Submitted:
03 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
Highlights
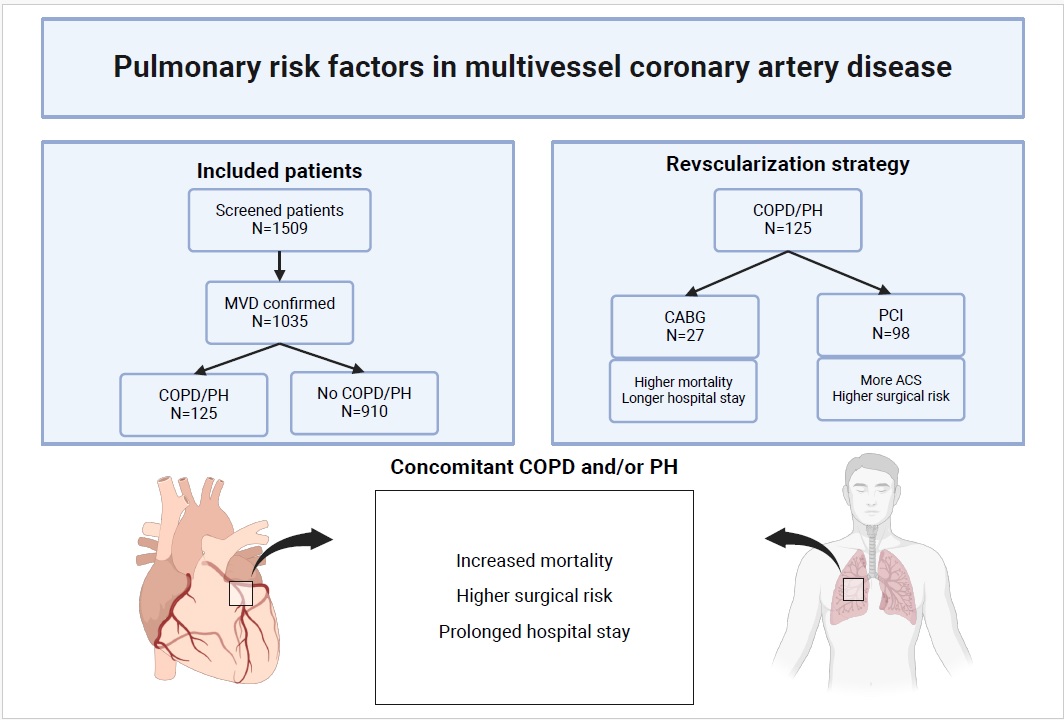
- Patients with severe coronary artery disease (CAD) and concomitant chronic obstructive pulmonary disease (COPD) or pulmonary hypertension (PH) have a higher risk of major adverse cardiac and cerebrovascular events (MACCE) than those with CAD without pulmonary disease.
- Despite high prevalence and clinical importance, there is a paucity of data regarding the impact of COPD and PH on the outcomes of multivessel coronary artery disease (MVD) interventional treatment.
- Using machine learning approach, we demonstrated pulmonary factors associated with adverse outcome after revascularization using coronary artery bypass grafting or percutaneous coronary intervention for the treatment of MVD.
Background
Objectives
Patients and Methods
Statistical Analysis
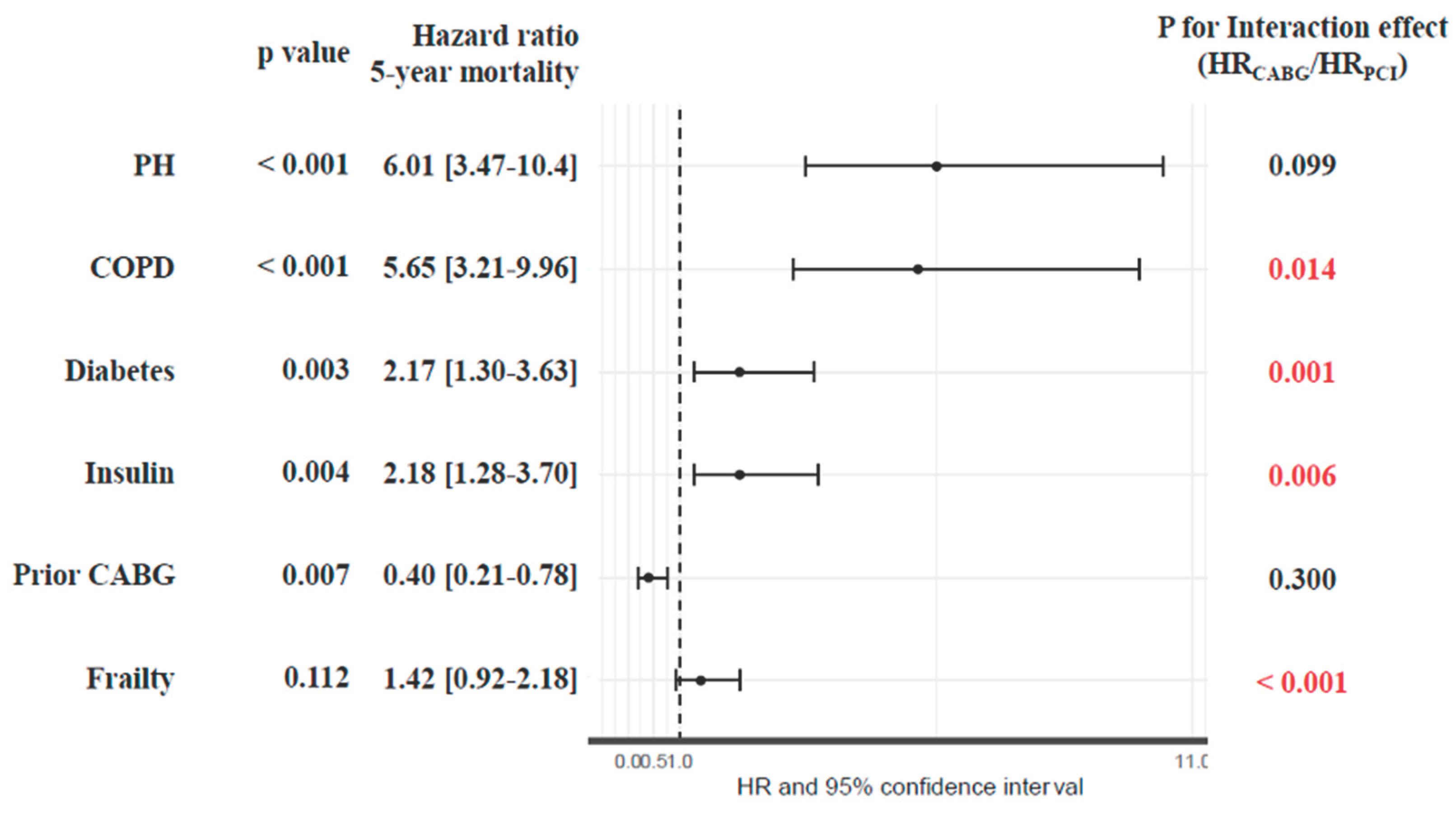
Results
Discussion
Limitations
Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- WHO. WHO - causes of death. Available online: https://www.who.int/data/gho/data/themes/mortality-and-global-health-estimates/ghe-leading-causes-of-death (accessed on 1 December 2024).
- Libby, P.; Theroux, P. Pathophysiology of coronary artery disease. Circulation 2005, 111, 3481–3488. [Google Scholar] [CrossRef]
- Knuuti, J.; Wijns, W.; Saraste, A.; et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. Eur. Heart J. 2020, 41, 407–477. [Google Scholar] [CrossRef] [PubMed]
- Barringhaus, K.G.; Park, K.L.; McManus, D.D.; et al. Outcomes from patients with multi-vessel disease following primary PCI: Staged PCI imparts very low mortality. Catheter Cardiovasc Interv. 2011, 77, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Ahn, J.M.; Lee, C.W.; et al. Long-Term Mortality After Coronary Revascularization in Nondiabetic Patients With Multivessel Disease. J. Am. Coll. Cardiol. 2016, 68, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Neumann, F.J.; Sousa-Uva, M.; Ahlsson, A.; et al. 2018 ESC/EACTS Guidelines on myocardial revascularization. Eur. Heart J. 2019, 40, 87–165. [Google Scholar] [CrossRef]
- Dehmer, G.J.; Grines, C.L.; Bakaeen, F.G.; et al. 2023 AHA/ACC Clinical Performance and Quality Measures for Coronary Artery Revascularization: A Report of the American College of Cardiology/American Heart Association Joint Committee on Performance Measures. Circ. Cardiovasc Qual. Outcomes 2023, 16, e00121. [Google Scholar] [CrossRef]
- Patterson, T.; McConkey, H.Z.R.; Ahmed-Jushuf, F.; et al. Long-Term Outcomes Following Heart Team Revascularization Recommendations in Complex Coronary Artery Disease. J. Am. Heart Assoc. 2019, 8, e011279. [Google Scholar] [CrossRef]
- Jonik, S.; Kageyama, S.; Ninomiya, K.; et al. Five-year outcomes in patients with multivessel coronary artery disease undergoing surgery or percutaneous intervention. Sci. Rep. 2024, 14, 3218. [Google Scholar] [CrossRef]
- Legrand, V.M.; Serruys, P.W.; Unger, F.; et al. Three-year outcome after coronary stenting versus bypass surgery for the treatment of multivessel disease. Circulation 2004, 109, 1114–1120. [Google Scholar] [CrossRef]
- Schwalbe, N.; Wahl, B. Artificial intelligence and the future of global health. Lancet 2020, 395, 1579–1586. [Google Scholar] [CrossRef]
- Harikrishnan, S.; Sanjay, G.; Ashishkumar, M.; et al. Pulmonary hypertension registry of Kerala, India (PRO-KERALA) - Clinical characteristics and practice patterns. Int. J. Cardiol. 2018, 265, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Shimony, A.; Eisenberg, M.J.; Rudski, L.G.; et al. Prevalence and impact of coronary artery disease in patients with pulmonary arterial hypertension. Am. J. Cardiol. 2011, 108, 460–464. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.M.; Xu, Z.B.; Yi, Q.; Ou, X.M.; Feng, Y.L. Association of chronic obstructive pulmonary disease with coronary artery disease. Chin. Med. J. (Engl) 2013, 126, 3205–3208. [Google Scholar] [CrossRef] [PubMed]
- Jonik, S.; Marchel, M.; Pedzich-Placha, E.; et al. Optimal Management of Patients with Severe Coronary Artery Disease following Multidisciplinary Heart Team Approach-Insights from Tertiary Cardiovascular Care Center. Int. J. Env. Res. Public Health 2022, 19. [Google Scholar] [CrossRef]
- Dutta, P.; Gomez, D.; Gladwin, M.T. Do BRD(4)S of a Feather Flock Together? How an Inflammation-Driven Epigenetic Regulator May Link Pulmonary Hypertension and Coronary Artery Disease. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1428–1430. [Google Scholar] [CrossRef]
- Roversi, S.; Roversi, P.; Spadafora, G.; Rossi, R.; Fabbri, L.M. Coronary artery disease concomitant with chronic obstructive pulmonary disease. Eur. J. Clin. Invest 2014, 44, 93–102. [Google Scholar] [CrossRef]
- Dursunoglu, N.; Dursunoglu, D.; Yildiz, A.I.; Uludag, B.; Alacam, Z.N.; Saricopur, A. Severity of coronary atherosclerosis in patients with COPD. Clin. Respir. J. 2017, 11, 751–756. [Google Scholar] [CrossRef]
- Almagro, P.; Lapuente, A.; Pareja, J.; et al. Underdiagnosis and prognosis of chronic obstructive pulmonary disease after percutaneous coronary intervention: A prospective study. Int. J. Chron. Obstruct Pulmon Dis. 2015, 10, 1353–1361. [Google Scholar] [CrossRef]
- Huang, L.; Pang, L.; Gu, Q.; et al. Prevalence, risk factors, and survival associated with pulmonary hypertension and heart failure among patients with underlying coronary artery disease: A national prospective, multicenter registry study in China. Chin. Med. J. (Engl) 2022, 135, 1837–1845. [Google Scholar] [CrossRef]
- Li, Y.; Zheng, H.; Yan, W.; et al. The impact of chronic obstructive pulmonary disease on the prognosis outcomes of patients with percutaneous coronary intervention or coronary artery bypass grafting: A meta-analysis. Heart Lung 2023, 60, 8–14. [Google Scholar] [CrossRef]
- Casselbrant, A.; Fedorowski, A.; Frantz, S.; Engstrom, G.; Wollmer, P.; Hamrefors, V. Common physiologic and proteomic biomarkers in pulmonary and coronary artery disease. PLoS ONE 2022, 17(3), e0264376. [Google Scholar] [CrossRef] [PubMed]
- Velioglu, Y.; Yuksel, A.; Topal, D.; et al. Does pulmonary hypertension affect early-term outcomes of off-pump coronary artery bypass surgery? Rev. Assoc. Med. Bras. (1992) 2022, 68, 1747–1752. [Google Scholar] [CrossRef]
| Characteristics | Overall (1035) | COPD/PH (125) | Non-COPD/PH (910) | p Value | |
|---|---|---|---|---|---|
| Preprocedural | Age, years; median (Q1-Q3) | 68 (64-69) | 67 (64-68) | 68 (64-69) | 0.07 |
| Gender, male; n(%) | 784 (75.7) | 96 (76.8) | 688 (75.6) | 0.77 | |
| BMI, kg/m2; median (Q1-O3) | 28 (25-29) | 27 (26-28) | 28 (25-29) | 0.06 | |
| Frailty; n(%) | 98 (9.5) | 17 (13.6) | 81 (8.9) | 0.09 | |
| Current smoking; n(%) | 197 (19.0) | 98 (78.4) | 99 (10.9) | <0.01 | |
| Diabetes; n(%) | 317 (30.6) | 40 (32.0) | 277 (30.4) | 0.72 | |
| with insulin; n(%) | 105 (10.1) | 13 (10.4) | 92 (10.1) | 0.93 | |
| Hypertension; n(%) | 868 (83.9) | 106 (84.8) | 762 (83.7) | 0.76 | |
| Dyslipidemia; n(%) | 840 (81.2) | 99 (79.2) | 741 (81.4) | 0.56 | |
| Heart failure; n(%) | 734 (70.9) | 91 (72.8) | 643 (70.7) | 0.63 | |
| LVEF, %; median (Q1-Q3) | 38 (32-39) | 37 (32-38) | 38 (32-39) | 0.12 | |
| CKD; n(%) | 270 (26.1) | 33 (26.4) | 237 (26.0) | 0.93 | |
| Anemia; n(%) | 332 (32.1) | 41 (32.8) | 291 (32.0) | 0.85 | |
| Prior MI; n(%) | 507 (49.0) | 62 (49.6) | 445 (48.9) | 0.88 | |
| Prior revascularization; n(%) | 399 (38.6) | 51 (40.8) | 348 (38.2) | 0.58 | |
| Indication; n(%) | |||||
| Stable CAD | 598 (57.8) | 74 (59.2) | 524 (57.6) | 0.74 | |
| ACS | 437 (42.2) | 51 (40.8) | 386 (42.4) | 0.75 | |
| STEMI | 121 (11.7) | 15 (12.0) | 106 (11.6) | 0.92 | |
| NSTEMI/UA | 316 (30.5) | 36 (28.8) | 280 (30.8) | 0.65 | |
| PAD; n(%) | 63 (6.1) | 11 (8.8) | 52 (5.7) | 0.19 | |
| Prior stroke/TIA; n(%) | 86 (8.3) | 14 (11.2) | 72 (7.9) | 0.21 | |
| Active cancer; n(%) | 27 (2.6) | 6 (4.8) | 21 (2.3) | 0.10 | |
| EuroSCORE II, %; median (Q1-Q3) | 5 (4-9) | 8 (4-10) | 5 (3-6) | <0.01 | |
| STS score, %; median (Q1-Q3) | 3 (2-4) | 5 (3-6) | 3 (2-4) | <0.01 | |
| Procedural | Complete revascularization; n(%) | 630 (60.9) | 75 (60.0) | 555 (61.0) | 0.84 |
| Angiographic | Number of lesion; median (Q1-Q3) | 4 (1-6) | 4 (1-7) | 4 (1-6) | 0.89 |
| LM disease; n(%) | 267 (25.8) | 32 (25.6) | 235 (25.8) | 0.96 | |
| Bifurcation; n(%) | 752 (72.7) | 93 (74.4) | 659 (72.4) | 0.64 | |
| Severe calcification; n(%) | 292 (28.2) | 42 (33.6) | 250 (27.5) | 0.15 | |
| Total occlusion; n(%) | 171 (16.5) | 25 (20.0) | 146 (16.0) | 0.26 | |
| SYNTAX score; median (Q1-Q3) | 30 (26-31) | 31 (27-31) | 30 (26-31) | 0.75 | |
| Characteristics | Overall (125) | CABG (27) | PCI (98) | p Value | |
|---|---|---|---|---|---|
| Preprocedural | Age, years; median (Q1-Q3) | 67 (63-70) | 66 (64-67) | 68 (63-70) | 0.06 |
| Gender, male; n(%) | 96 (76.8) | 22 (81.5) | 74 (75.5) | 0.52 | |
| BMI, kg/m2; median (Q1-Q3) | 27 (25-28) | 26 (25-27) | 27 (25-28) | 0.09 | |
| Frailty; n(%) | 17 (13.6) | 2 (7.4) | 15 (15.3) | 0.29 | |
| Current smoking; n(%) | 98 (78.4) | 21 (77.8) | 77 (78.6) | 0.93 | |
| Diabetes; n(%) | 40 (32.0) | 8 (29.6) | 32 (32.7) | 0.78 | |
| with insulin; n(%) | 13 (10.4) | 3 (11.1) | 10 (10.2) | 0.90 | |
| Hypertension; n(%) | 106 (84.8) | 23 (85.2) |
83 (84.7) | 0.95 | |
| Dyslipidemia; n(%) | 99 (79.2) | 21 (77.8) | 78 (79.6) | 0.85 | |
| Heart failure; n(%) | 91 (72.8) | 18 (66.7) | 73 (74.5) | 0.42 | |
| LVEF, %; median (Q1-Q3) | 37 (31-41) | 40 (31-42) | 36 (31-40) | <0.01 | |
| CKD; n(%) | 33 (26.4) | 4 (14.8) | 29 (29.6) | 0.12 | |
| Anemia; n(%) | 41 (32.8) | 5 (18.5) | 36 (36.7) | 0.08 | |
| Prior MI; n(%) | 62 (49.6) | 12 (44.4) | 50 (51.0) | 0.55 | |
| Prior revascularization; n(%) | 51 (40.8) | 9 (33.3) | 42 (42.9) | 0.38 | |
| Indication; n(%) | |||||
| CCS | 74 (59.2) | 17 (63.0) | 57 (58.2) | 0.66 | |
| ACS | 51 (40.8) | 10 (37.0) | 41 (41.8) | 0.67 | |
| STEMI | 15 (12.0) | 0 (0.0) | 15 (15.3) | 0.03 | |
| NSTEMI/UA | 36 (28.8) | 10 (37.0) | 26 (26.5) | 0.29 | |
| PAD; n(%) | 11 (8.8) | 2 (7.4) | 9 (9.2) | 0.78 | |
| Prior stroke/TIA; n(%) | 14 (11.2) | 2 (7.4) | 12 (12.2) | 0.48 | |
| Active cancer; n(%) | 6 (4.8) | 0 (0.0) | 6 (6.1) | 0.19 | |
| EuroSCORE II, %; mean(SD) | 8 (4-10) | 6 (5-8) | 9 (4-11) | <0.01 | |
| STS score, %; median(Q1-Q3) | 5 (3-8) | 4 (1-6) | 6 (3-8) | <0.01 | |
| Procedural | Complete revascularization; n(%) | 75 (60.0) | 18 (66.7) | 57 (58.2) | 0.43 |
| Angiographic | Number of lesion; median (Q1-Q3) | 4 (1-5) | 4 (1-6) | 4 (1-5) | 0.13 |
| LM disease; n(%) | 32 (25.6) | 10 (37.0) | 22 (22.4) | 0.14 | |
| Bifurcation; n(%) | 93 (74.4) | 21 (77.8) | 72 (73.5) | 0.65 | |
| Severe calcification; n(%) | 42 (33.6) | 8 (29.6) | 34 (34.7) | 0.64 | |
| Total occlusion; n(%) | 25 (20.0) | 5 (18.5) | 20 (20.4) | 0.83 | |
| SYNTAX score; median (Q1-Q3) | 31 (25-34) | 32 (25-35) | 31 (25-33) | 0.08 | |
| Endpoints | Overall (1035) | COPD/PH (125) | Non-COPD/PH (910) | p Value |
|---|---|---|---|---|
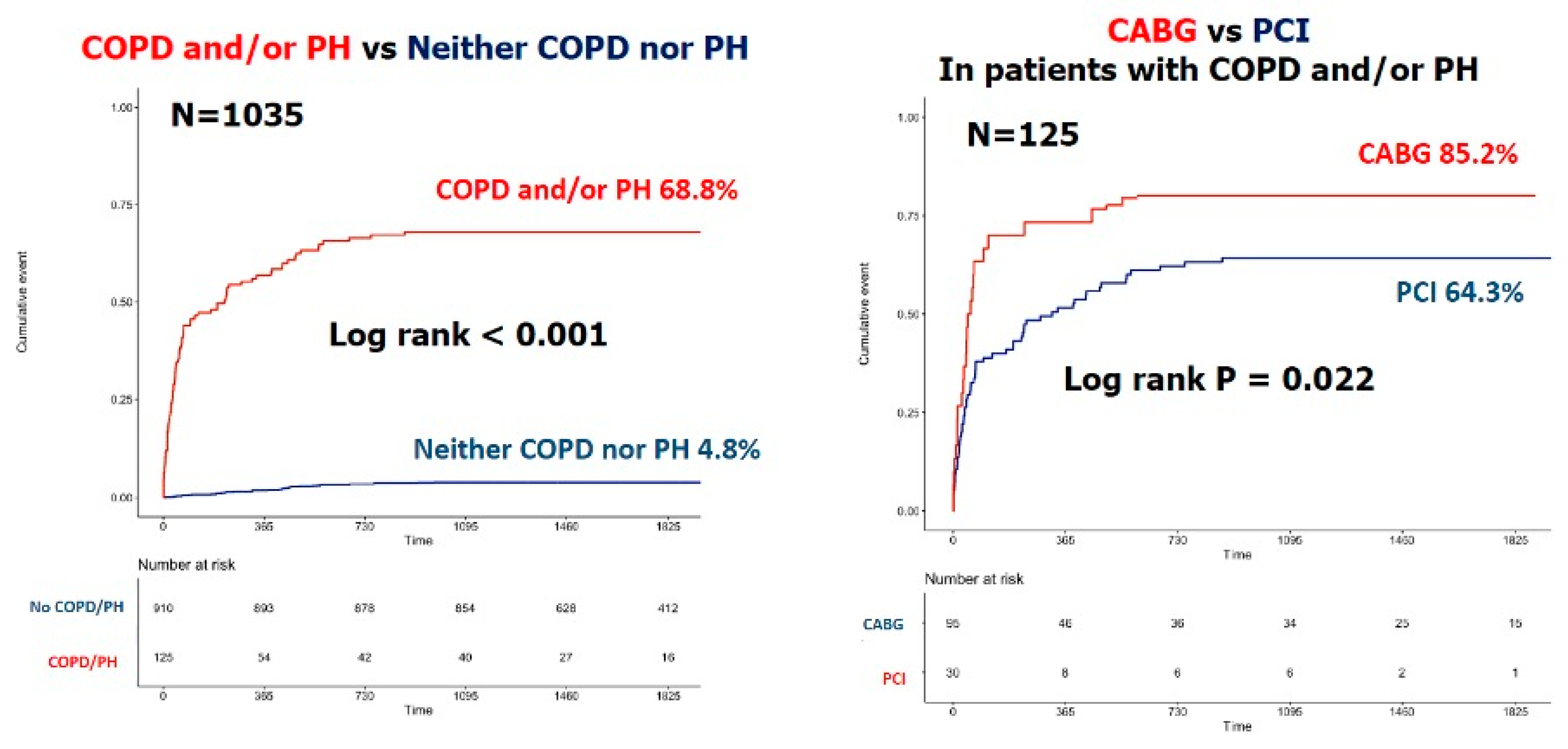
| All-cause death, n(%) | 130 (12.6) | 86 (68.8) | 44 (4.8) | <0.01 |
| MI, n(%) | 121 (11.7) | 16 (12.8) | 105 (11.5) | 0.70 |
| Stroke, n(%) | 47 (4.5) | 7 (5.6) | 40 (4.4) | 0.55 |
| Repeat revascularization, n(%) | 261 (25.2) | 29 (23.2) | 232 (25.5) | 0.59 |
| MACCE, n(%) | 525 (50.7) | 121 (96.8) | 404 (44.4) | <0.01 |
| In-hospital mortality, n(%) | 42 (4.1) | 10 (8.0) | 32 (3.5) | 0.02 |
| Postprocedural hospital stay, days; median (Q1-Q3) | 7 (2-11) | 9 (2-13) | 6 (1-9) | <0.01 |
| Endpoints | Overall (125) | CABG (27) | PCI (98) | p Value |
|---|---|---|---|---|
| All-cause death, n(%) | 86 (68.8) | 23 (85.2) | 63 (64.3) | 0.04 |
| MACCE, n(%) | 121 (96.8) | 26 (96.3) | 95 (96.9) | 0.16 |
| MI, n(%) | 16 (12.8) | 2 (7.4) | 14 (14.3) | 0.42 |
| Stroke, n(%) | 7 (5.6) | 4 (14.8) | 3 (3.1) | 0.02 |
| Repeat revascularization, n(%) | 29 (23.2) | 4 (14.8) | 25 (25.5) | 0.26 |
| In-hospital mortality, n(%) | 10 (8.0) | 6 (22.2) | 4 (4.1) | <0.01 |
| Postprocedural hospital stay, days; median (Q1-Q3) | 9 (3-14) | 14 (6-24) | 7 (1-10) | <0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




