Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
Collection and Acclimation of Coral
Experimental Design
Coral-ACTIS
Commercial HSI
Coral Pigment Analysis
Statistical Analysis
Results
Comparing Coral Reflectance Between Instruments
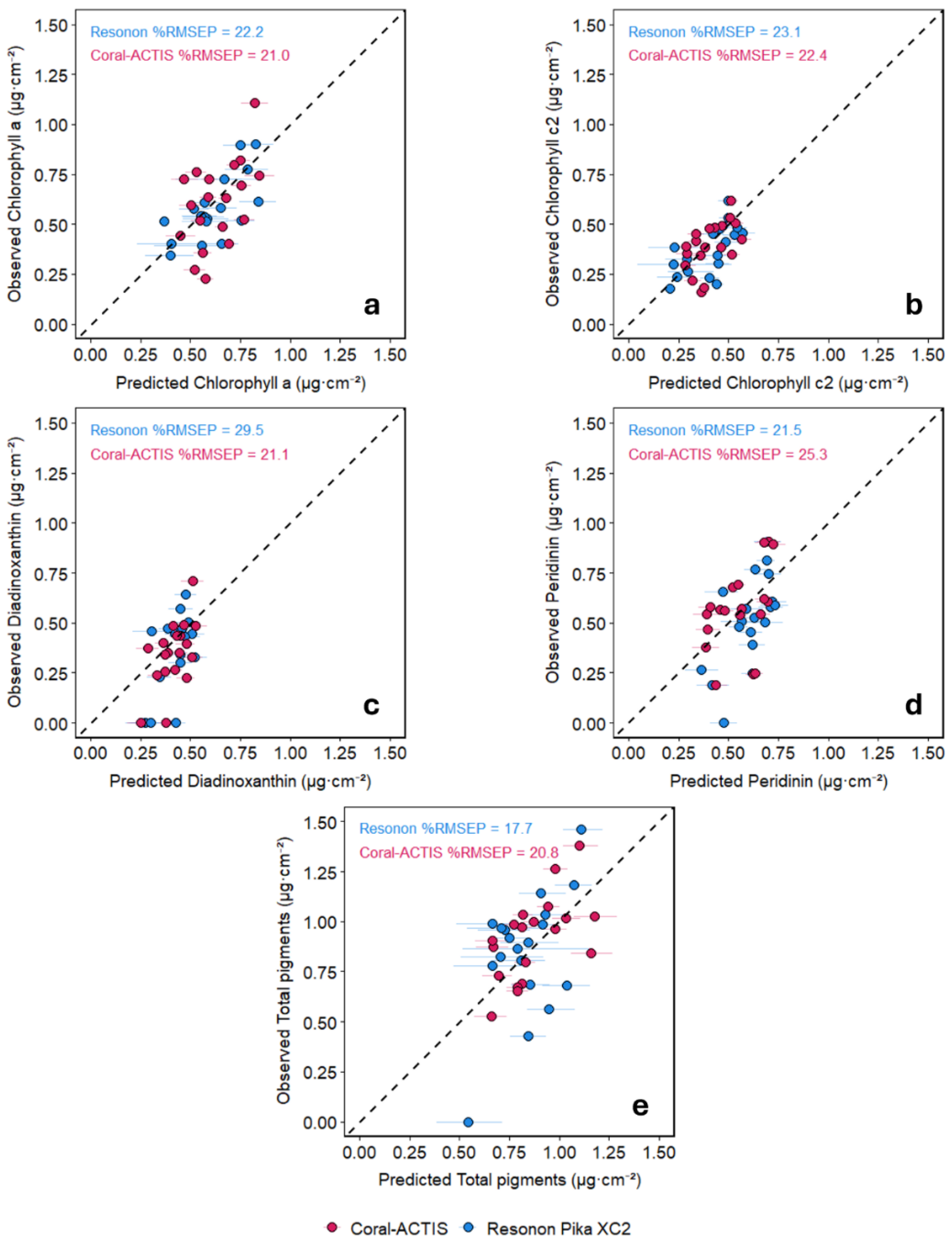
Predicting Coral Pigment Concentration
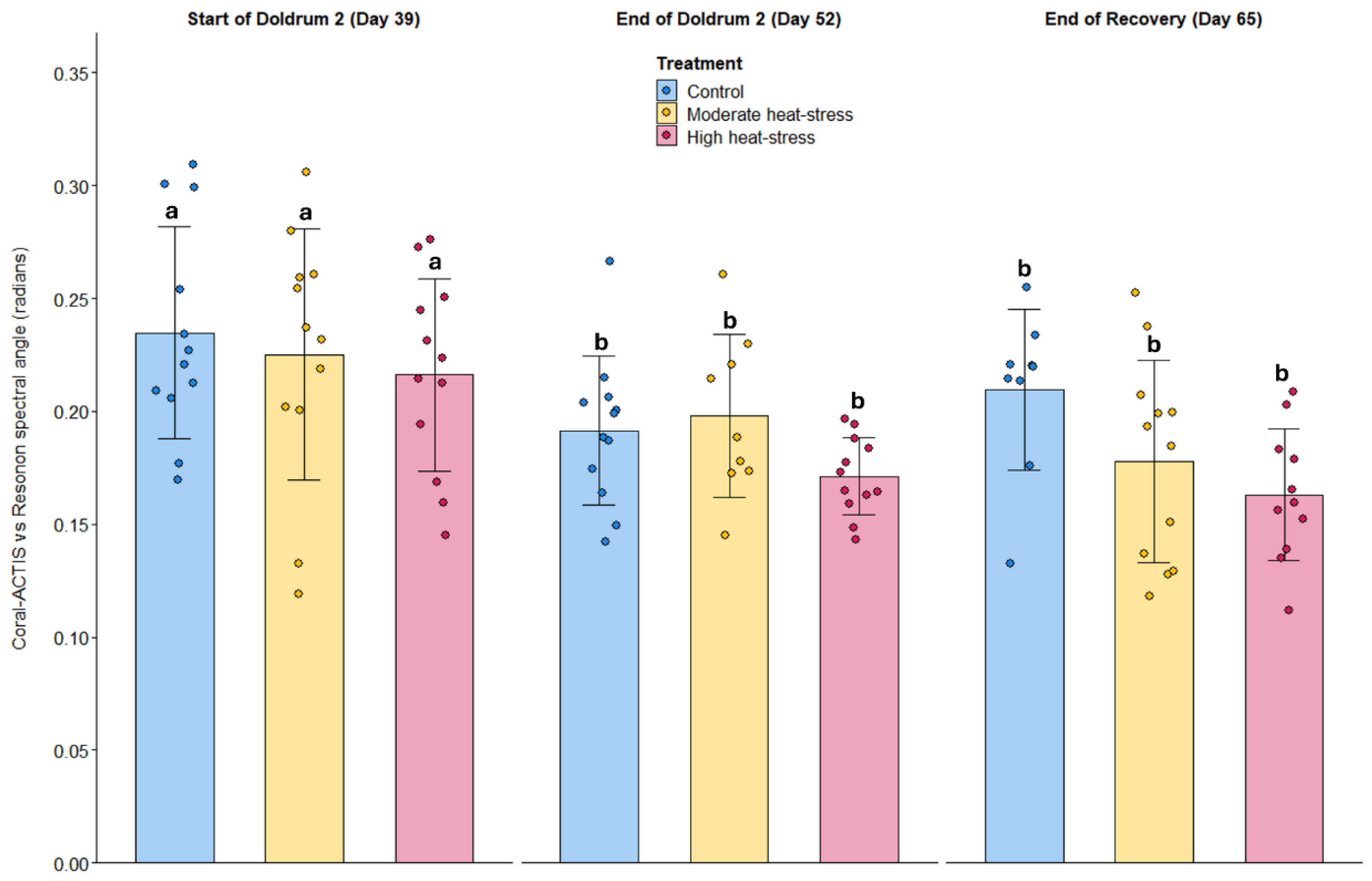
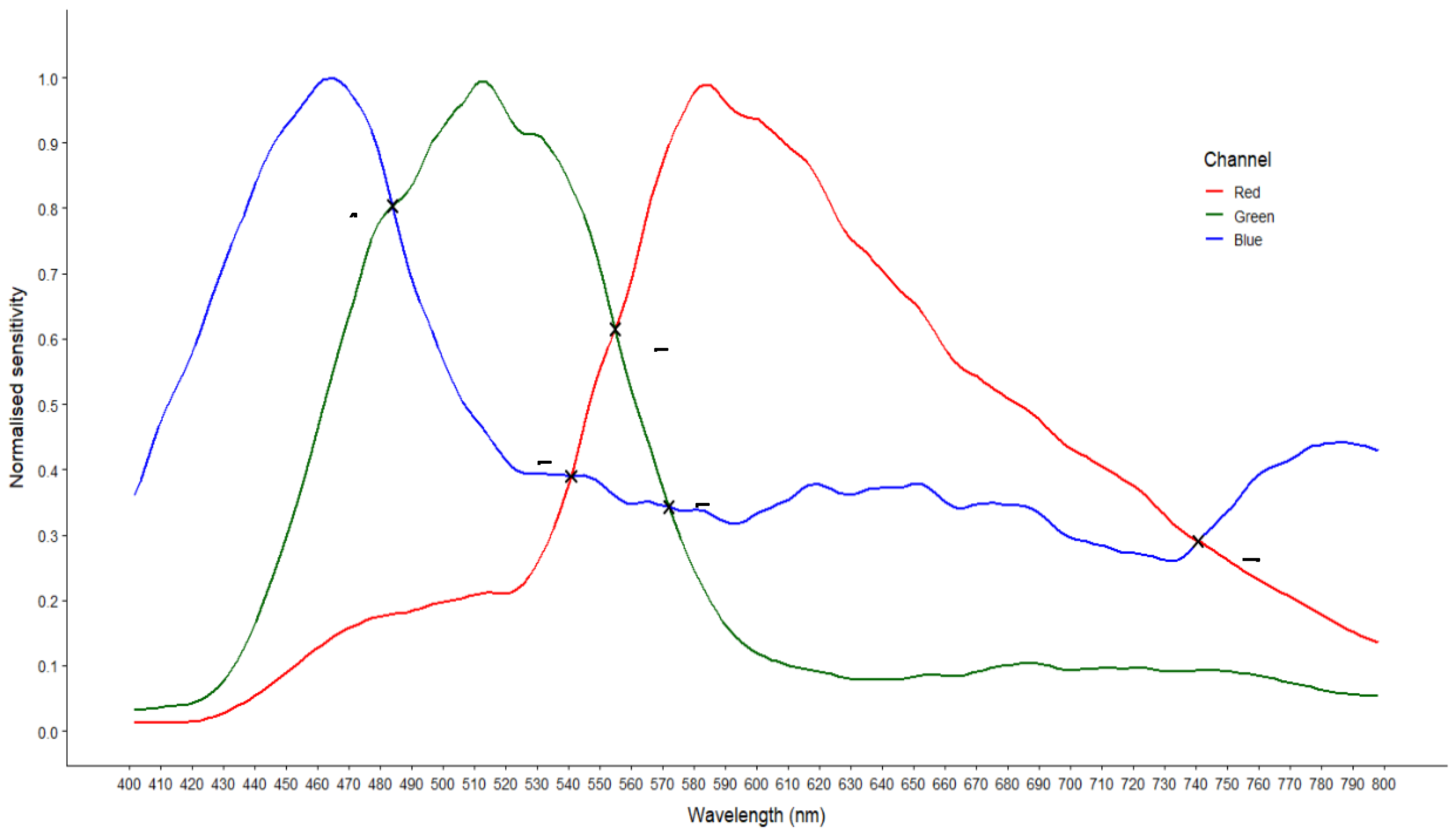
Camera Sensitivities and Comparison of Reference Spectra
Discussion
Coral Reflectance Comparison
PLSR and Prediction of Pigments
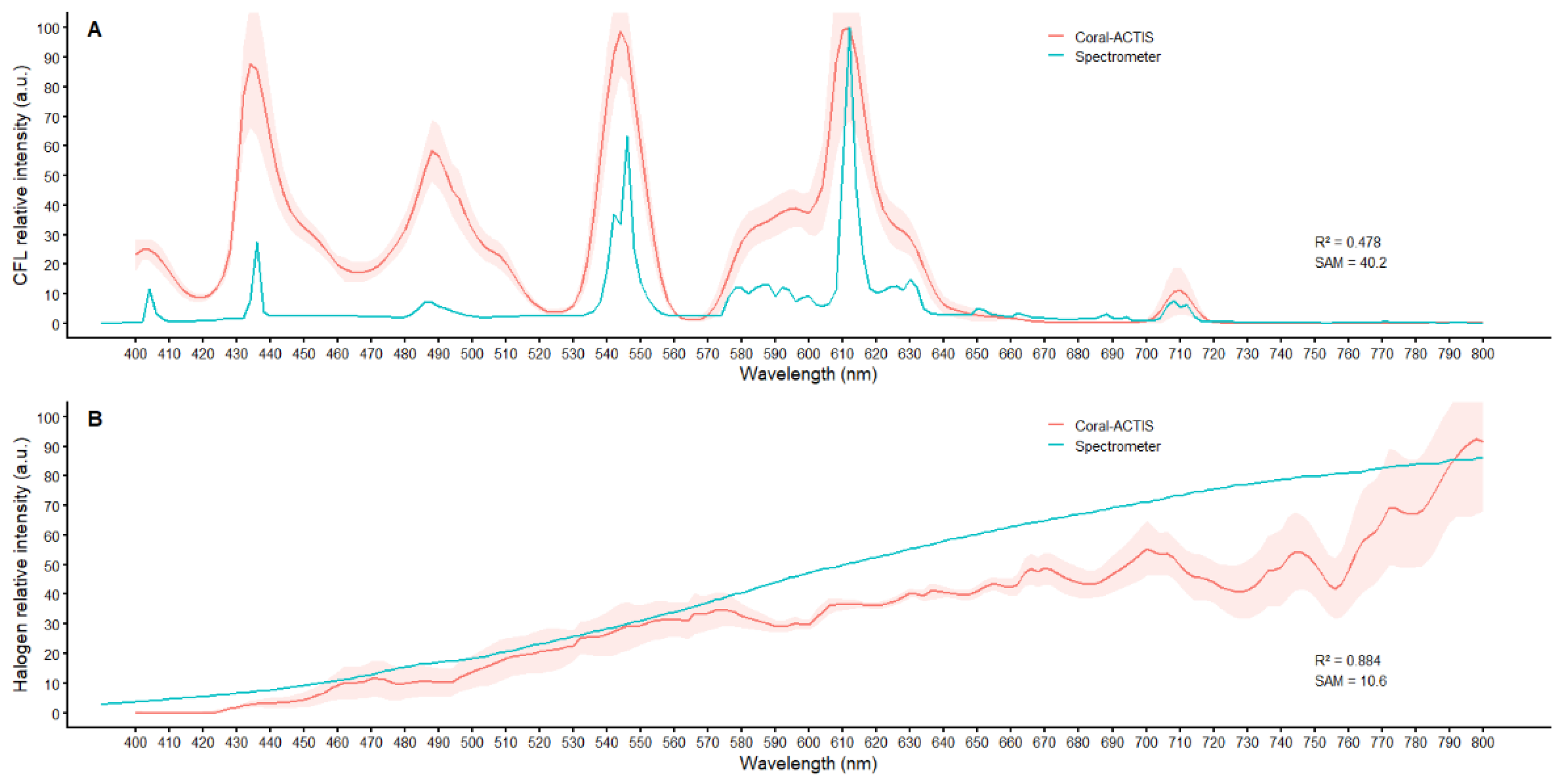
Coral-ACTIS vs Reference Light Spectra
Future Coral Spectral Imaging
Conclusion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Declaration of Competing Interest
Data availability
References
- Abd-Elrahman, A.; Pande-Chhetri, R.; Vallad, G. Design and development of a multi-purpose low-cost hyperspectral imaging system. Remote Sensing 2011, 3(3), 570–586. [Google Scholar] [CrossRef]
- Ahmad, A.; Kumar, A.; Dubey, V.; Butola, A.; Ahluwalia, B. S.; Mehta, D. S. Characterization of color cross-talk of CCD detectors and its influence in multispectral quantitative phase imaging. Optics Express 2019, 27(4), 4572–4589. [Google Scholar] [CrossRef]
- Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral Ecology 2001, 26(1), 32–46. [Google Scholar] [CrossRef]
- Asner, G. P.; Drury, C.; Vaughn, N. R.; Hancock, J. R.; Martin, R. E. Variability in Symbiont Chlorophyll of Hawaiian Corals from Field and Airborne Spectroscopy. Remote Sensing 2024, 16(5), 732. [Google Scholar] [CrossRef]
- Bhargava, A.; Sachdeva, A.; Sharma, K.; Alsharif, M. H.; Uthansakul, P.; Uthansakul, M. Hyperspectral imaging and its applications: A review. Heliyon 2024, 10(12). [Google Scholar] [CrossRef]
- Burnett, A. C.; Anderson, J.; Davidson, K. J.; Ely, K. S.; Lamour, J.; Li, Q.; Morrison, B. D.; Yang, D.; Rogers, A.; Serbin, S. P. A best-practice guide to predicting plant traits from leaf-level hyperspectral data using partial least squares regression. Journal of Experimental Botany 2021, 72(18), 6175–6189. [Google Scholar] [CrossRef] [PubMed]
- Butcherine, P.; Fiori, A.; Ellis, S. L.; Hendrickson, C. A.; Harrison, D. P. Shading Acropora kenti during doldrum-like conditions can reduce mortality risk during high-thermal stress events. Review 2026. [Google Scholar] [CrossRef]
- Carrascal, L. M.; Galván, I.; Gordo, O. Partial least squares regression as an alternative to current regression methods used in ecology. Oikos 2009, 118(5), 681–690. [Google Scholar] [CrossRef]
- Cheng, M.-F.; Mukundan, A.; Karmakar, R.; Valappil, M. A. E.; Jouhar, J.; Wang, H.-C. Modern Trends and Recent Applications of Hyperspectral Imaging: A Review. Technologies 2025, 13(5), 170. [Google Scholar] [CrossRef]
- Chennu, A.; Färber, P.; De’ath, G.; de Beer, D.; Fabricius, K. E. A diver-operated hyperspectral imaging and topographic surveying system for automated mapping of benthic habitats. Scientific Reports 2017, 7(1), 7122. [Google Scholar] [CrossRef]
- Eöllös-Jarošíková, K.; Guerra-Yánez, C.; Zvánovec, S.; Komanec, M. Fog resilient optical camera communication using wavelength division multiplexing. Optics Express 2025, 33(19), 39426–39437. [Google Scholar] [CrossRef]
- Ferrara, E. F.; Bauer, L.; Puntin, G.; Bautz, F.; Celayir, S.; Do, M.-S.; Eck, F.; Heider, M.; Wissel, P.; Arnold, A.; Wilke, T.; Reichert, J.; Ziegler, M. RGB color indices as proxy for symbiont cell density and chlorophyll content during coral bleaching (p. 2024.12.20.629333). bioRxiv 2024. [Google Scholar] [CrossRef]
- Hochberg, E. J.; Atkinson, M. J.; Apprill, A.; Andréfouët, S. Spectral reflectance of coral. Coral Reefs 2004, 23(1), 84–95. [Google Scholar] [CrossRef]
- Hochberg, E. J.; Ducret, H.; Dierssen, H. M. Revised empirical models for estimating coral pigment concentrations from optical reflectance spectra. Applied Optics 2025, 64(35), 10603–10610. [Google Scholar] [CrossRef] [PubMed]
- Holden, H.; LeDrew, E. Spectral Discrimination of Healthy and Non-Healthy Corals Based on Cluster Analysis, Principal Components Analysis, and Derivative Spectroscopy. Remote Sensing of Environment 1998, 65(2), 217–224. [Google Scholar] [CrossRef]
- Joyce, K. E.; Phinn, S. R. Hyperspectral analysis of chlorophyll content and photosynthetic capacity of coral reef substrates. Limnology and Oceanography 2003, 48(1part2), 489–496. [Google Scholar] [CrossRef]
- Khaled, M. A.; Abdelsalam, A. A. Discrimination of some red sea coral reef species based on hyperspectral signature field data. Scientific African 2025, 28, e02696. [Google Scholar] [CrossRef]
- Kruse, F. A.; Lefkoff, A. B.; Boardman, J. W.; Heidebrecht, K. B.; Shapiro, A. T.; Barloon, P. J.; Goetz, A. F. H. The spectral image processing system (SIPS)—Interactive visualization and analysis of imaging spectrometer data. Remote Sensing of Environment, Airbone Imaging Spectrometry 1993, 44(2), 145–163. [Google Scholar] [CrossRef]
- Lukac, R.; Plataniotis, K. N. Color filter arrays: Design and performance analysis. IEEE Transactions on Consumer Electronics 2005, 51(4), 1260–1267. [Google Scholar] [CrossRef]
- Luo, J.; Wang, J. Image Demosaicing Based on Generative Adversarial Network. Mathematical Problems in Engineering 2020, 2020(1), 7367608. [Google Scholar] [CrossRef]
- Melamed, M. B.; Martin, R. E.; Allen, M.; Asner, G. P. Reflections: Spectral Investigation of Black Band Disease in Hawaiian Corals. Remote Sensing 2025, 17(18), 3241. [Google Scholar] [CrossRef]
- Menon, D.; Calvagno, G. Color image demosaicking: An overview. Signal Processing: Image Communication 2011, 26(8), 518–533. [Google Scholar] [CrossRef]
- Mevik, B.-H.; Wehrens, R. The pls package: Principal component and partial least squares regression in R. Journal of Statistical Software 2007, 18, 1–23. [Google Scholar] [CrossRef]
- Mishra, D. R.; Narumalani, S.; Rundquist, D.; Lawson, M.; Perk, R. Enhancing the detection and classification of coral reef and associated benthic habitats: A hyperspectral remote sensing approach. Journal of Geophysical Research: Oceans 2007, 112(C8). [Google Scholar] [CrossRef]
- Monakhova, K.; Yanny, K.; Aggarwal, N.; Waller, L. Spectral DiffuserCam: Lensless snapshot hyperspectral imaging with a spectral filter array. Optica 2020, 7(10), 1298–1307. [Google Scholar] [CrossRef]
- Montes-Herrera, J. C.; Cimoli, E.; Cummings, V.; Hill, N.; Lucieer, A.; Lucieer, V. Underwater hyperspectral imaging (UHI): A review of systems and applications for proximal seafloor ecosystem studies. Remote Sensing 2021, 13(17), 3451. [Google Scholar] [CrossRef]
- Podlesnykh, I.; Kovalev, M.; Platonov, P. Towards the Future of Ubiquitous Hyperspectral Imaging: Innovations in Sensor Configurations and Cost Reduction for Widespread Applicability. Technologies 2024, 12(11), 221. [Google Scholar] [CrossRef]
- Polder, G.; Gowen, A. The hype in spectral imaging; Spectroscopy Europe, 2021; Available online: https://library.wur.nl/WebQuery/wurpubs/fulltext/574548.
- Rashid, A. R.; Chennu, A. A Trillion Coral Reef Colors: Deeply Annotated Underwater Hyperspectral Images for Automated Classification and Habitat Mapping. Data 2020, 5(1), Article 1. [Google Scholar] [CrossRef]
- Richards, L. S.; Siems, S. T.; Huang, Y.; Zhao, W.; Harrison, D. P.; Manton, M. J.; Reeder, M. J. The meteorological drivers of mass coral bleaching on the central Great Barrier Reef during the 2022 La Niña. Scientific Reports 2024, 14(1), 1–17. [Google Scholar] [CrossRef]
- Salazar-Vazquez, J.; Mendez-Vazquez, A. A plug-and-play Hyperspectral Imaging Sensor using low-cost equipment. HardwareX 2020, 7, e00087. [Google Scholar] [CrossRef]
- Savitzky, Abraham.; Golay, M. J. E. Smoothing and Differentiation of Data by Simplified Least Squares Procedures. Analytical Chemistry 1964, 36(8), 1627–1639. [Google Scholar] [CrossRef]
- Siebeck, U. E.; Marshall, N. J.; Klüter, A.; Hoegh-Guldberg, O. Monitoring coral bleaching using a colour reference card. Coral Reefs 2006, 25(3), 453–460. [Google Scholar] [CrossRef]
- Sony Semiconductor Solutions Corporation. IMX219PQ CMOS Image Sensor Datasheet. 2019. Available online: https://www.opensourceinstruments.com/Electronics/Data/IMX219PQ.pdf.
- Stuart, M. B.; McGonigle, A. J. S.; Davies, M.; Hobbs, M. J.; Boone, N. A.; Stanger, L. R.; Zhu, C.; Pering, T. D.; Willmott, J. R. Low-Cost Hyperspectral Imaging with A Smartphone. Journal of Imaging 2021, 7(8), Article 8. [Google Scholar] [CrossRef]
- Teague, J.; Megson-Smith, D. A.; Allen, M. J.; Day, J. C.; Scott, T. B. A Review of Optical Techniques for Coral Monitoring & Introducing Low-Cost Hyperspectral Imaging, 2021.
- Teague, J.; Willans, J.; Megson-Smith, D. A.; Day, J. C. C.; Allen, M. J.; Scott, T. B. Using Colour as a Marker for Coral ‘Health’: A Study on Hyperspectral Reflectance and Fluorescence Imaging of Thermally Induced Coral Bleaching. Oceans 2022, 3(4), Article 4. [Google Scholar] [CrossRef]
- Thrane, J.-E.; Kyle, M.; Striebel, M.; Haande, S.; Grung, M.; Rohrlack, T.; Andersen, T. Spectrophotometric Analysis of Pigments: A Critical Assessment of a High-Throughput Method for Analysis of Algal Pigment Mixtures by Spectral Deconvolution. PLOS ONE 2015, 10(9), e0137645. [Google Scholar] [CrossRef]
- Tucsen. Quantum Efficiency in Scientific Cameras: A Beginner’s Guide; Tucsen, 30 September 2025; Available online: https://www.tucsen.com/learning/quantum-efficiency-in-scientific-cameras/.
- Veal, C. J.; Carmi, M.; Fine, M.; Hoegh-Guldberg, O. Increasing the accuracy of surface area estimation using single wax dipping of coral fragments. Coral Reefs 2010, 29, 893–897. [Google Scholar] [CrossRef]
- Wang, L.; Jin, J.; Song, Z.; Wang, J.; Zhang, L.; Rehman, T. U.; Ma, D.; Carpenter, N. R.; Tuinstra, M. R. LeafSpec: An accurate and portable hyperspectral corn leaf imager. Computers and Electronics in Agriculture 2020, 169, 105209. [Google Scholar] [CrossRef]
- Wang, Y.; Pawlowski, M. E.; Cheng, S.; Dwight, J. G.; Stoian, R. I.; Lu, J.; Alexander, D.; Tkaczyk, T. S. Light-guide snapshot imaging spectrometer for remote sensing applications. Optics Express 2019, 27(11), 15701–15725. [Google Scholar] [CrossRef] [PubMed]
- Warner, M.; Chilcoat, G.; McFarland, F.; Fitt, W. Seasonal fluctuations in the photosynthetic capacity of photosystem II in symbiotic dinoflagellates in the Caribbean reef-building coral Montastraea. Marine Biology 2002, 141, 31–38. [Google Scholar] [CrossRef]
- Watty, K.; Schoepf, V.; Johnson, K. W.; Littke, S.; van der Zande, R. M. Using reflectance to measure chlorophyll a in corals: Calibration and implications of skeletal optical properties; Coral Reefs, 2026. [Google Scholar] [CrossRef]
- Zeng, K.; Xu, Z.; Yang, Y.; Liu, Y.; Zhao, H.; Zhang, Y.; Xie, B.; Zhou, W.; Li, C.; Cao, W. In situ hyperspectral characteristics and the discriminative ability of remote sensing to coral species in the South China Sea. GIScience & Remote Sensing 2022, 59(1), 272–294. [Google Scholar] [CrossRef]
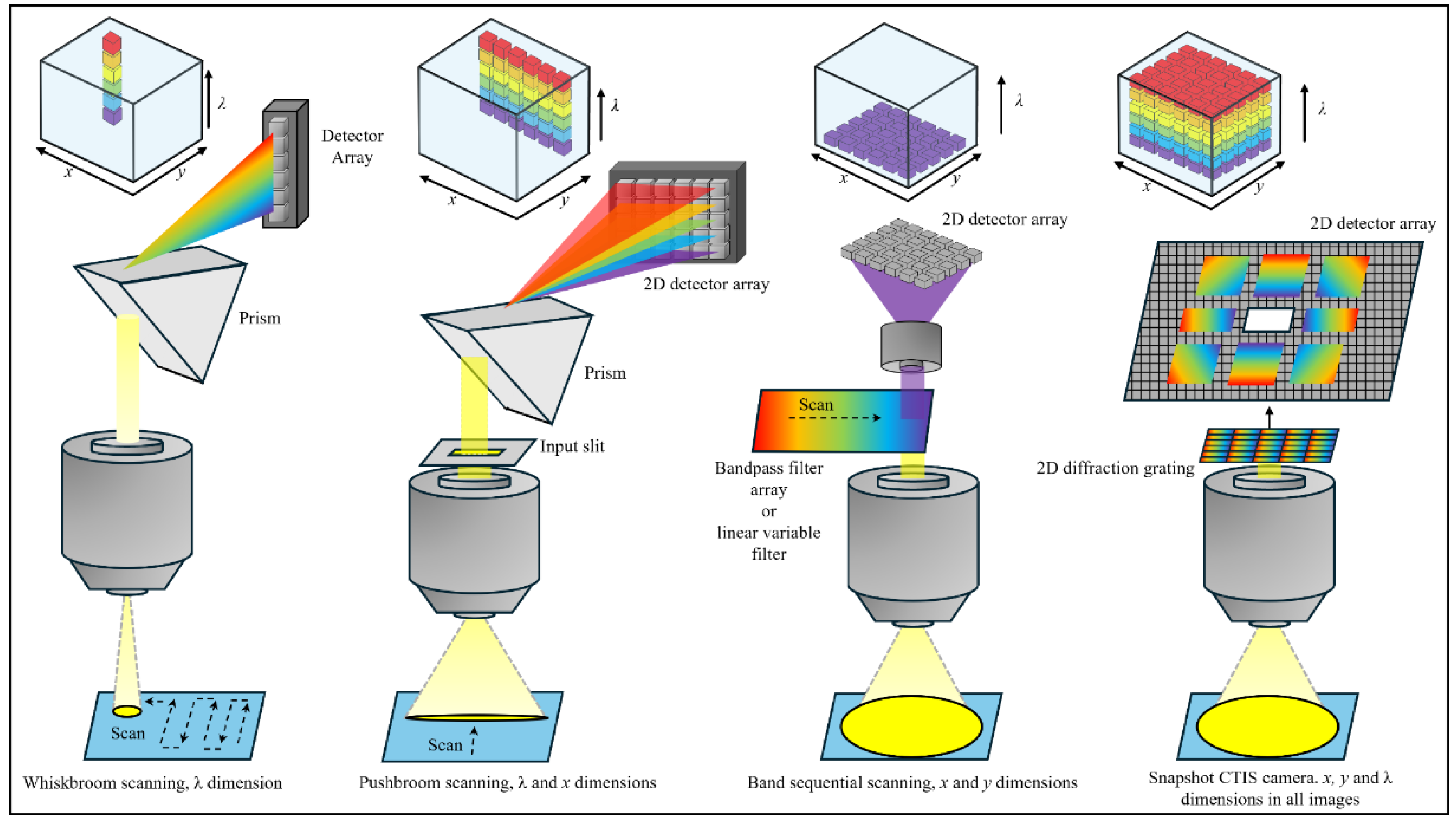
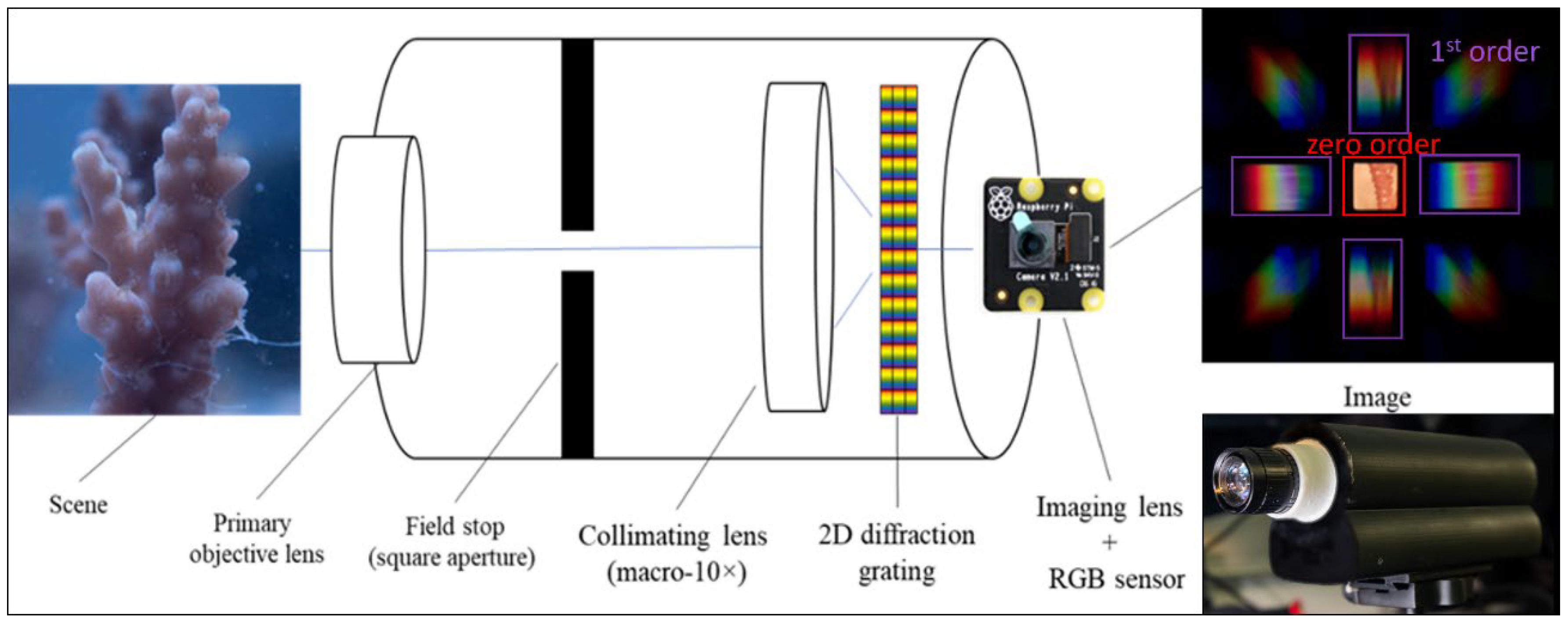
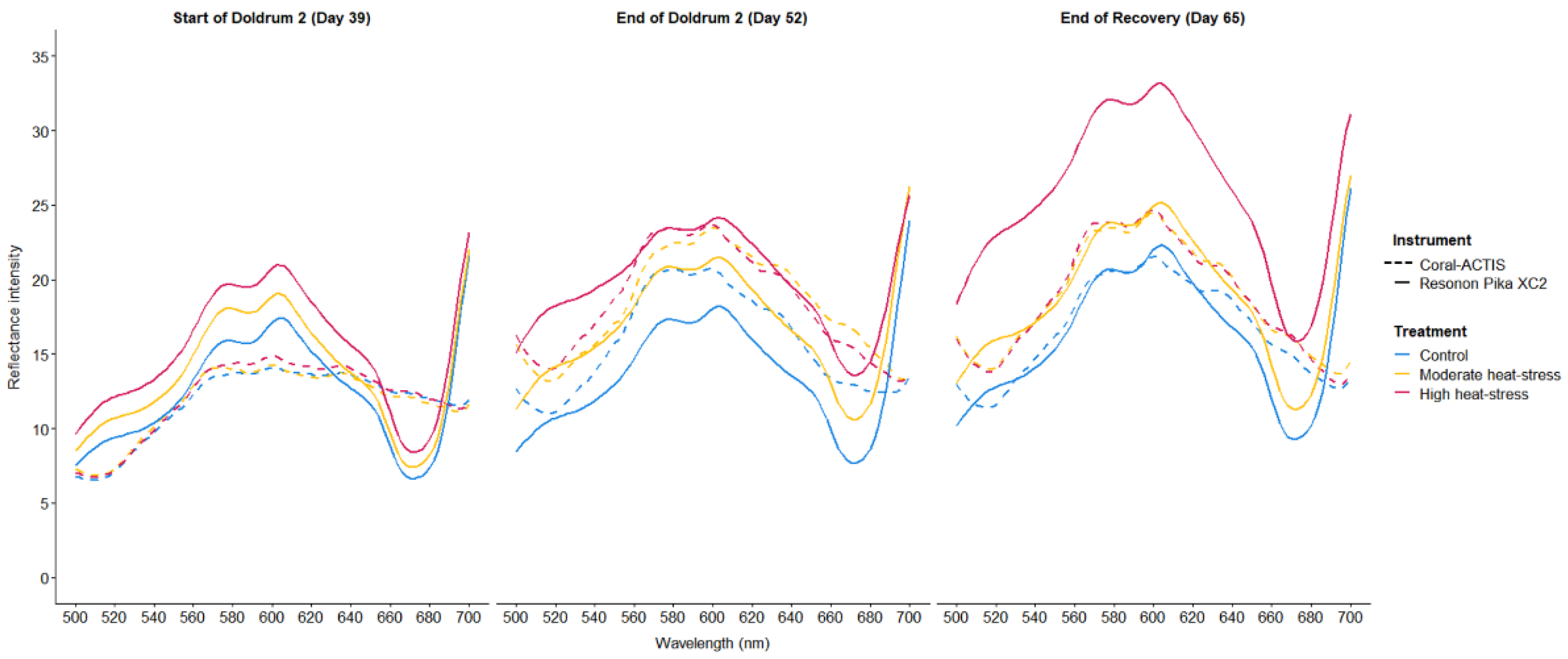
| Instrument. | Reflectance variable | Day | Temp | Day×Temp | Tank | Day×Tank | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| F | P | F | P | F | P | F | P | F | P | ||
| Resonon Pika XC2 | 542 nm | 11.0930 | 0.0004 | 6.6131 | 0.0139 | 1.2761 | 0.3276 | 2.0603 | 0.0459 | 1.1048 | 0.3602 |
| 560 nm | 15.2380 | 0.0003 | 7.5320 | 0.0133 | 1.5524 | 0.2269 | 1.9872 | 0.0518 | 0.9901 | 0.4765 | |
| 676 nm | 8.9971 | 0.0023 | 7.0610 | 0.0111 | 0.9586 | 0.4504 | 2.1819 | 0.0331 | 1.6281 | 0.0836 | |
| 694 nm | 27.4860 | 0.0001 | 8.1869 | 0.0135 | 1.5657 | 0.2302 | 2.7707 | 0.0091 | 1.4286 | 0.1543 | |
| 10 nm waveband (538 – 546 nm) | 11.1160 | 0.0008 | 6.6158 | 0.0138 | 1.2775 | 0.3164 | 2.0606 | 0.0450 | 1.1039 | 0.3604 | |
| 10 nm waveband (556 – 564 nm) | 15.2780 | 0.0003 | 7.5442 | 0.0130 | 1.5526 | 0.2358 | 1.9844 | 0.0511 | 0.9889 | 0.4734 | |
| 10 nm waveband (690 – 698 nm) | 27.3690 | 0.0001 | 8.0708 | 0.0108 | 1.5482 | 0.2281 | 2.7627 | 0.0098 | 1.4495 | 0.1477 | |
| 50 nm waveband (510 – 560 nm) | 10.7760 | 0.0012 | 6.4498 | 0.0176 | 2.0762 | 0.0568 | 6.4498 | 0.0176 | 6.4498 | 0.0176 | |
| 50 nm waveband (570 – 620 nm) | 21.7020 | 0.0001 | 21.7020 | 0.0001 | 1.7757 | 0.1869 | 1.9818 | 0.0533 | 0.9286 | 0.5348 | |
| 50 nm waveband (650 – 700 nm) | 13.9740 | 0.0004 | 7.5606 | 0.0108 | 1.0656 | 0.4098 | 7.5606 | 0.0108 | 1.0656 | 0.4098 | |
| Coral-ACTIS | 542 nm | 48.2960 | 0.0001 | 2.3631 | 0.1611 | 1.2583 | 0.3287 | 1.8690 | 0.0698 | 1.1959 | 0.2933 |
| 560 nm | 46.6430 | 0.0001 | 2.3087 | 0.1579 | 0.8950 | 0.4917 | 2.0313 | 0.0516 | 1.4634 | 0.1432 | |
| 676 nm | 46.4310 | 0.0001 | 2.8527 | 0.1240 | 4.5216 | 0.1370 | 1.4033 | 0.2056 | 0.3838 | 0.9848 | |
| 694 nm | 13.9790 | 0.0002 | 0.3620 | 0.7100 | 0.8974 | 0.4975 | 1.8476 | 0.0757 | 0.7115 | 0.7784 | |
| 10 nm waveband (538 – 548 nm) | 48.4130 | 0.0001 | 2.3646 | 0.1586 | 1.2543 | 0.3281 | 1.8785 | 0.0727 | 1.2026 | 0.2909 | |
| 10 nm waveband (556 – 566 nm) | 46.9930 | 0.0001 | 2.3160 | 0.1636 | 0.8948 | 0.4876 | 2.0334 | 0.0483 | 1.4627 | 0.1420 | |
| 10 nm waveband (690 – 700 nm) | 14.9610 | 0.0004 | 0.3756 | 0.7003 | 0.9202 | 0.4741 | 1.8622 | 0.0746 | 0.7007 | 0.7826 | |
| 50 nm waveband (510 – 560 nm) | 55.7290 | 0.0001 | 2.4832 | 0.1486 | 1.2832 | 0.3195 | 2.1078 | 0.0415 | 1.2546 | 0.2503 | |
| 50 nm waveband (570 – 620 nm) | 89.4000 | 0.0001 | 2.9411 | 0.1324 | 0.8197 | 0.5181 | 1.9763 | 0.0563 | 1.2258 | 0.2765 | |
| 50 nm waveband (650 – 700 nm) | 64.0800 | 0.0001 | 2.0895 | 0.1928 | 4.1928 | 0.2030 | 1.6090 | 0.1288 | 0.3642 | 0.9878 | |
| Coral-ACTIS vs Resonon | Spectral similarity (SAM) | 9.7210 | 0.0019 | 3.0434 | 0.0911 | 0.7595 | 0.5624 | 1.6382 | 0.1206 | 1.3143 | 0.2141 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




