Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
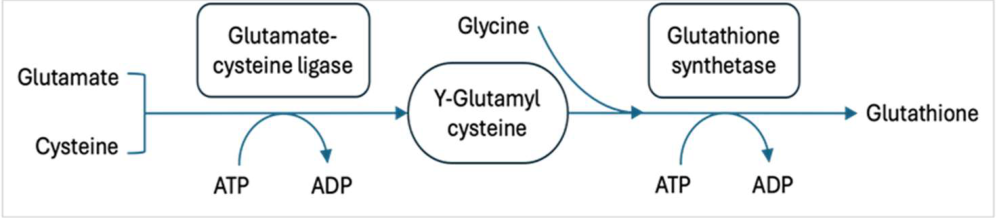
2. Materials and Methods
3. Results and Discussion
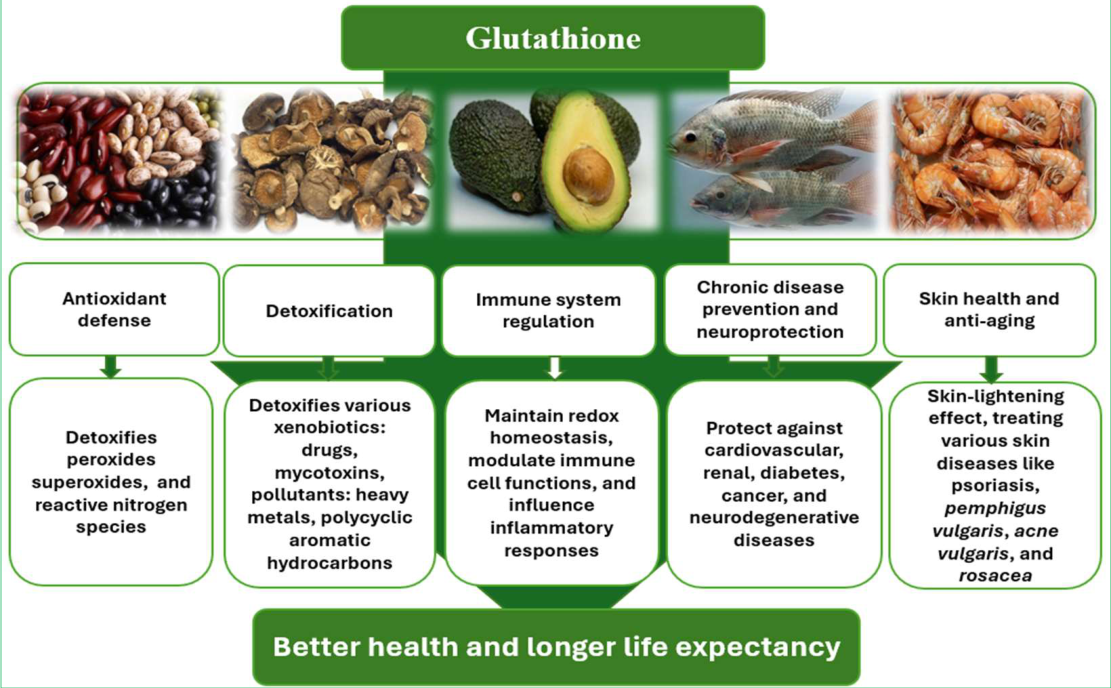
3.1. Benefits of Glutathione for the Body and Role in Disease Prevention
3.1.1. Antioxidant Defense
3.1.2. Detoxification
3.1.3. Immune System Regulation
3.1.4. Mitochondrial Protection
3.1.5. Chronic Disease Prevention
3.1.6. Neuroprotection
3.1.7. Skin Health and Anti-Aging
3.2. The Role of Diet in Increasing GSH Levels
3.2.1. The Best Dietary Sources of GSH
3.2.2. Foods to Increase Dietary Glutathione
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GSH | Glutathione |
| Nrf2 | Nuclear factor erythroid-2-related factor 2 |
| RNS | Reactive nitrogen species |
| ROS | Reactive oxygen species |
References
- Chai, Y.C.; Mieyal, J.J. Glutathione and glutaredoxin—key players in cellular redox homeostasis and signaling. Antioxidants 2023, 12(8), 1553. [Google Scholar] [CrossRef]
- Di Giacomo, C.; Malfa, G.A.; Tomasello, B.; Bianchi, S.; Acquaviva, R. Natural compounds and glutathione: Beyond mere antioxidants. Antioxidants 2023, 12, 1445. [Google Scholar] [CrossRef]
- Lana, J.F.S.D.; Rios, A.; Takeyama, R.; Santos, N.F.D.; Pires, L.; Santos, G.S.; Rodrigues, I.A.; Jeyaraman, M.; Purita, J.; Lana, J.F. Nebulized Glutathione as a Key Antioxidant for the Treatment of Oxidative Stress in Neurodegenerative Conditions. Nutrients 2024, 16, 2476. [Google Scholar] [CrossRef]
- Zandi, P.; Schnug, E. Reactive oxygen species, antioxidant responses and implications from a microbial modulation perspective. Biology 2022, 11, 155. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, S.; Tomar, A.; Yen, F.; Unlu, G.; Ropek, N.; Weber, R.; Wang, Y.; Khan, A.; Gad, M.; et al. Autoregulatory control of mitochondrial glutathione homeostasis. Science 2023, 382, 820–828. [Google Scholar] [CrossRef]
- Lian, G.; Gnanaprakasam, J.N.R.; Wang, T.; Wu, R.; Chen, X.; Liu, L.; Shen, Y.; Yang, M.; Yang, J.; Chen, Y.; et al. Glutathione de novo synthesis but not recycling process coordinates with glutamine catabolism to control redox homeostasis and directs murine T cell differentiation. eLife 2018, 7, e36158. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.C. Dysregulation of glutathione synthesis in liver disease. Liver Res. 2020, 4, 64–73. [Google Scholar] [CrossRef]
- Ikeda, Y.; Fujii, J. The Emerging Roles of γ-Glutamyl Peptides Produced by γ-Glutamyltransferase and the Glutathione Synthesis System. Cells 2023, 12, 2831. [Google Scholar] [CrossRef] [PubMed]
- Vairetti, M.; Di Pasqua, L.G.; Cagna, M.; Richelmi, P.; Ferrigno, A.; Berardo, C. Changes in Glutathione Content in Liver Diseases: An Update. Antioxidants 2021, 10, 364. [Google Scholar] [CrossRef]
- Vázquez-Meza, H.; Vilchis-Landeros, M.M.; Vázquez-Carrada, M.; Uribe-Ramírez, D.; Matuz-Mares, D. Cellular Compartmentalization, Glutathione Transport and Its Relevance in Some Pathologies. Antioxidants 2023, 12, 834. [Google Scholar] [CrossRef] [PubMed]
- Vašková, J.; Kočan, L.; Vaško, L.; Perjési, P. Glutathione-Related Enzymes and Proteins: A Review. Molecules 2023, 28, 1447. [Google Scholar] [CrossRef]
- Gasmi, A.; Nasreen, A.; Lenchyk, L.; Lysiuk, R.; Peana, M.; Shapovalova, N.; Piscopo, S.; Komissarenko, M.; Shanaida, M.; Smetanina, K.; et al. An Update on Glutathione's Biosynthesis, Metabolism, Functions, and Medicinal Purposes. Curr. Med. Chem. 2023, 31, 4579–4601. [Google Scholar] [CrossRef]
- Dwivedi, D.; Megha, K.; Mishra, R.; Mandal, P.K. Glutathione in Brain: Overview of Its Conformations, Functions, Biochemical Characteristics, Quantitation and Potential Therapeutic Role in Brain Disorders. Neurochem. Res. 2020, 45, 1461–1480. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, K. Glutathione in the Brain. Int. J. Mol. Sci. 2021, 22, 5010. [Google Scholar] [CrossRef] [PubMed]
- Panda, C.; Komarnytsky, S.; Fleming, M.; Marsh, C.; Barron, K.; Brun-Blashka, S.; Metzger, B. Guided Metabolic Detoxification Program Supports Phase II Detoxification Enzymes and Antioxidant Balance in Healthy Participants. Nutrients 2023, 15, 2209. [Google Scholar] [CrossRef]
- Narayanankutty, A.; Jdevagiri, J.S.; Narayanankutty, V.K. Glutathione, an antioxidant tripeptide: Dual roles in Carcinogenesis and Chemoprevention. Curr. Protein Pept. Sci. 2019, 20, 907–917. [Google Scholar] [CrossRef]
- Chen, F.; Zhang, X.; Wang, Z.; Xu, C.; Hu, J.; Liu, L.; Zhou, J.; Sun, B. Dual-responsive and NIR-driven free radical nanoamplifier with glutathione depletion for enhanced tumor-specific photothermal/thermodynamic/chemodynamic synergistic Therapy. Biomater. Sci. 2022, 10, 5912–5924. [Google Scholar] [CrossRef]
- Wróblewska, J.; Wróblewski, M.; Hołyńska-Iwan, I.; Modrzejewska, M.; Nuszkiewicz, J.; Wróblewska, W.; Woźniak, A. The Role of Glutathione in Selected Viral Diseases. Antioxidants 2023, 12, 1325. [Google Scholar] [CrossRef] [PubMed]
- Hristov, B. The role of glutathione metabolism in chronic illness development and its potential use as a novel therapeutic target. Cureus 2022, 14, e29015. [Google Scholar] [CrossRef]
- Çerkezi, S.; Nakova, M.; Gorgoski, I.; Ferati, K.; Bexheti-Ferati, A.; Palermo, A.; Inchingolo, A.M.; Ferrante, L.; Inchingolo, A.D.; Inchingolo, F.; et al. The Role of Sulfhydryl (Thiols) Groups in Oral and Periodontal Diseases. Biomedicines 2024, 12, 882. [Google Scholar] [CrossRef]
- Kulcsár, S.; Kövesi, B.; Balogh, K.; Zándoki, E.; Ancsin, Z.; Erdélyi, M.; Mézes, M. The Co-Occurrence of T-2 Toxin, Deoxynivalenol, and Fumonisin B1 Activated the Glutathione Redox System in the EU-Limiting Doses in Laying Hens. Toxins 2023, 15, 305. [Google Scholar] [CrossRef] [PubMed]
- Muller, M.; Merrett, N.D. Mechanism for glutathione-mediated protection against the Pseudomonas aeruginosa redox toxin, pyocyanin. Chem. Biol. Interact. 2015, 232, 30–37. [Google Scholar] [CrossRef]
- Branco, V.; Matos, B.; Mourato, C.; Diniz, M.; Carvalho, C.; Martins, M. Synthesis of glutathione as a central aspect of PAH toxicity in liver cells: A comparison between phenanthrene, Benzo[b]Fluoranthene and their mixtures. Ecotoxicol. Environ. Saf. 2021, 208, 111637. [Google Scholar] [CrossRef] [PubMed]
- Sauser, L.; Mohammed, T.O.; Kalvoda, T.; Feng, S.; Spingler, B.; Rulíšek, L.; Shoshan, M.S. Thiolation and Carboxylation of Glutathione Synergistically Enhance Its Lead-Detoxification Capabilities. Inorg. Chem. 2021, 60, 18620–18624. [Google Scholar] [CrossRef]
- Minich, D.M.; Brown, B.I. A Review of Dietary (Phyto)Nutrients for Glutathione Support. Nutrients 2019, 11, 2073. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Gevezova, M.; Sarafian, V.; Maes, M. Redox regulation of the immune response. Cell. Mol. Immunol. 2022, 19, 1079–1101. [Google Scholar] [CrossRef]
- Kurniawan, H.; Franchina, D.G.; Guerra, L.; Bonetti, L.; Baguet, L.S.; Grusdat, M.; Schlicker, L.; Hunewald, O.; Dostert, C.; Merz, M.P.; et al. Glutathione Restricts Serine Metabolism to Preserve Regulatory T Cell Function. Cell Metab. 2020, 31, 920–936. [Google Scholar] [CrossRef]
- Xu, C.; Sun, S.; Johnson, T.; Qi, R.; Zhang, S.; Zhang, J.; Yang, K. The glutathione peroxidase Gpx4 prevents lipid peroxidation and ferroptosis to sustain Treg cell activation and suppression of antitumor immunity. Cell Rep. 2021, 35, 109235. [Google Scholar] [CrossRef]
- Ferreira, A.C.; Koeken, V.A.C.M.; Matzaraki, V.; Kostidis, S.; Alarcon-Barrera, J.C.; De Bree, L.C.J.; Moorlag, S.J.C.F.M.; Mourits, V.P.; Novaković, B.; Giera, M.; et al. Glutathione Metabolism Contributes to the Induction of Trained Immunity. Cells 2021, 10, 971. [Google Scholar] [CrossRef]
- Abnousian, A.; Vásquez, J.; Sasaninia, K.; Kelley, M.; Venketaraman, V. Glutathione Modulates Efficacious Changes in the Immune Response against Tuberculosis. Biomedicines 2023, 11, 1340. [Google Scholar] [CrossRef]
- Wrotek, S.; Sobocińska, J.; Kozłowski, H.M.; Pawlikowska, M.; Jędrzejewski, T.; Dzialuk, A. New Insights into the Role of Glutathione in the Mechanism of Fever. Int. J. Mol. Sci. 2020, 21, 1393. [Google Scholar] [CrossRef]
- Zhang, T.; Tsutsuki, H.; Li, X.; Sawa, T. New insights into the regulatory roles of glutathione in NLRP3-inflammasome-mediated immune and inflammatory responses. J. Biochem. 2022, 171, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Marí, M.; De Gregorio, E.; De Dios, C.; Roca-Agujetas, V.; Cucarull, B.; Tutusaus, A.; Morales, A.; Colell, A. Mitochondrial Glutathione: Recent Insights and Role in Disease. Antioxidants 2020, 9, 909. [Google Scholar] [CrossRef]
- Chen, T.Y.; Wang, H.T.; Chang, C.C.; Lee, S.C. Mitochondrial Glutathione in Cellular Redox Homeostasis and Disease Manifestation. Int. J. Mol. Sci. 2024, 25, 1314. [Google Scholar] [CrossRef] [PubMed]
- Okhai, I.; Fedichkina, R.; Goshovska, Y.; Sagach, V. Mitochondrial function after endogenous glutathione synthesis stimulation in old rats heart. Cardiovasc. Res. 2022, 118, cvac066.028. [Google Scholar] [CrossRef]
- Strutynska, N.; Goshovska, Y.; Mys, L.; Strutynskyi, R.; Luchkova, A.; Fedichkina, R.; Okhai, I.; Korkach, Y.; Sagach, V. Glutathione restores the mitochondrial redox status and improves the function of the cardiovascular system in old rats. Front. Physiol. 2023, 13, 1093388. [Google Scholar] [CrossRef]
- Daniel, T.; Faruq, H.; Magdalena, J.; Manuela, G.; Horst, L. Role of GSH and Iron-Sulfur Glutaredoxins in Iron Metabolism—Review. Molecules 2020, 25, 3860. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.A.; Lin, T.H.; Sheehan, A.E.; Van Der Goes Van Naters, W.; Neukomm, L.J.; Graves, H.; Bis-Brewer, D.M.; Züchner, S.; Freeman, M.R. Glutathione S-Transferase Regulates Mitochondrial Populations in Axons through Increased Glutathione Oxidation. Neuron 2019, 103, 52–65. [Google Scholar] [CrossRef]
- Zhang, X.; Huang, Y.; Han, X.; Wang, Y.; Zhang, L.; Chen, L. Evaluating the protective effects of mitochondrial glutathione on cerebral ischemia/reperfusion injury via near-infrared fluorescence imaging. Anal. Chem. 2019, 91, 14728–14736. [Google Scholar] [CrossRef]
- Wang, Y.; Yen, F.S.; Zhu, X.G.; Timson, R.C.; Weber, R.; Xing, C.; Liu, Y.; Allwein, B.; Luo, H.; Yeh, H.W.; et al. SLC25A39 is necessary for mitochondrial glutathione import in mammalian cells. Nature 2021, 599, 136–140. [Google Scholar] [CrossRef]
- Young, A.; Gill, R.; Mailloux, R.J. Protein S-glutathionylation: The linchpin for the transmission of regulatory information on redox buffering capacity in mitochondria. Chem. Biol. Interact. 2019, 299, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Osahon, O.W.; Sekhar, R.V. GlyNAC (Glycine and N-Acetylcysteine) Supplementation in Mice Increases Length of Life by Correcting Glutathione Deficiency, Oxidative Stress, Mitochondrial Dysfunction, Abnormalities in Mitophagy and Nutrient Sensing, and Genomic Damage. Nutrients 2022, 14, 1114. [Google Scholar] [CrossRef]
- Søndergård, S.D.; Cintin, I.; Kuhlman, A.B.; Morville, T.; Bergmann, M.L.; Kjær, L.K.; Poulsen, H.E.; Giustarini, D.; Rossi, R.; Dela, F.; et al. The effects of 3 weeks of oral glutathione supplementation on whole body insulin sensitivity in obese males with and without type 2 diabetes: a randomized trial. Appl. Physiol. Nutr. Metab. 2021, 46, 1133–1142. [Google Scholar] [CrossRef]
- González-Menéndez, P.; Hevia, D.; Alonso-Arias, R.; Álvarez-Artime, A.; Rodríguez-García, A.; Kinet, S.; Gonzalez-Pola, I.; Taylor, N.; Mayo, J.C.; Sainz, R.M. GLUT1 protects prostate cancer cells from glucose deprivation-induced oxidative stress. Redox Biol. 2018, 17, 112–127. [Google Scholar] [CrossRef]
- Deka, B.; Barge, S.R.; Bharadwaj, S.; Kashyap, B.; Manna, P.; Borah, J.C.; Talukdar, N.C. Beneficial effect of the methanolic leaf extract of Allium hookeri on stimulating glutathione biosynthesis and preventing impaired glucose metabolism in type 2 diabetes. Arch. Biochem. Biophys. 2021, 708, 108961. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, L.; Sandhu, J.K.; Harper, M.E.; Cuperlovic-Culf, M. Role of Glutathione in Cancer: From Mechanisms to Therapies. Biomolecules 2020, 10, 1429. [Google Scholar] [CrossRef]
- Asantewaa, G.; Harris, I.S. Glutathione and its precursors in cancer. Curr. Opin. Biotechnol. 2021, 68, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Potęga, A. Glutathione-Mediated Conjugation of Anticancer Drugs: An Overview of Reaction Mechanisms and Biological Significance for Drug Detoxification and Bioactivation. Molecules 2022, 27, 5252. [Google Scholar] [CrossRef]
- Niu, B.; Liao, K.; Zhou, Y.; Wen, T.; Quan, G.; Pan, X.; Wu, C. Application of glutathione depletion in cancer therapy: Enhanced ROS-based therapy, ferroptosis, and chemotherapy. Biomaterials 2021, 277, 121110. [Google Scholar] [CrossRef]
- Kalinina, E. Glutathione-Dependent Pathways in Cancer Cells. Int. J. Mol. Sci. 2024, 25, 8423. [Google Scholar] [CrossRef]
- Roy, N.; Paira, P. Glutathione Depletion and Stalwart Anticancer Activity of Metallotherapeutics Inducing Programmed Cell Death: Opening a New Window for Cancer Therapy. ACS Omega 2024, 9, 20670–20701. [Google Scholar] [CrossRef]
- Zhang, X.; Sui, S.; Wang, L.; Li, H.; Zhang, L.; Xu, S.; Zheng, X. Inhibition of tumor propellant glutathione peroxidase 4 induces ferroptosis in cancer cells and enhances anticancer effect of cisplatin. J. Cell. Physiol. 2020, 235, 3425–3437. [Google Scholar] [CrossRef]
- Jain, S.K.; Justin Margret, J.; Abrams, S.A.; Levine, S.N.; Bhusal, K. The impact of Vitamin D and L-Cysteine co-supplementation on upregulating glutathione and Vitamin D-metabolizing genes and in the treatment of circulating 25-Hydroxy Vitamin D deficiency. Nutrients 2024, 16, 2004. [Google Scholar] [CrossRef]
- Limongi, R.; Jeon, P.; Théberge, J.; Palaniyappan, L. Counteracting effects of glutathione on the glutamate-driven excitation/inhibition imbalance in first-episode schizophrenia: a 7T MRS and dynamic causal modeling study. Antioxidants 2021, 10, 75. [Google Scholar] [CrossRef]
- Yamada, H.; Ono, S.; Wada, S.; Aoi, W.; Park, E.Y.; Nakamura, Y.; Sato, K. Statuses of food-derived glutathione in intestine, blood, and liver of rat. npj Sci. Food 2018, 2, 3. [Google Scholar] [CrossRef] [PubMed]
- Duperray, J.; Sergheraert, R.; Chalothorn, K.; Tachalerdmanee, P.; Perin, F. The effects of the oral supplementation of L-Cystine associated with reduced L-Glutathione-GSH on human skin pigmentation: a randomized, double-blinded, benchmark- and placebo-controlled clinical trial. J. Cosmet. Dermatol. 2021, 21, 802–813. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Yadav, V.; Yadav, T.; P, J.; Mandal, I. Glutathione as a skin-lightening agent and in melasma: a systematic review. Int. J. Dermatol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, G.; Malhotra, S.; Kaur, T.; Tyagi, S.; Bassan, R. Glutathione: The master antioxidant – Beyond skin lightening agent. Pigment Int. 2021, 8, 144–152. [Google Scholar] [CrossRef]
- Cui, X.; Mi, T.; Xiao, X.; Zhang, H.; Dong, Y.; Huang, N.; Gao, P.; Lee, J.S.; Guelakis, M.; Gu, X. Topical glutathione amino acid precursors protect skin against environmental and oxidative stress. J. Eur. Acad. Dermatol. Venereol. 2024, 38, 11–13. [Google Scholar] [CrossRef]
- Sitohang, I.B.E.; Ninditya, S. Systemic Glutathione as a Skin-Whitening Agent in Adult. Dermatol. Res. Pract. 2020, 2020, 8547960. [Google Scholar] [CrossRef]
- Yousefian, F.; Krejci, J. How to Put Your Patients on a “Hair Diet”. Int. Soc. Hair Restor. Surg. 2023, 33, 121–128. [Google Scholar] [CrossRef]
- Kumar, P.; Liu, C.; Suliburk, J.W.; Hsu, J.W.; Muthupillai, R.; Jahoor, F.; Minard, C.G.; Taffet, G.E.; Sekhar, R.V. Supplementing Glycine and N-Acetylcysteine (GlyNAC) in Older Adults Improves Glutathione Deficiency, Oxidative Stress, Mitochondrial Dysfunction, Inflammation, Physical Function, and Aging Hallmarks: A Randomized Clinical Trial. J. Gerontol. A Biol. Sci. Med. Sci. 2023, 78, 75–89. [Google Scholar] [CrossRef]
- Sekhar, R.V.; Kumar, P.; Hsu, J.W.; Suliburk, J.W.; Taffet, G.E.; Minard, C.G.; Jahoor, F.; Liu, C.Y. Correcting Glutathione Deficiency and Mitochondrial Dysfunction in Older Humans: A Randomized Clinical Trial. Innov. Aging 2019, 3, S416. [Google Scholar] [CrossRef]
- Sekhar, R.V.; Taffet, G.E.; Kumar, P. Glynac Supplementation in Older Adults Protects From Meal Driven Oxidative Stress and Inflammation: Results of a RCT. Innov. Aging 2022, 6, 814–815. [Google Scholar] [CrossRef]
- Sekhar, R.V.; Kumar, P.; Taffet, G.E.; Minard, C.G.; Suliburk, J.W.; Jahoor, F.; Liu, C.Y.; Muthupillai, R. Glynac Supplementation in Older Adults Improves Glutathione, Mitochondria, Aging Hallmarks, and Function. Innov. Aging 2024, 8, 203. [Google Scholar] [CrossRef]
- Agarwal, P.; Kant, K.; Kaushik, S.; Soni, A.; Fernandes, L.; Gowthami, R.S.; Mandal, I.; Padaki, V.; Kandangkel, L.; Khanna, R. Assessment of Anti-aging Efficacy of the Master Antioxidant Glutathione. Int. J. Sci. Basic Appl. Res. 2017, 33, 257–265. [Google Scholar]
- Detcheverry, F.; Senthil, S.; Narayanan, S.; Badhwar, A. Changes in levels of the antioxidant glutathione in brain and blood across the age span of healthy adults: A systematic review. NeuroImage Clin. 2023, 40, 103503. [Google Scholar] [CrossRef]
- Choi, I.Y.; Lee, P.; Denney, D.R.; Spaeth, K.; Nast, O.V.; Ptomey, L.T.; Roth, A.K.; Lierman, J.A.; Sullivan, D.K. Dairy intake is associated with brain glutathione concentration in older adults. Am. J. Clin. Nutr. 2015, 101, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Kalaras, M.D.; Richie, J.P.; Calcagnotto, A.; Beelman, R.B. Mushrooms: A rich source of the antioxidants ergothioneine and glutathione. Food Chem. 2017, 233, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Fujita, M. Exogenous glutathione confers high temperature stress tolerance in mung bean (Vigna radiata L.) by modulating antioxidant defense and methylglyoxal detoxification system. Environ. Exp. Bot. 2015, 112, 44–54. [Google Scholar] [CrossRef]
- Colville, L.; Sáez, C.; Lewis, G.P.; Kranner, I. The distribution of glutathione and homoglutathione in leaf, root and seed tissue of 73 species across the three sub-families of the Leguminosae. Phytochemistry 2015, 115, 175–183. [Google Scholar] [CrossRef]
- Demirkol, O.; Adams, C.; Ercal, N. Biologically important thiols in various vegetables and fruits. J. Agric. Food Chem. 2004, 52, 8151–8154. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.P.; Coates, R.J.; Flagg, E.W.; Eley, J.W.; Block, G.; Greenberg, R.S.; Schoeller, D.A.; Lanza, E.; Jackson, B. Glutathione in foods listed in the National Cancer Institute's health habits and history food frequency questionnaire. Nutr. Cancer 1992, 17, 57–75. [Google Scholar] [CrossRef]
- Mikulková, K.; Illek, J.; Bezděková, Z.; Šimková, I. Glutathione as an antioxidant marker: determination of glutathione concentration in the breast muscles and liver of broilers supplemented with different selenium sources. Acta Vet. Brno 2019, 88, 157–163. [Google Scholar] [CrossRef]
- Rakowska, R.; Sadowska, A.; Waszkiewicz-Robak, B. Influence of pre-and post-slaughter factors on the reduced glutathione content of beef muscles. Meat Sci. 2017, 124, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Atli, G.; Canli, M. Responses of metallothionein and reduced glutathione in a freshwater fish Oreochromis niloticus following metal exposures. Environ. Toxicol. Pharmacol. 2008, 25, 33–38. [Google Scholar] [CrossRef]
- Mika, A.; Skorkowski, E.F.; Stepnowski, P. The use of different MS techniques to determine glutathione levels in marine tissues. Food Anal. Methods 2013, 6, 789–802. [Google Scholar] [CrossRef]
- Monteiro, D.A.; Rantin, F.T.; Kalinin, A.L. Inorganic mercury exposure: toxicological effects, oxidative stress biomarkers and bioaccumulation in the tropical freshwater fish matrinxã, Brycon amazonicus (Spix and Agassiz, 1829). Ecotoxicology 2010, 19, 105–123. [Google Scholar] [CrossRef]
- Grim, J.M.; Simonik, E.A.; Semones, M.C.; Kuhn, D.E.; Crockett, E.L. The glutathione-dependent system of antioxidant defense is not modulated by temperature acclimation in muscle tissues from striped bass, Morone saxatilis. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2013, 164, 383–390. [Google Scholar] [CrossRef]
- Ognjanović, B.I.; Milovanović, J.G.; Đorđević, N.Z.; Marković, S.D.; Žikić, R.V.; Štajn, A.Š.; Saičić, Z.S. Parameters of oxidative stress in liver and white muscle of hake (Merluccius merluccius L.) from the Adriatic Sea. Kragujev. J. Sci. 2008, 30, 137–144. [Google Scholar]
- Kovačević, T.B.; Borković, S.S.; Pavlović, S.Z.; Despotović, S.G.; Saičić, Z.S. Glutathione as a suitable biomarker in hepatopancreas, gills and muscle of three freshwater crayfish species. Arch. Biol. Sci. 2008, 60, 59–66. [Google Scholar] [CrossRef]
- Pazos, M.; González, M.J.; Gallardo, J.M.; Torres, J.L.; Medina, I. Preservation of the endogenous antioxidant system of fish muscle by grape polyphenols during frozen storage. Eur. Food Res. Technol. 2005, 220, 514–519. [Google Scholar] [CrossRef]
- Al-Temimi, A.A.; Al-Mossawi, A.A.; Al-Hilifi, S.A.; Korma, S.A.; Esatbeyoglu, T.; Rocha, J.M.; Agarwal, V. Glutathione for Food and Health Applications with Emphasis on Extraction, Identification, and Quantification Methods: A Review. Metabolites 2023, 13, 465. [Google Scholar] [CrossRef]
- Bruggeman, B.K.; Storo, K.E.; Fair, H.M.; Wommack, A.J.; Carriker, C.R.; Smoliga, J.M. The absorptive effects of orobuccal non-liposomal nano-sized glutathione on blood glutathione parameters in healthy individuals: A pilot study. PLoS ONE 2019, 14, e0215815. [Google Scholar] [CrossRef] [PubMed]
- Giustarini, D.; Milzani, A.; Dalle-Donne, I.; Rossi, R. How to increase cellular glutathione. Antioxidants 2023, 12, 1094. [Google Scholar] [CrossRef]
- Gould, R.L.; Pazdro, R. Impact of Supplementary Amino Acids, Micronutrients, and Overall Diet on Glutathione Homeostasis. Nutrients 2019, 11, 1056. [Google Scholar] [CrossRef] [PubMed]
- Gut, P.; Lizzo, G.; Migliavacca, E.; Karagounis, L.; Heise, T.; von Eynatten, M. Effects of glycine and n-acetylcysteine on glutathione levels and mitochondrial energy metabolism in healthy aging. Innov. Aging 2021, 5, 689–690. [Google Scholar] [CrossRef]
- He, W.; Posey, E.A.; Steele, C.C.; Savell, J.W.; Bazer, F.W.; Wu, G. Dietary glycine supplementation enhances glutathione availability in tissues of pigs with intrauterine growth restriction. J. Anim. Sci. 2024, 102, skae025. [Google Scholar] [CrossRef]
- Rodrigues, C.; Percival, S.S. Immunomodulatory Effects of Glutathione, Garlic Derivatives, and Hydrogen Sulfide. Nutrients 2019, 11, 295. [Google Scholar] [CrossRef]
- Distefano, A.; Orlando, L.; Giallongo, S.; Tropea, E.; Spampinato, M.; Santisi, A.; Sciacca, S.; Nicosia, A.; Zingale, V.D.; Li Volti, G.; et al. Fish oil containing pro-resolving mediators enhances the antioxidant system and ameliorates LPS-induced inflammation in human bronchial epithelial cells. Pharmaceuticals 2024, 17, 1066. [Google Scholar] [CrossRef]
- Biswas, P.; Dellanoce, C.; Vezzoli, A.; Mrakic-Sposta, S.; Malnati, M.; Beretta, A.; Accinni, R. Antioxidant Activity with Increased Endogenous Levels of Vitamin C, E and A Following Dietary Supplementation with a Combination of Glutathione and Resveratrol Precursors. Nutrients 2020, 12, 3224. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.; El-Dreny, E.; Shaheen, M. The Effect of Dietary Supplement on Glutathione Level in Rats. Eur. J. Nutr. Food Saf. 2022, 14, 1–11. [Google Scholar] [CrossRef]
- Wang, D.; Wang, T.; Li, Z.; Guo, Y.; Granato, D. Green tea polyphenols upregulate the Nrf2 signaling pathway and suppress oxidative stress and inflammation markers in D-galactose-induced liver aging in mice. Front. Nutr. 2022, 9, 836112. [Google Scholar] [CrossRef] [PubMed]
- Dludla, P.V.; Ziqubu, K.; Mabhida, S.E.; Mazibuko-Mbeje, S.E.; Hanser, S.; Nkambule, B.B.; Basson, A.K.; Pheiffer, C.; Tiano, L.; Kengne, A.P. Dietary Supplements Potentially Target Plasma Glutathione Levels to Improve Cardiometabolic Health in Patients with Diabetes Mellitus: A Systematic Review of Randomized Clinical Trials. Nutrients 2023, 15, 944. [Google Scholar] [CrossRef] [PubMed]
- Deth, R.; Clarke, A.; Ni, J.; Trivedi, M. Clinical evaluation of glutathione concentrations after consumption of milk containing different subtypes of β-casein: results from a randomized, cross-over clinical trial. Nutr. J. 2015, 15, 1–6. [Google Scholar] [CrossRef]
- De Souza, R.G.M.; Gomes, A.C.; Navarro, A.M.; Cunha, L.C.V.; Silva, M.S.; Júnior, F.B.A.; Mota, J.F. Baru Almonds Increase the Activity of Glutathione Peroxidase in Overweight and Obese Women: A Randomized, Placebo-Controlled Trial. Nutrients 2019, 11, 1750. [Google Scholar] [CrossRef]
- Li, Y.; Clark, C.; Abdulazeem, H.; Salehisahlabadi, A.; Rahmani, J.; Zhang, Y. The effect of Brazil nuts on selenium levels, Glutathione peroxidase, and thyroid hormones: A systematic review and meta-analysis of randomized controlled trials. J. King Saud. Univ. Sci. 2020, 32, 1845–1852. [Google Scholar] [CrossRef]
- Guardado-Félix, D.; Antunes-Ricardo, M.; Rocha-Pizaña, M.R.; Martínez-Torres, A.C.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Chickpea (Cicer arietinum L.) sprouts containing supranutritional levels of selenium decrease tumor growth of colon cancer cells xenografted in immune-suppressed mice. J. Funct. Foods 2019, 53, 76–84. [Google Scholar] [CrossRef]
- Ahmad, P.; Latef, A.A.H.; Hashem, A.; Abd_Allah, E.F.; Gucel, S.; Tran, L.-S.P. Nitric Oxide Mitigates Salt Stress by Regulating Levels of Osmolytes and Antioxidant Enzymes in Chickpea. Front. Plant Sci. 2016, 7, 347. [Google Scholar] [CrossRef]
- Kumar, A.; Dubey, A.; Kumar, V.; Ansari, M.A.; Narayan, S.; Kumar, M.; Kumar, S.; Pandey, V.; Shirke, P.A.; Pande, V.; et al. Over-expression of chickpea glutaredoxin (CaGrx) provides tolerance to heavy metals by reducing metal accumulation and improved physiological and antioxidant defence system. Ecotoxicol. Environ. Saf. 2020, 192, 110252. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, V.; Dubey, A.; Ansari, M.A.; Narayan, S.; Kumar, M.; Kumar, S.; Pandey, V.; Pande, V.; Sanyal, I. Chickpea glutaredoxin (CaGrx) gene mitigates drought and salinity stress by modulating the physiological performance and antioxidant defense mechanisms. Physiol. Mol. Biol. Plants 2021, 27, 923–944. [Google Scholar] [CrossRef]
- Zhang, P.; Li, S.; Guo, Z.; Lu, S. Nitric oxide regulates glutathione synthesis and cold tolerance in forage legumes. Environ. Exp. Bot. 2019, 167, 103851. [Google Scholar] [CrossRef]
- Yoo, G.; Kim, E.; Kang, H.; Kim, J.; Yeo, W.S. Mass spectrometric investigation of concentration-dependent effect of curcumin and oxidative stress on intracellular glutathione levels. Anal. Bioanal. Chem. 2020, 412, 2873–2880. [Google Scholar] [CrossRef] [PubMed]
- Pressman, P.; Bridge, W.; Zarka, M.; Hayes, A.W.; Clemens, R. Dietary γ -Glutamylcysteine: Its Impact on Glutathione Status and Potential Health Outcomes. J. Diet. Suppl. 2022, 19, 259–270. [Google Scholar] [CrossRef]
- Mangkalopakorn, P.; Segsarnviriya, C.; Keeratipranon, M.; Maiprasert, M. Glutathione Levels after Glutathione Supplementation: A Systematic Review and Meta-analysis. J. Curr. Sci. Technol. 2025, 15, 81–101. [Google Scholar] [CrossRef]
- Braidy, N.; Zarka, M.; Jugder, B.E.; Welch, J.; Jayasena, T.; Chan, D.K.P.; Wen, W.; Sachdev, P.S.; Bridge, W. The precursor to glutathione (GSH), γ Glutamylcysteine (GGC), can ameliorate oxidative damage and neuroinflammation induced by Aβ40 oligomers in human astrocytes. Front. Aging Neurosci. 2019, 11, 177. [Google Scholar] [CrossRef]
- Lizzo, G.; Migliavacca, E.; Lamers, D.; Frézal, A.; Corthésy, J.; Vinyes-Parés, G.; Bosco, N.; Karagounis, L.G.; Hövelmann, U.; Heise, T.; et al. A Randomized Controlled Clinical Trial in Healthy Older Adults to Determine Efficacy of Glycine and N-Acetylcysteine Supplementation on Glutathione Redox Status and Oxidative Damage. Front. Aging 2022, 3, 852569. [Google Scholar] [CrossRef]
- Weschawalit, S.; Thongthip, S.; Phutrakool, P.; Asawanonda, P. Glutathione and its antiaging and antimelanogenic effects. Clin. Cosmet. Investig. Dermatol. 2017, 10, 147–153. [Google Scholar] [CrossRef]
- Alzahrani, T.; Alotaibi, S.N.; Alzahrani, A.S.; Alzahrani, A.A.; Alturki, L.S.; Alshammari, M.D.; Alharbi, R.K.; Alanazi, S.F.; Alshammari, W.F.; Algarni, A.S. Exploring the Safety and Efficacy of Glutathione Supplementation for Skin Lightening: A Narrative Review. Cureus 2025, 17, e78045. [Google Scholar] [CrossRef]
- Camillerapp, C.; Mayfield, D.; Papineni, S.; Leroux, A.; Jeanne, J. Safety Assessment of S-Acetyl Glutathione for Use in Foods and Dietary Supplements. Food Chem. Toxicol. 2025, 115279. [Google Scholar] [CrossRef]
- Kalamkar, S.; Acharya, J.; Madathil, A.; Gajjar, V.; Divate, U.; Karandikar-Iyer, S.; Goel, P.; Ghaskadbi, S. Randomized Clinical Trial of How Long-Term Glutathione Supplementation Offers Protection from Oxidative Damage and Improves HbA1c in Elderly Type 2 Diabetic Patients. Antioxidants 2022, 11, 1026. [Google Scholar] [CrossRef]
- Tuell, D.; Ford, G.; Los, E.; Stone, W.L. The Role of Glutathione and Its Precursors in Type 2 Diabetes. Antioxidants 2024, 13, 184. [Google Scholar] [CrossRef]
- Marini, H.R.; Facchini, B.A.; Di Francia, R.; Freni, J.; Puzzolo, D.; Montella, L.; Ieni, A.; Irrera, N.; Minutoli, L. Glutathione: Lights and Shadows in Cancer Patients. Biomedicines 2023, 11, 2226. [Google Scholar] [CrossRef]
| Food Source | GSH Content (mg per 100 g) | Reference(s) |
| Plant foods | ||
| Mushrooms, dried | 11.00–241.00 | [69] |
| Legumes (mung beans, black beans, kidney beans, pinto beans etc.) | 15.00–37.00 | [70,71] |
| Spinach | 9.62–28.90 | [72,73] |
| Asparagus | 10.73–21.80 | [73] |
| Avocado | 10.42–20.60 | [72] |
| Squash, zucchini | 8.40–11.40 | |
| Potatoes (baked or boiled) | 10.20–11.00 | [72] |
| Broccoli, brussels sprouts | 1.90–10.00 | |
| Carrots, tomatoes | 5.90–7.50 | [73] |
| Strawberries, grapefruit, cantaloupe, | 6.10–6.90 | |
| Green, red sweet peppers, garlic | 3.40–5.50 | |
| Nectarines, peaches, melons, watermelons | 4.90–5.00 | |
| Oranges, lemons, papaya, mangoes | 4.18–4.80 | |
| Cauliflower, cucumbers | 3.78–4.00 | |
| Bananas, pears, nuts and seeds, walnuts | 3.30–3.70 | |
| Meat, subproducts | ||
| Chicken breast | 36.26 | [74] |
| Veal cutlet | 26.30 | [73] |
| Pork | 13.70–18.90 | |
| Beef, liver (chicken) | 11.80–15.34 | [73] [75] |
| Chicken | 6.50–7.70 | [73] |
| Fish, seafood | ||
| O. Niloticus | 245.86 | [76] |
| C. Crangon | 169.03 | [77] |
| B. amazonicus, heart | 83.59 | [78] |
| C. harengus membras | 73.76 | [77] |
| M. saxatilis, heart | 49.17 | [79] |
| P. maxima | 39.34 | [77] |
| Merluccius merluccius | 23.00 | [80] |
| B. amazonicus, muscle | 17.83 | [77] |
| Morone saxatilis, striped bass (sceletal muscle) | 12.29 | [79] |
| Austropotamobius torrentium, Astacus astacus, | [81] | |
| Orconectes limosus | 6.76–8.30 | [73] |
| Fish (cod and perch), pan fried | 5.70 | [82] |
| Food/Supplement | Mechanism of Action | Reference(s) |
| Cysteine, glycine, glutamate | These amino acids are direct precursors for GSH synthesis, enhancing tissue GSH levels. | [25,42,86,87] |
| Glycine | Enhances synthesis and concentration of GSH in various tissues. | [88] |
| Garlic, onions | Contains sulfur compounds such as S-allylcysteine that enhance GSH levels. Onion extracts and flavonoids like quercetin can increase intracellular GSH levels by activating the gamma-glutamylcysteine synthetase promoter, which is essential for GSH synthesis. | [89] |
| Brassica vegetables (sulforaphane), lipoic acid | Nrf2 activators in the body; Nrf2 upregulates enzymes for GSH synthesis and cystine uptake; Nrf2 activators increase GSH in many cell types | [25] |
| Omega-3 fatty acids | Modulate the GSH network by activating Nrf2 and boosting synthesis and GPx activity in many normal tissues | [90] |
| GSH and resveratrol precursors | Increase endogenous levels of vitamins C, E, and A, enhancing antioxidant activity. | [91] |
| Plant species Allium hookeri | Rich in organosulfur compounds, it increases GSH levels and regulates glucose metabolism. | [45] |
| Spirulina, turmeric | These foods increase antioxidant enzymes and GSH levels, reducing oxidative stress. | [92] |
| Green tea (especially EGCG-rich polyphenols) | increases GSH by activating Nrf2 signaling, up-regulating GSH-related enzymes, and enhancing antioxidant and detoxification pathways | [93] |
| Vitamins C and E, along with selenium | Vitamins and micronutrients are known to support GSH levels by maintaining its reduced form and enhancing its antioxidant capacity. | [86,91,94] |
| Milk | Milk containing A2 β-casein can promote the production of GSH in humans. | [68,95] |
| Nuts and seeds (particularly baru almonds and Brazil nuts) | Source of selenium, a key component of GSH peroxidase, can increase GSH levels in the body by enhancing the activity of GSH peroxidase. | [96,97] |
| Legumes, e.g., lentils, chickpeas (Cicer arietinum L) | Provide protein and may aid in the synthesis of GSH. Contain compounds that enhance the activity of antioxidant enzymes, including GSH reductase and GSH peroxidase. They express glutaredoxin, a protein that works with GSH to reduce oxidative stress and glutaredoxin helps maintain GSH levels. Chickpeas contain bioactive compounds like selenium and isoflavonoids, which can enhance the activity of GSH peroxidase. |
[98,99,100,101] |
| Legumes like Medicago falcata and Medicago truncatula | Reach of nitric oxide (NO), which plays a crucial role in regulating GSH synthesis and influences the expression of genes involved in GSH synthesis, such as γ-glutamylcysteine synthetase and GSH synthetase. | [102] |
| GlyNAC (Glycine and N-Acetylcysteine) supplements | Improves GSH deficiency and mitochondrial function in older adults, lowers oxidative stress. | [42] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




