Submitted:
30 April 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Preparation of Solutions
2.2. UVC-Irradiation Procedure
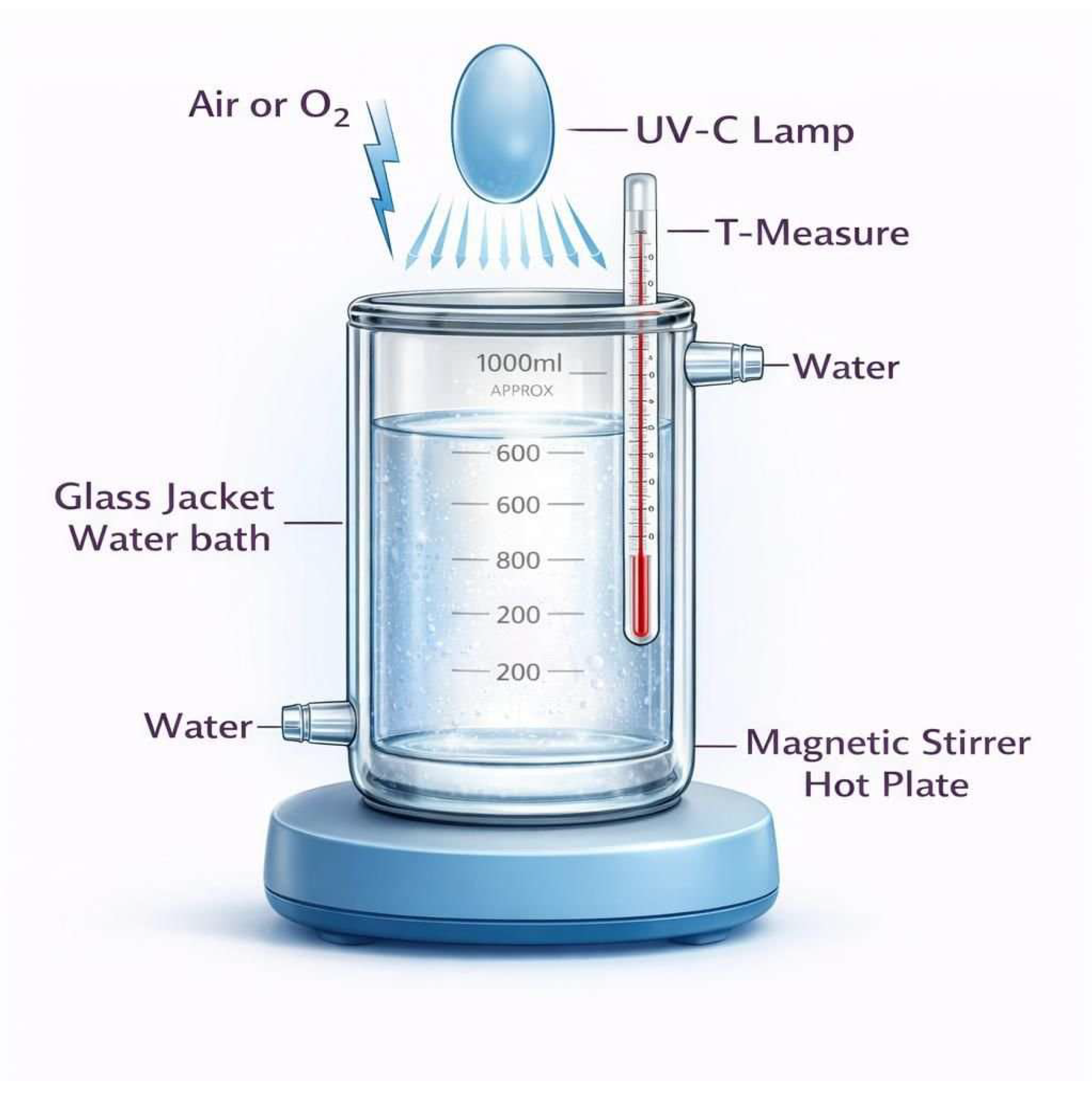
2.3. Viscosity and Molar Mass Determination
2.4. Photodegradation Kinetics
| Parameter | Equation | Eq. No. |
|---|---|---|
| Overall First-order Rate Law | // | (4) |
| Half-life | (5) | |
| helf-life (90%) | // | (6) |
| Arrhenius-type relation | (7) | |
| Eyring equation | (8) |
2.5. Conformational Modelling
3. Results and Discussion
3.1. Photo-Modification Kinetics
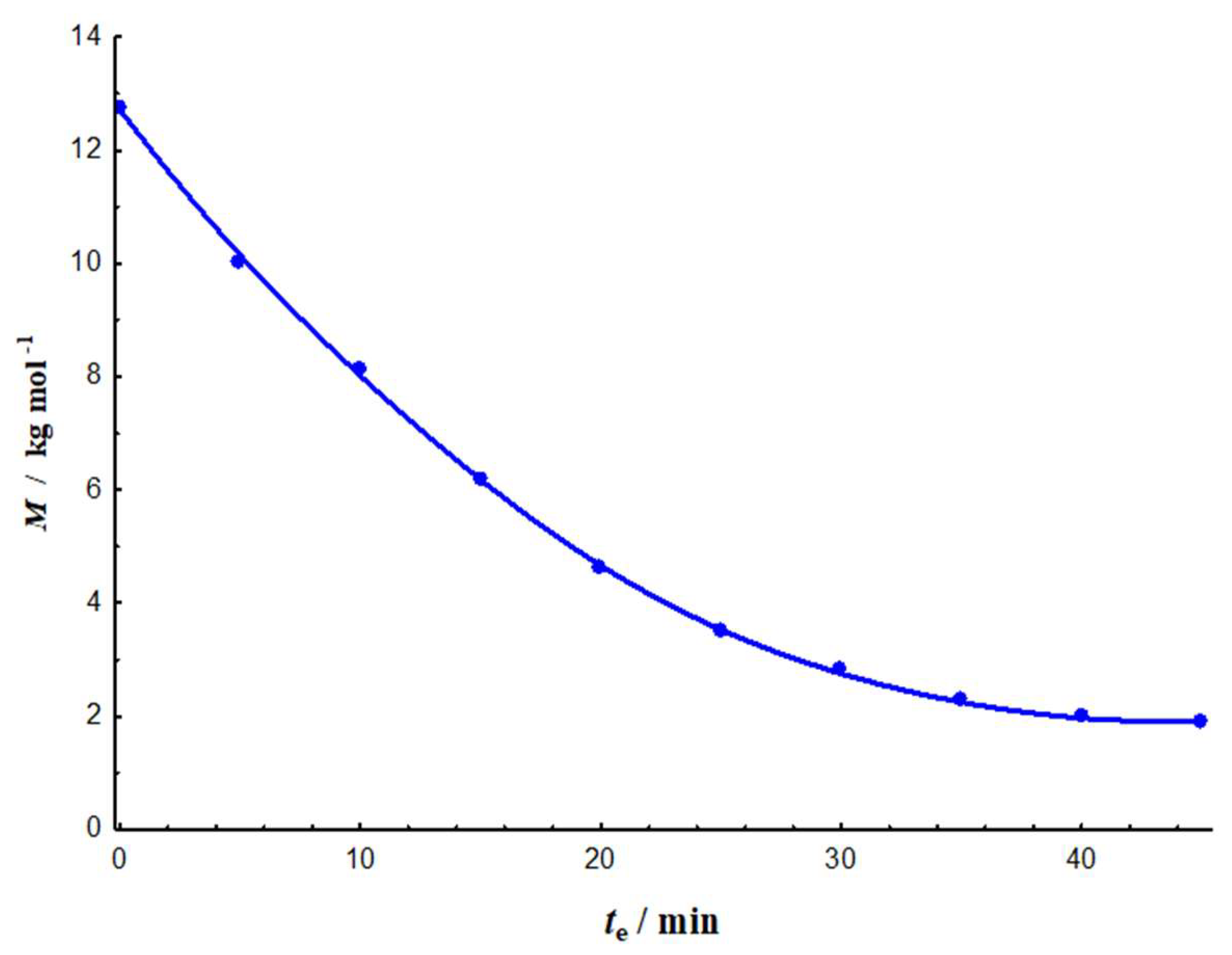
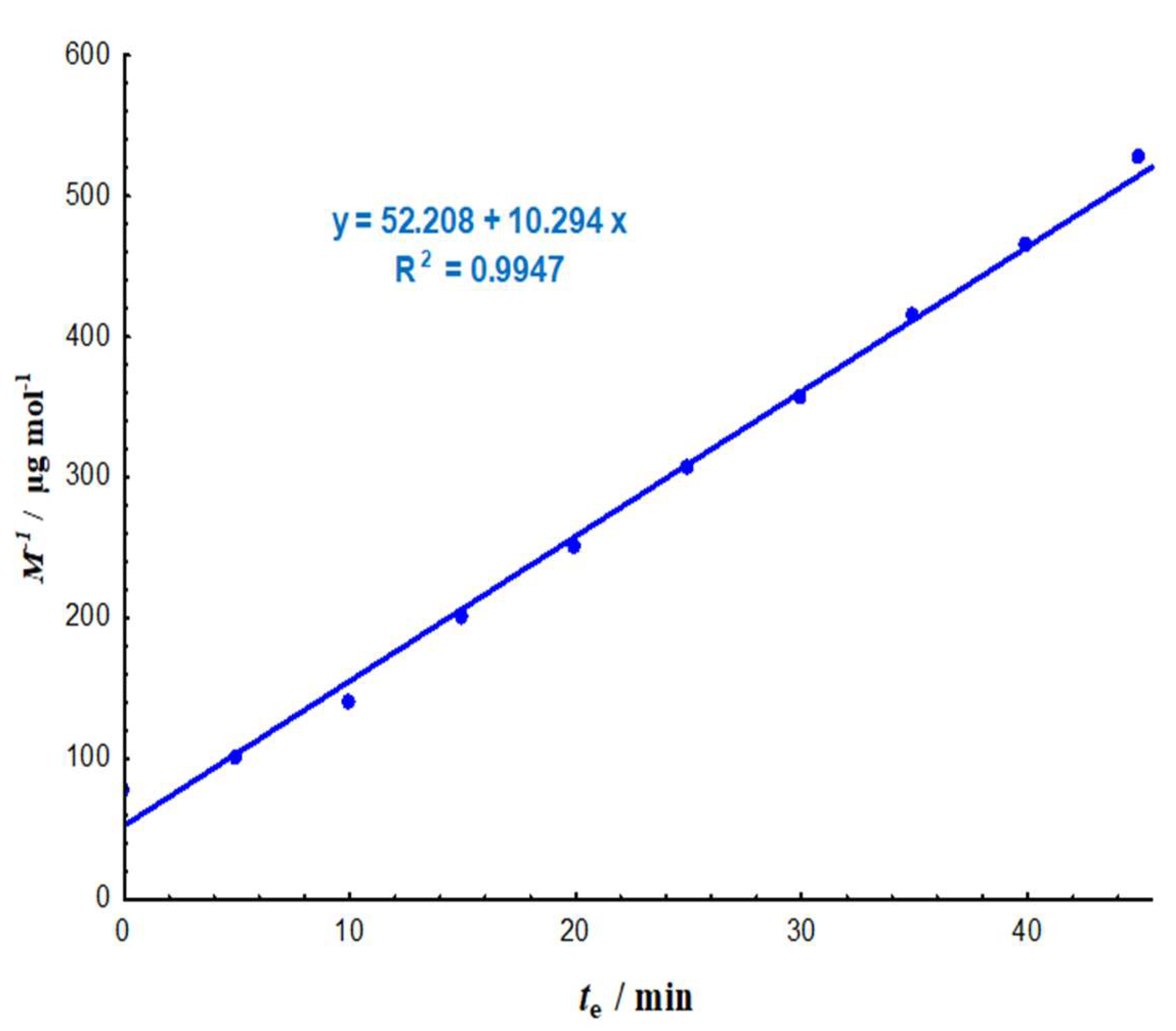
3.1.1. Degradation Rate and Lifetimes
3.1.2. Thermodynamic Interpretation
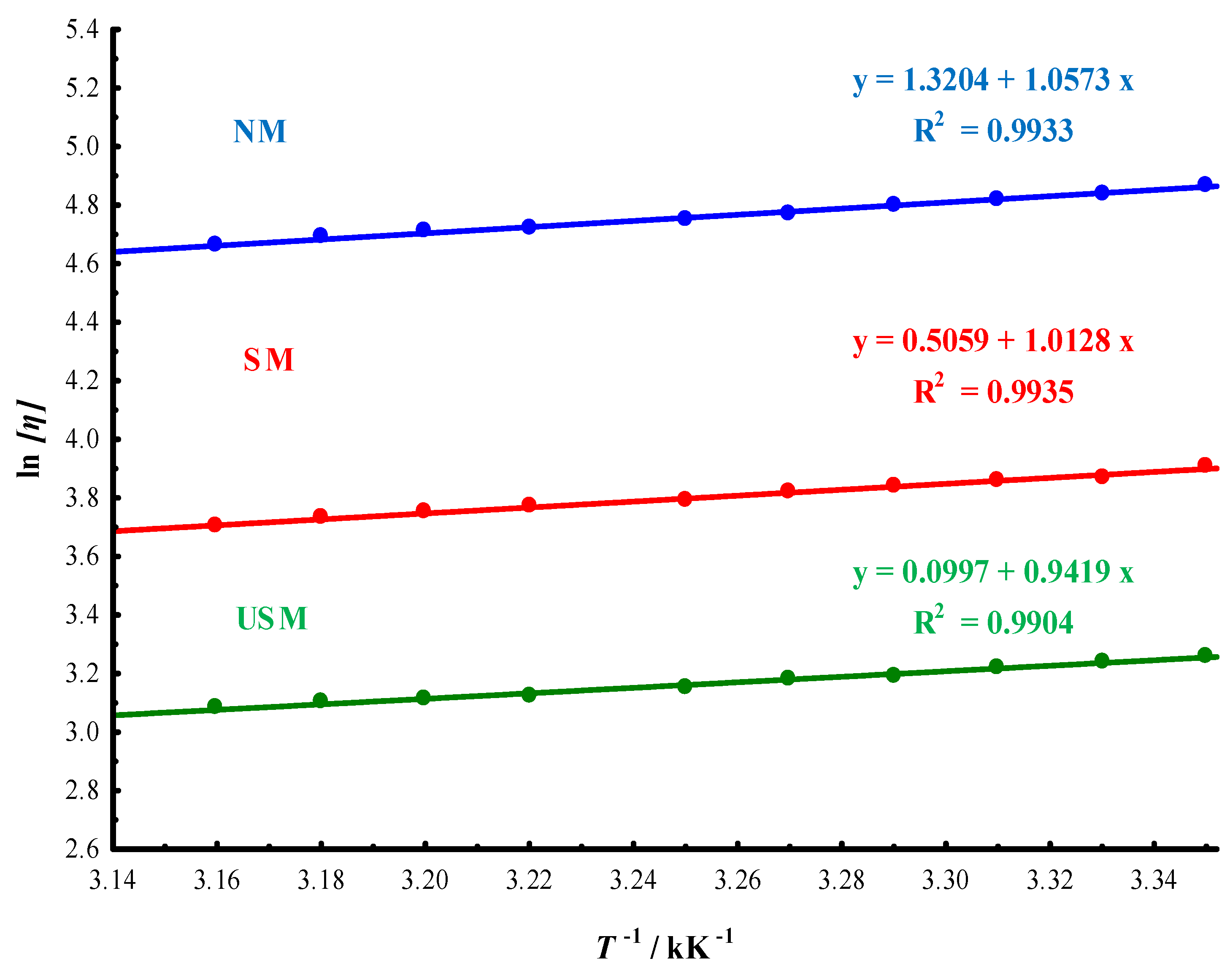
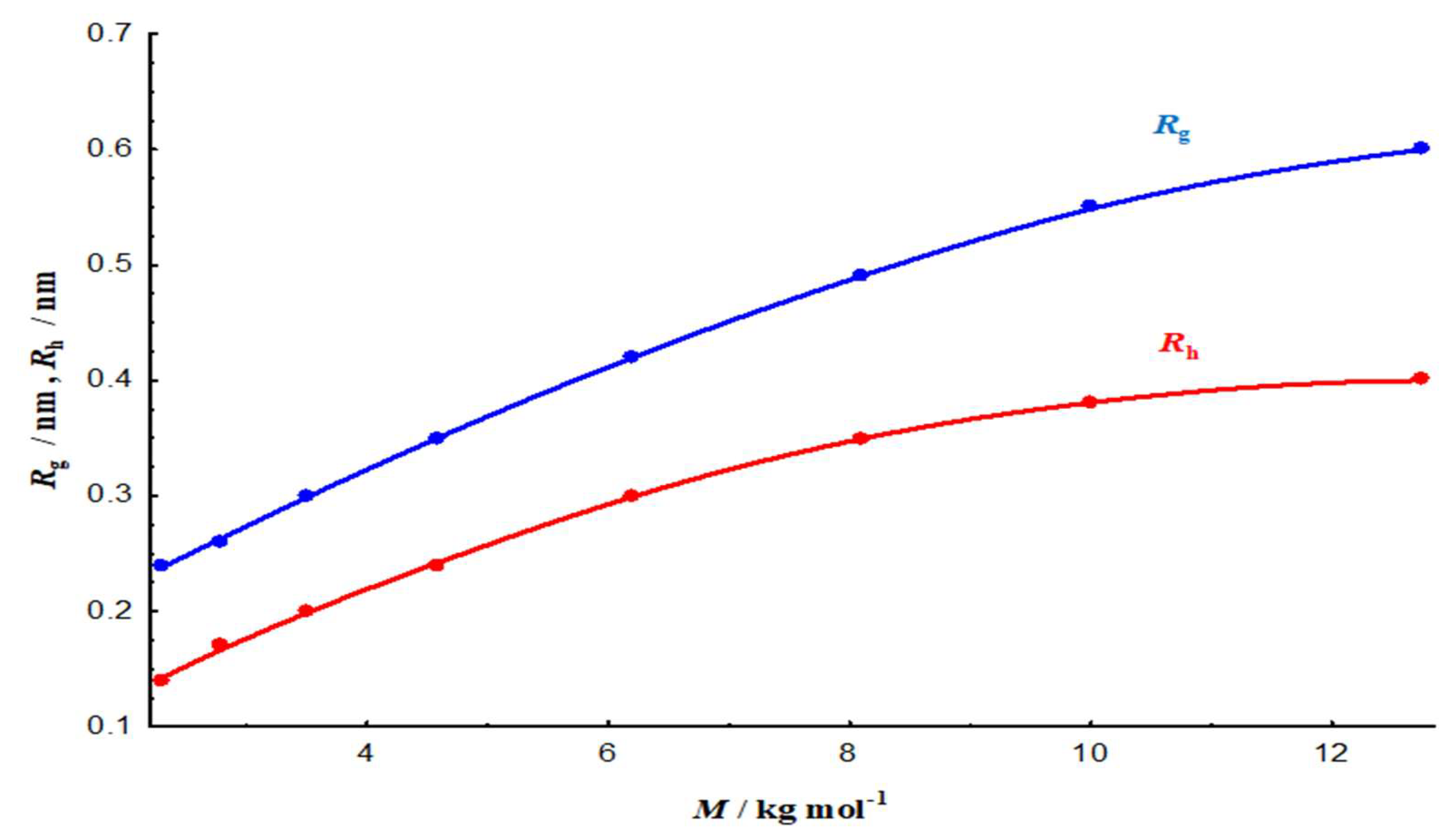
3.2. Macromolecular Characteristics
3.2.1. Molecular-Weight Dependence and Size Behaviour
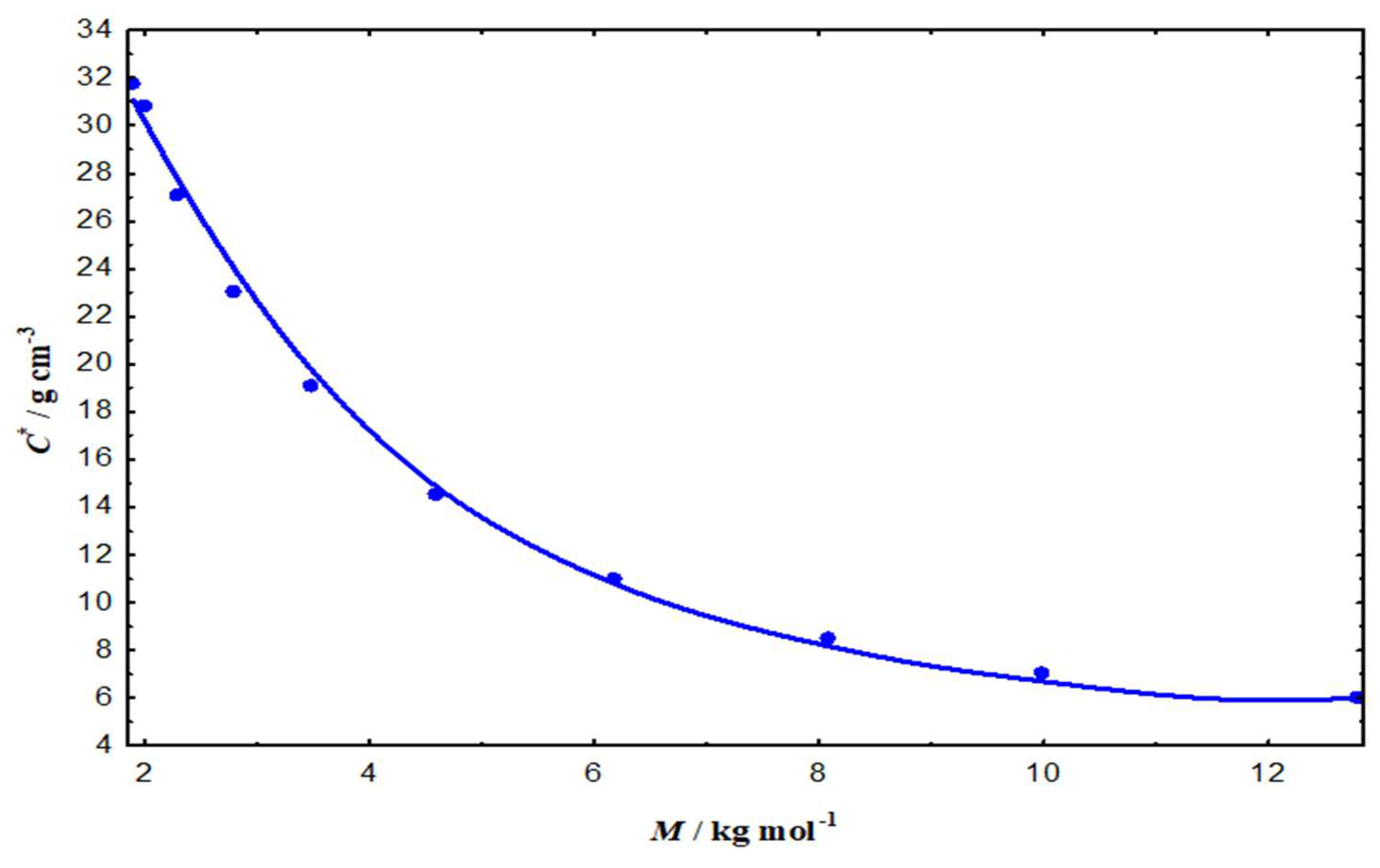
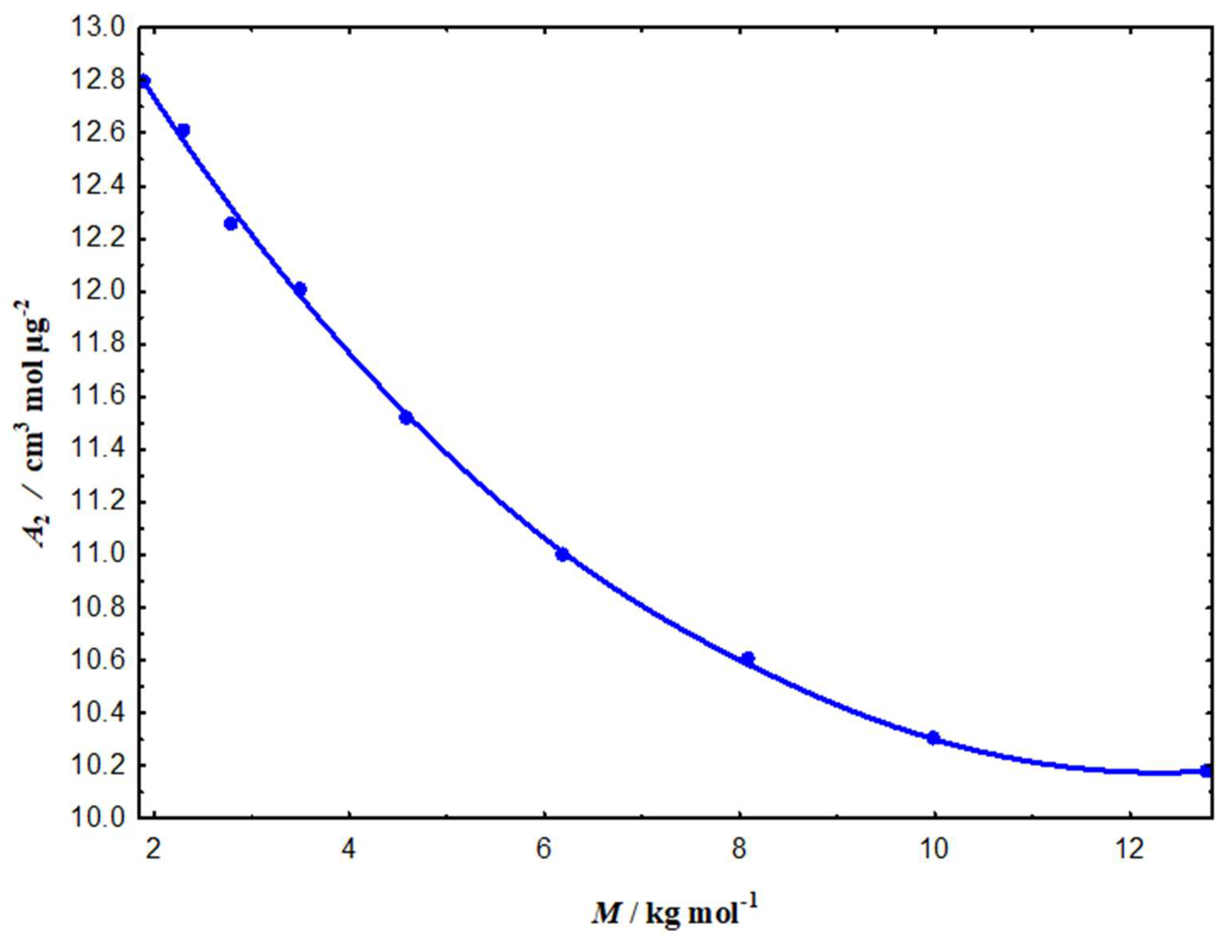
3.2.2. Impact on Critical Overlap and Thermodynamic Stability
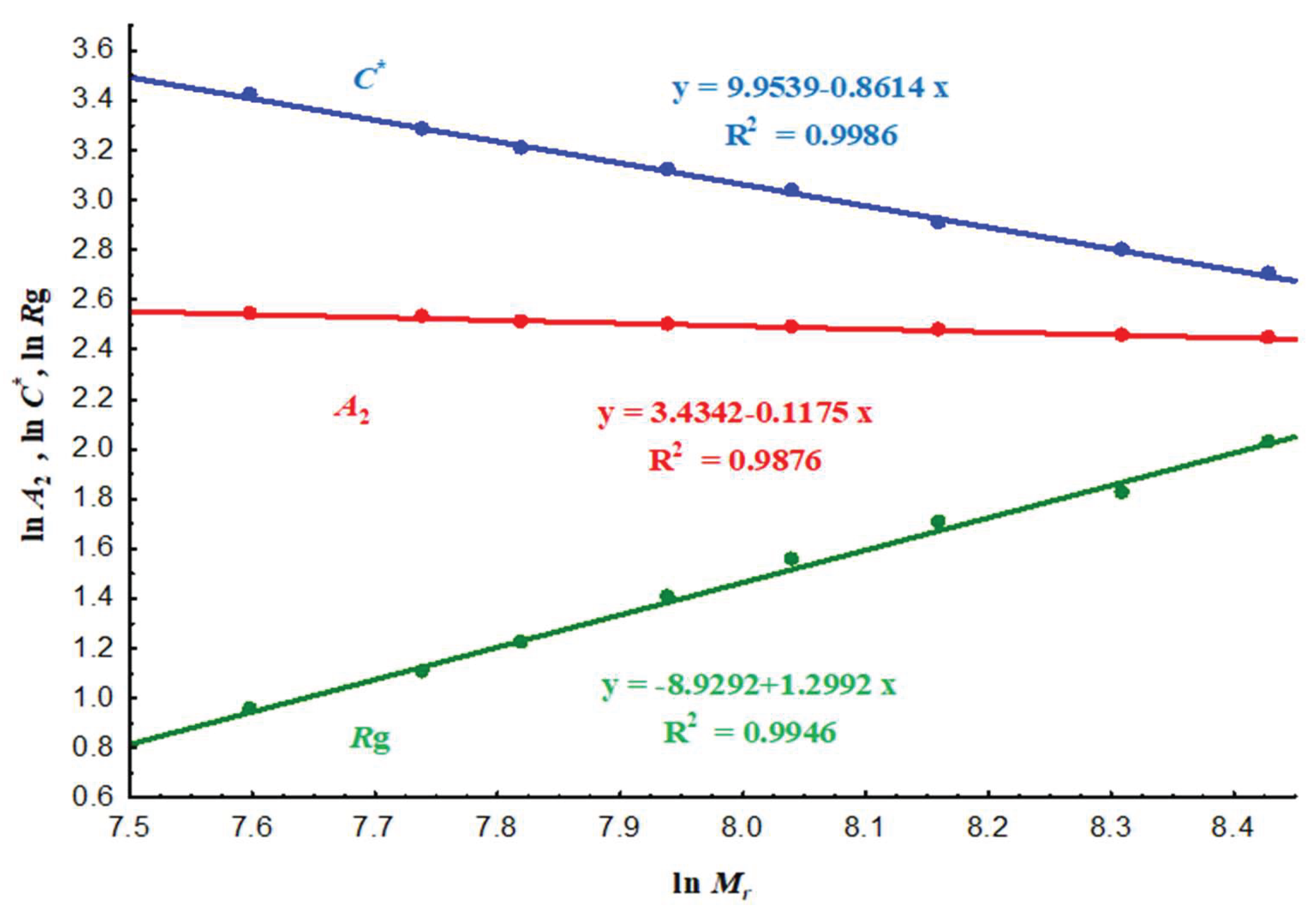
3.2.3. Scaling laws and conformational regime
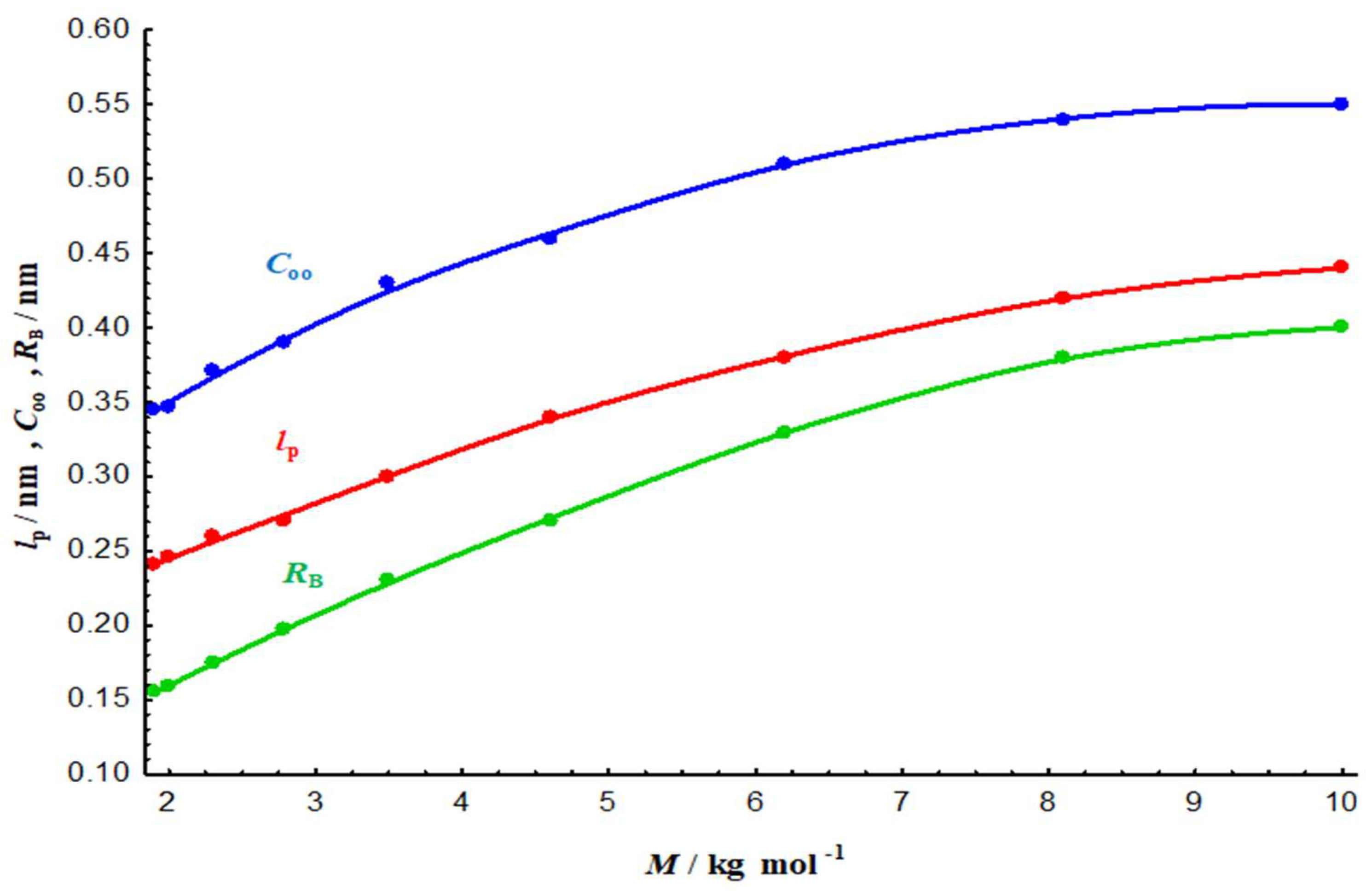
3.2.4. Ideal chain length parameters and chain flexibility
3.3. Conformational Consequences of Photo-Tailing
3.4. Overall Structural Interpretation
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, J.; Ahmed, H.H.; Hussein, A.M.; Kaur, M.; Jameel, M.K.; Kaur, H.; Tillaeva, U.; Al-Hussainy, A.F.; Sameer, H.N.; Hameed, H.G.; et al. Advances in polysaccharide-based materials for biomedical and pharmaceutical applications: A comprehensive review. Arch. Pharm. 2025, 358, e2400854. [Google Scholar] [CrossRef]
- Hamed, Y.S.; Hassan, K.R.; Salem, M.E.; Shen, M.; Wang, J.; Bu, T.; Cao, Y.; Xia, Q.; Youssef, K.M.; Yang, K. Gamma rays irradiated polysaccharides: A review of structure, physicochemical properties, biological activity alteration, and future food applications. Carbohydr. Polym. 2025, 123, 123326. [Google Scholar] [CrossRef]
- Elashhab, F.; Sheha, L.; Elzawi, N.; Youssef, A.E.A. Viscosity Analysis of Electron-Beam Degraded Gellan in Dilute Aqueous Solution. Physchem 2025, 5, 40. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Islam, M.; Hasan, M.K.; Nam, K.W. A comprehensive review of radiation-induced hydrogels: Synthesis, properties, and multidimensional applications. Gels 2024, 10, 381. [Google Scholar] [CrossRef]
- Sultana, R.; Kamihira, M. Multifaceted heparin: Diverse applications beyond anticoagulant therapy. Pharmaceuticals 2024, 17, 1362. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Guo, S.; Xu, J. Multifunctional applications and research advances of low-molecular-weight heparin. Front. Pharmacol. 2025, 16, 1585762. [Google Scholar] [CrossRef]
- Hogwood, J.; Mulloy, B.; Lever, R.; Gray, E.; Page, C.P. Pharmacology of heparin and related drugs: An update. Pharmacol. Rev. 2023, 75, 328–379. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Song, Y.; Zhao, Y.; Wang, N.; Wei, B.; Linhardt, R.J.; Dordick, J.S.; Zhang, F.; Wang, H. Quality control, safety assessment, and preparation approaches of low-molecular-weight heparin. Carbohydr. Polym. 2024, 339, 122216. [Google Scholar] [CrossRef] [PubMed]
- Tuaeva, N.O.; Trukhan, V.M.; Kardonskii, D.A.; et al. Preparation of low-molecular-weight heparins by radiation-induced destruction. Pharm. Chem. J. 2018, 52, 122–126. [Google Scholar] [CrossRef]
- Higashi, K.; Hosoyama, S.; Ohno, A.; Masuko, S.; Yang, B.; Sterner, E.; Wang, Z.; Linhardt, R.J.; Toida, T. Photochemical preparation of a novel low molecular weight heparin. Carbohydr. Polym. 2012, 87, 1737–1743. [Google Scholar] [CrossRef]
- Kulick, W.M.; Clasen, C. Viscosimetry of Polymers and Polyelectrolytes; Springer: Berlin/Heidelberg, Germany, 2004. [Google Scholar]
- Solomon, O.F.; Ciută, I.Z. Détermination de la viscosité intrinsèque de solutions de polymères par une simple détermination de la viscosité. J. Appl. Polym. Sci. 1962, 6, 683–686. [Google Scholar] [CrossRef]
- Mark, H. Überdie Entstehung und eigenschaften hoch polymer festkörper. In Der Festekörper; Sanger, R., Ed.; Hirzel: Leipzig, Germany, 1938; pp. 65–104. [Google Scholar]
- Houwink, R. Zusammenhang zwischen viscosimetrisch und osmotisch bestimmten Polymerisationsgraden bei Hochpolymeren. J. Prakt. Chem. 1940, 157, 15–18. [Google Scholar] [CrossRef]
- Bertini, S.; Bisio, A.; Torri, G.; Bensi, D.; Terbojevich, M. Molecular weight determination of heparin and dermatan sulfate by size exclusion chromatography with a triple detector array. Biomacromolecules 2005, 6, 168–173. [Google Scholar] [CrossRef]
- Sinko, P.J. Martin’s Physical Pharmacy and Pharmaceutical Sciences, 7th ed.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2017; pp. 412–415. [Google Scholar]
- Khalil, R.A. Direct Mathematical Models for Estimating the Shelf Life of Second- and Zero-Order Degradation Relationships of Food and Drugs. Iran. J. Math. Chem. 2023, 14, 161–169. [Google Scholar]
- Glassstone, S.; Laidler, K.J.; Eyring, H. The Theory of Rate Processes; McGraw-Hill: New York, NY, USA, 1941. [Google Scholar]
- Moore, W.R. Viscosities of dilute polymer solutions. Prog. Polym. Sci. 1967, 1, 1–43. [Google Scholar] [CrossRef]
- Teraoka, I. Polymer Solutions: An Introduction to Physical Properties; Wiley-Interscience: New York, NY, USA, 2002. [Google Scholar]
- Qian, J.W.; Wang, M.L.; Han, D.L.; Cheng, R.S. Eur. Polym. J. 2001, 37, 1403. [CrossRef]
- Calvini, P. The role of the Ekenstam equation on the kinetics of cellulose hydrolytic degradation. Cellulose 2012, 19, 313–318. [Google Scholar] [CrossRef]
- Emsley, A.M.; Stevens, G.C. Kinetics and Mechanisms of the low temperature degradation of cellulose. Cellulose 1994, 1, 26–56. [Google Scholar] [CrossRef]
- Kauzmann, W.; Eyring, H. J. Am. Chem. Soc. 1940, 62, 3113. [CrossRef]
- Seyrek, E.; Dubin, P.L.; Tribet, C.; Jasupaithum, A. Conformation of heparin studied with macromolecular hydrodynamic methods and X-ray scattering. Biomacromolecules 2003, 4, 273–280. [Google Scholar] [CrossRef]
- Beirne, J.; Mourier, P.; Rudd, T.R.; Guichard, O.; Bazin, R.; Guerrini, M.; Torri, G.; Yates, E.A. Characterisation of currently marketed heparin products: Analysis of molecular weight and heparinase-I digest patterns. Anal. Bioanal. Chem. 2011, 401, 2825–2832. [Google Scholar]
- Dobrynin, A.V.; Rubinstein, M. Theory of polyelectrolytes in solutions and at surfaces. Prog. Polym. Sci. 2005, 30, 1049–1118. [Google Scholar] [CrossRef]
- de Gennes, P.-G. Scaling Concepts in Polymer Physics; Cornell University Press: Ithaca, NY, USA, 1979. [Google Scholar]
- Yates, E.A.; Santini, F.; Guerrini, M.; Naggi, A.; Torri, G.; Casu, B. 1D and 2D NMR spectroscopy of low molecular weight heparins: A correlation and approach to the definition of structures. Carbohydr. Res. 2004, 339, 239–248. [Google Scholar]
- Papadopoulou, A.; Gani, E.N.; Katsifa, A.; Kyritsis, A.; Efthimiadou, E.K. Solution properties and chain flexibility of sodium heparin in aqueous media. J. Mol. Liq. 2016, 219, 247–254. [Google Scholar]
- Rubinstein, M.; Colby, R.H. Polymer Physics; Oxford University Press: New York, NY, USA, 2003. [Google Scholar]
- Hong, S.; et al. Photochemical preparation of a novel low molecular weight heparin. Carbohydr. Polym. 2012, 87, 1181–1185. [Google Scholar] [CrossRef]
- Zatsepina, O.; et al. Heparin in Acid and Alkaline Environments—A Study of the Correlations between Hydrodynamic Properties and Desulphation. Polysaccharides 2023, 4, 88–98. [Google Scholar] [CrossRef]
| Model / Parameter | Equation | Eq. No. |
|---|---|---|
| Self-Avoiding Walk Model (SAWM) | ||
| Radius of Gyration | , | (9) |
| Critical Concentration | (10) | |
| Expansion Ratio | (11) | |
| Mark-Houwink Scaling | , = 3 – 1 | (12) |
| Scaling Law | (13) | |
| Scaling Law | (14) | |
| Random Walk Model (RWM): Theta -Conditions | ||
| Unperturbed Dimensions | , | (15) |
| Flory Characteristic Ratio | , cm | (16) |
| Persistence Length | (17) | |
| Theoretical Viscosity | , | (18) |
| Sample | |||||
|---|---|---|---|---|---|
| NM | 12.76 | 8.80 | 3.75 | -240.50 | |
| SM | 4.00 | 8.42 | 1.66 | -215.98 | |
| USM | 2.12 | 7.83 | 1.10 | -200.10 |
| Sample | ||||||
|---|---|---|---|---|---|---|
| NM | 12.76 | 0.77 | 0.44 | 1.19 | ||
| SM | 4.00 | 0.40 | 0.23 | 1.19 | ||
| USM | 2.12 | 0.26 | 0.14 | 1.19 |
| Sample | |||||
|---|---|---|---|---|---|
| NM | 12.76 | 5.92 | 10.18 | ||
| SM | 4.00 | 14.53 | 11.52 | ||
| USM | 2.12 | 30.80 | 12.5 |
| Coefficient | ||||
|---|---|---|---|---|
| Value | 1.30 | - 0.86 | - 0.12 |
| Sample | |||||
|---|---|---|---|---|---|
| NM | 12.76 | 0.4 | 0.55 | 0.44 | |
| SM | 4.00 | 0.23 | 0.43 | 0.30 | |
| USM | 2.12 | 0.16 | 0.34 | 0.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




