaphylaxis: Contamination of wheat flour by mites determined by enzyme-linked immunosorbent assay. J. Dermatol. 2022, 49, e59–e60. [Google Scholar] [CrossRef]
- Röckmann, H.; van Geel, M. J.; Knulst, A. C.; Huiskes, J.; Bruijnzeel-Koomen, C. A.; de Bruin-Weller, M. S. Food allergen sensitization pattern in adults in relation to severity of atopic dermatitis. Clin. Transl. Allergy 2014, 4, 9. [Google Scholar] [CrossRef]
- He, H.; Duca, E. D.; Diaz, A.; Kim, H. J.; Gay-Mimbrera, J.; Zhang, N.; Wu, J.; Beaziz, J.; Estrada, Y.; Krueger, J. G. Mild atopic dermatitis lacks systemic inflammation and shows reduced nonlesional skin abnormalities. J. Allergy Clin. Immunol. 2021, 147, 1369–1380. [Google Scholar] [CrossRef]
- Morita, E.; Matsuo, H.; Chinuki, Y.; Takahashi, H.; Dahlström, J.; Tanaka, A. Food-dependent exercise-induced anaphylaxis -importance of omega-5 gliadin and HMW-glutenin as causative antigens for wheat-dependent exercise-induced anaphylaxis-. Allergol. Int. 2009, 58, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Tanei, R. Clinical Characteristics, Treatments, and Prognosis of Atopic Eczema in the Elderly. J. Clin. Med. 2015, 4, 979–997. [Google Scholar] [CrossRef] [PubMed]
- Sallmann, E.; Reininger, B.; Brandt, S.; Duschek, N.; Hoflehner, E.; Garner-Spitzer, E.; Platzer, B.; Dehlink, E.; Hammer, M.; Holcmann, M.; et al. High-Affinity IgE Receptors on Dendritic Cells Exacerbate Th2-Dependent Inflammation. J. Immunol. 2011, 187, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Matsuoka, K.; Taya, C.; Suzuki, H.; Yokozeki, H.; Nishioka, K.; Hirokawa, K.; Etori, M.; Yamashita, M.; Kubota, T.; et al. Basophils play a critical role in the development of IgE-mediated chronic allergic inflammation independently of T cells and mast cells. Immunity 2005, 23, 191–202. [Google Scholar] [CrossRef]
- Redhu, D.; Franke, K.; Aparicio-Soto, M.; Kumari, V.; Pazur, K.; Illerhaus, A.; Hartmann, K.; Worm, M.; Babina, M. Mast cells instruct keratinocytes to produce thymic stromal lymphopoietin: Relevance of the tryptase/protease-activated receptor 2 axis. J. Allergy Clin. Immunol. 2022, 149, 2053–2061. [Google Scholar] [CrossRef]
- Izuhara, K.; Arima, K.; Ohta, S.; Suzuki, S.; Inamitsu, M.; Yamamoto, K. Periostin in Allergic Inflammation. Allergol. Int. 2014, 63, 143–151. [Google Scholar] [CrossRef]
- Shimizu, Y.; Shichijo, M.; Hiramatsu, K.; Takeuchi, M.; Nagai, H.; Takagi, K. Mite antigen-induced IL-4 and IL-13 production by basophils derived from atopic asthma patients. Clin. Exp. Allergy 1998, 28, 497–503. [Google Scholar] [CrossRef]
- Valenta, R.; Seiberler, S.; Natter, S.; Mahler, V.; Mossabeb, R.; Ring, J.; Stingl, G. Autoallergy: A pathogenetic factor in atopic dermatitis? J. Allergy Clin. Immunol. 2000, 105, 432–437. [Google Scholar] [CrossRef]
- Bieber, T. Atopic dermatitis 2.0: from the clinical phenotype to the molecular taxonomy and stratified medicine. Allergy 2012, 67, 1475–82. [Google Scholar] [CrossRef]
- Kitagaki, H.; Ono, N.; Hayakawa, K.; Kitazawa, T.; Watanabe, K.; Shiohara, T. Repeated elicitation of contact hypersensitivity induces a shift in cutaneous cytokine milieu from a T helper cell type 1 to a T helper cell type 2 profile. J. Immunol. 1997, 159, 2484–2491. [Google Scholar] [CrossRef]
- Mellanby, K. Man's reaction to mosquito bites. Nature 1946, 158, 554. [Google Scholar] [CrossRef]
- Campana, R.; Dzoro, S.; Mittermann, I.; Fedenko, E.; Elisyutina, O.; Khaitov, M.; Karaulov, A.; Valentaet, R. Molecular aspects of allergens in atopic dermatitis. Curr. Opin. Allergy Clin. Immunol. 2017, 17, 269–277. [Google Scholar] [CrossRef]
- Katayama, I.; Tanei, R.; Yokozeki, H.; Nishioka, K.; Dohi, Y. Induction of Eczematous Skin Reaction in Experimentally Induced Hyperplastic Skin of Balb/C Mice by Monoclonal Anti-DNP IgE Antibody: Possible Implications for Skin Lesion Formation in Atopic Dermatitis. Int. Arch. Allergy Immunol. 1990, 93, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Ohtani, T.; Memezawa, A.; Okuyama, R.; Sayo, T.; Sugiyama, Y.; Inoue, S.; Aiba, S. Increased hyaluronan production and decreased E-cadherin expression by cytokine-stimulated keratinocytes lead to spongiosis formation. J. Investig. Dermatol. 2009, 129, 1412–1420. [Google Scholar] [CrossRef]
- Oyama, K. Cutaneous late-phase reaction to environmental antigen in patients with atopic dermatitis. Dermatology 1993, 187, 182–185. [Google Scholar] [CrossRef]
- Ogawa-Momohara, M.; Muro, Y.; Murase, C.; Taki, T.; Tanahashi, K.; Yamashita, Y.; Koizumi, H.; Fukaura, R.; Takeichi, T.; Akiyama, M. Allergen-specific IgG4 increase in atopic dermatitis with long-term dupilumab use. Br. J. Dermatol. 2023, 189, 472–474. [Google Scholar] [CrossRef]
- Redhu, D.; Francuzik, W.; Globig, P.; Worm, M. T cell immunophenotypes and IgE responses in patients with moderate-to-severe atopic dermatitis receiving dupilumab. Clin. Transl. Allergy 2025, 15, e70062. [Google Scholar] [CrossRef]
- Ständer, S.; Pinter, A.; Hougeir, F. G.; Guyot, P.; Xu, Y.; Praestgaard, A. H.; Freemantle, N.; Rossi, A. B.; Bégo-Le-Bagousse, G.; Wang, Z.; et al. Dupilumab versus Lebrikizumab Demonstrates Greater Likelihood of Achieving and Maintaining Improvements in Efficacy Outcomes Using a Placebo-Adjusted Indirect Treatment Comparison. Dermatol. Ther. (Heidelb) 2025, 15, 2537–2551. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-S. Comparative efficacy and safety of tralokinumab and dupilumab in moderate-to-severe atopic dermatitis: A narrative review. J. Clin. Med. 2025, 14, 4960. [Google Scholar] [CrossRef] [PubMed]
- Bogacz-Piaseczyńska, A.; Bożek, A.; Krupka-Olek, M.; Kawczyk-Krupka, A.; Zalejska-Fiolka, J.; Canonica, G. W. Dupilumab and House Dust Mite Immunotherapy in Patients with Atopic Dermatitis: A Preliminary Study. Vaccines 2024, 12, 1046. [Google Scholar] [CrossRef]
- Silva, I. C.; Daher, R.; Khattri, S. Combined use of Omalizumab and dupilumab: safety and efficacy data from a large academic center. Arch. Dermatol. Res. 2025, 317, 674. [Google Scholar] [CrossRef] [PubMed]
- Fomina, D. S.; Mukhina, O. A.; Sedova, E. L.; Lebedkina, M. S.; Bobrikova, E. N.; Karaulov, A. V.; Lysenko, M. A.; Renz, H. Case Series: Combination of dupilumab and omalizumab as a way to reduce dupilumab-associated adverse events in severe atopic dermatitis. Front. Allergy 2026, 6, 1696897. [Google Scholar] [CrossRef] [PubMed]
- Sroka-Tomaszewska, J.; Trzeciak, M. Molecular Mechanisms of Atopic Dermatitis Pathogenesis. Int. J. Mol. Sci. 2021, 22, 4130. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



