Submitted:
02 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Serum Samples
2.2. Chemicals and Reagents
2.3. Instrumentation and Analytical Conditions
2.4. Preparation of Calibration Standards and Quality Control Samples
2.5. Sample Preparation
2.6. Method Validation
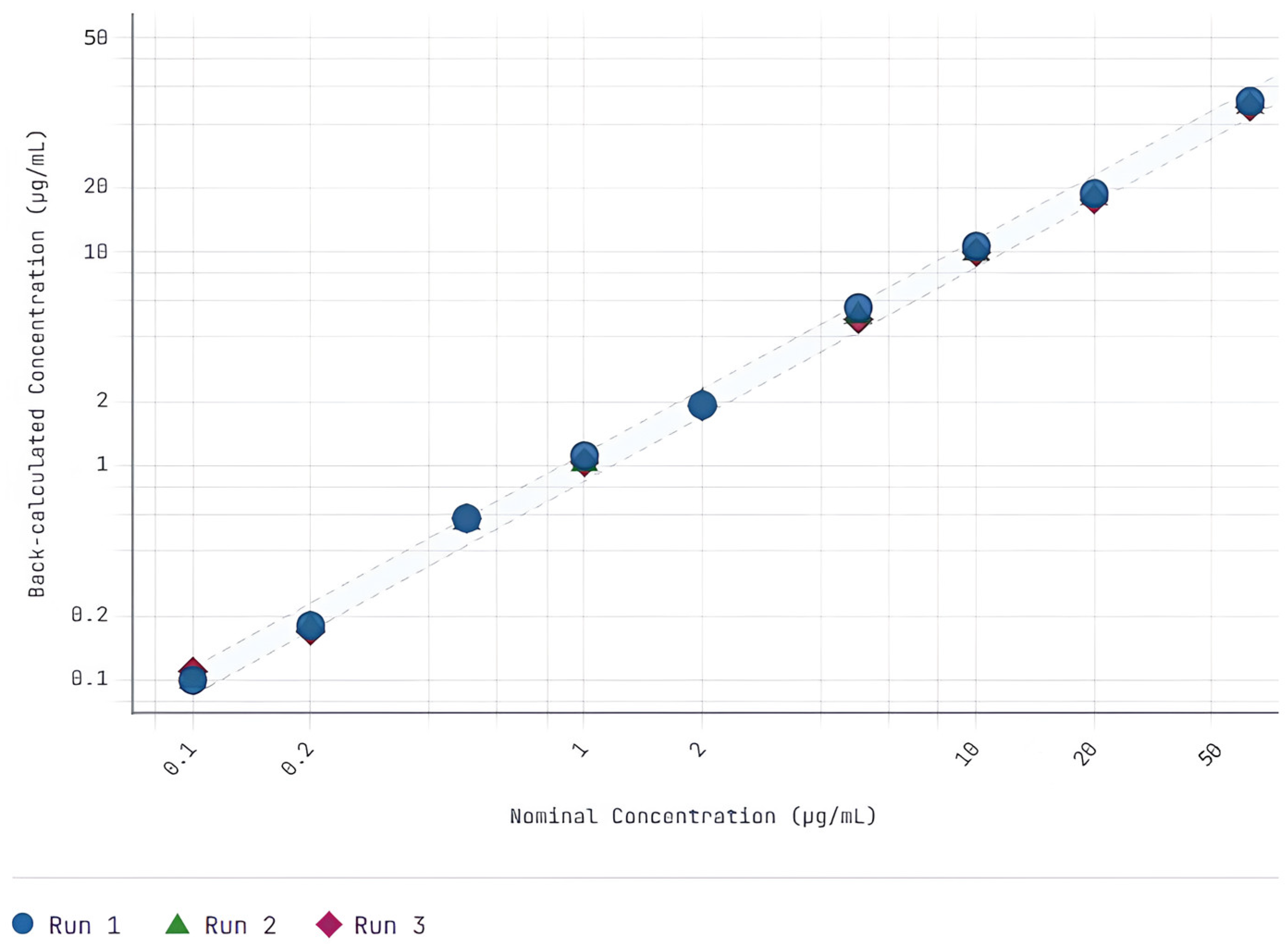
2.6.1. Calibration Curve Performance and Lower Limit of Quantification
2.6.2. Accuracy and Precision
2.6.3. Matrix Effect and Extraction Recovery
2.6.4. Carryover and Dilution Integrity
2.6.5. Selectivity
2.6.6. Stability Studies
2.7. Pharmaceutical Quality Control Application
2.8. Statistical Analysis
3. Results
3.1. Method Validation
3.2. Stability, Processed Sample Stability and Carryover
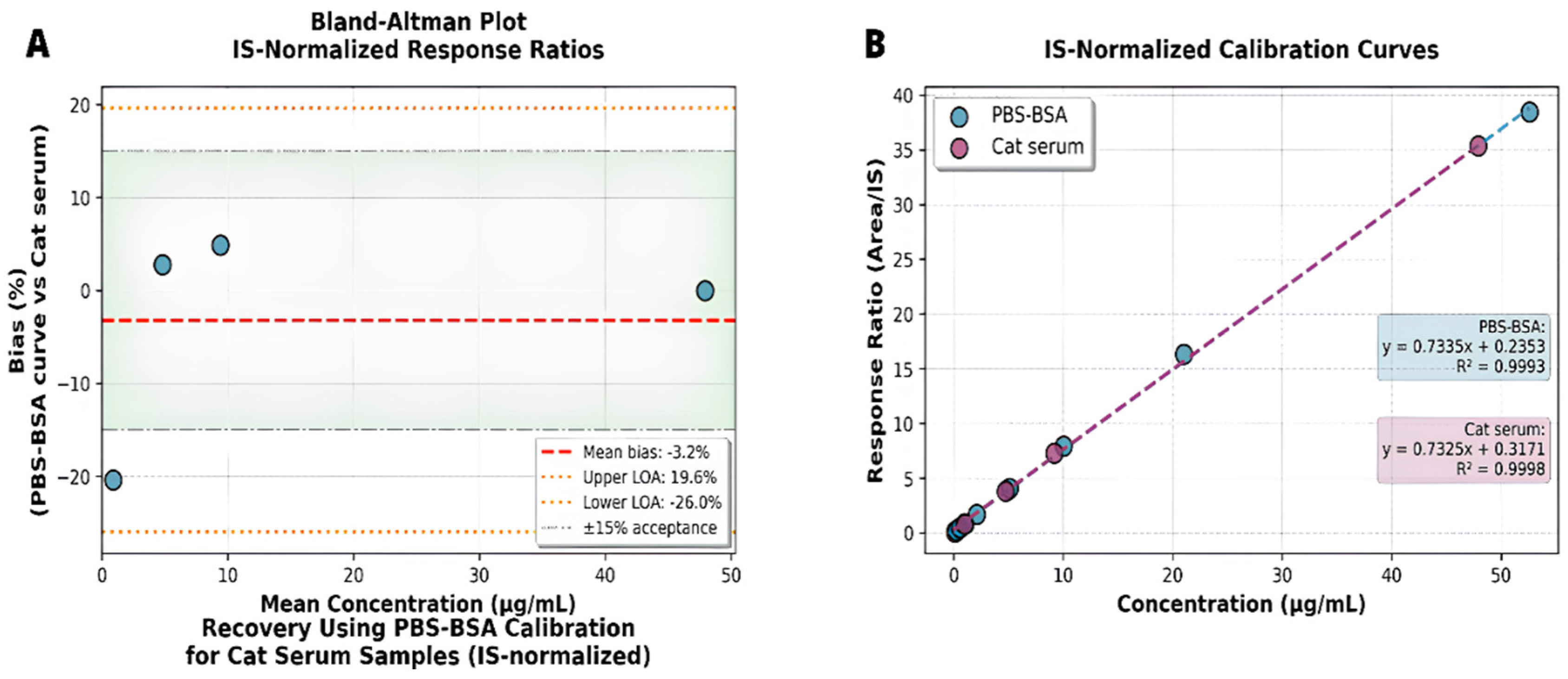
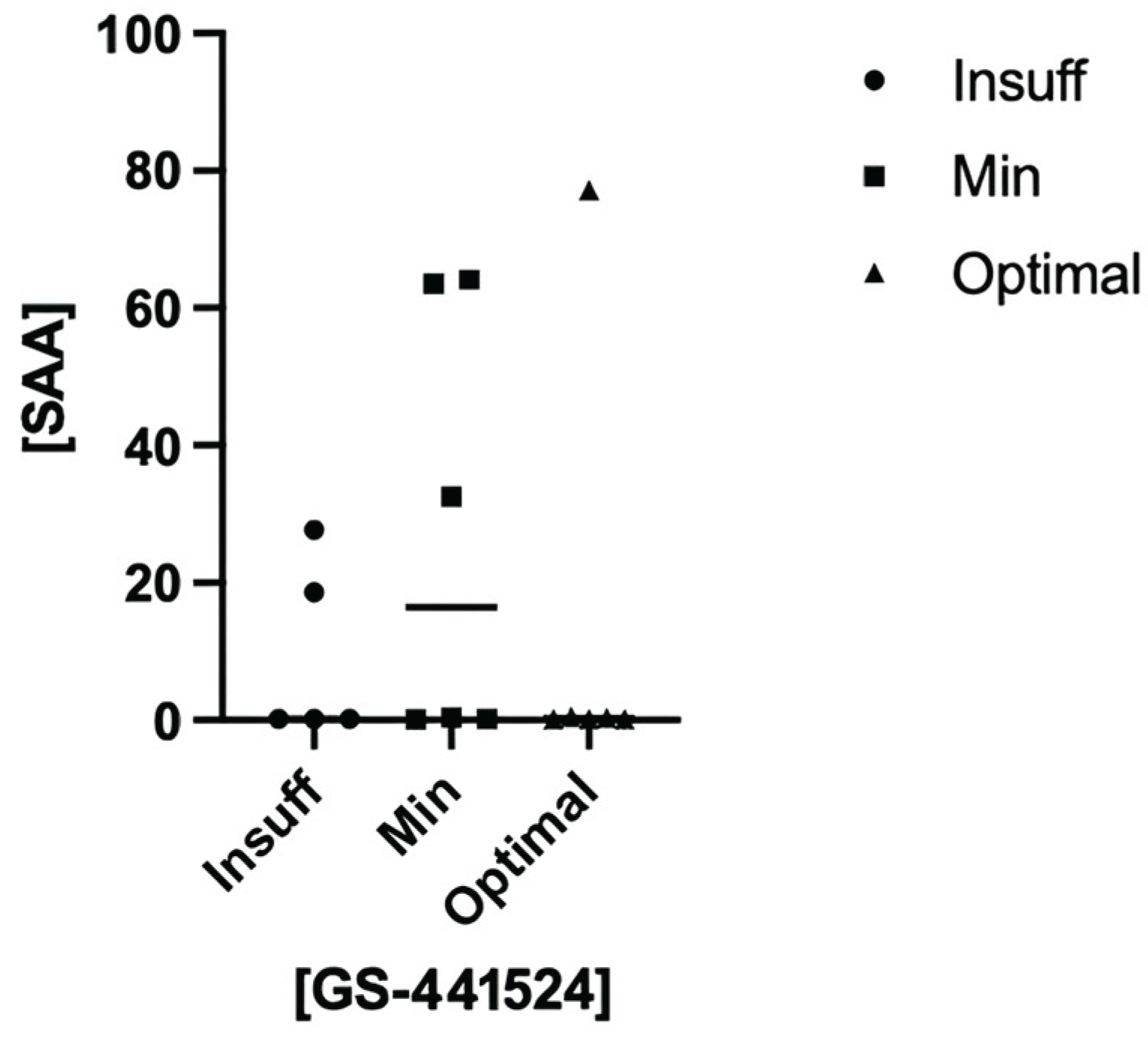
3.3. Matrix Equivalence and Surrogate Matrix Validation
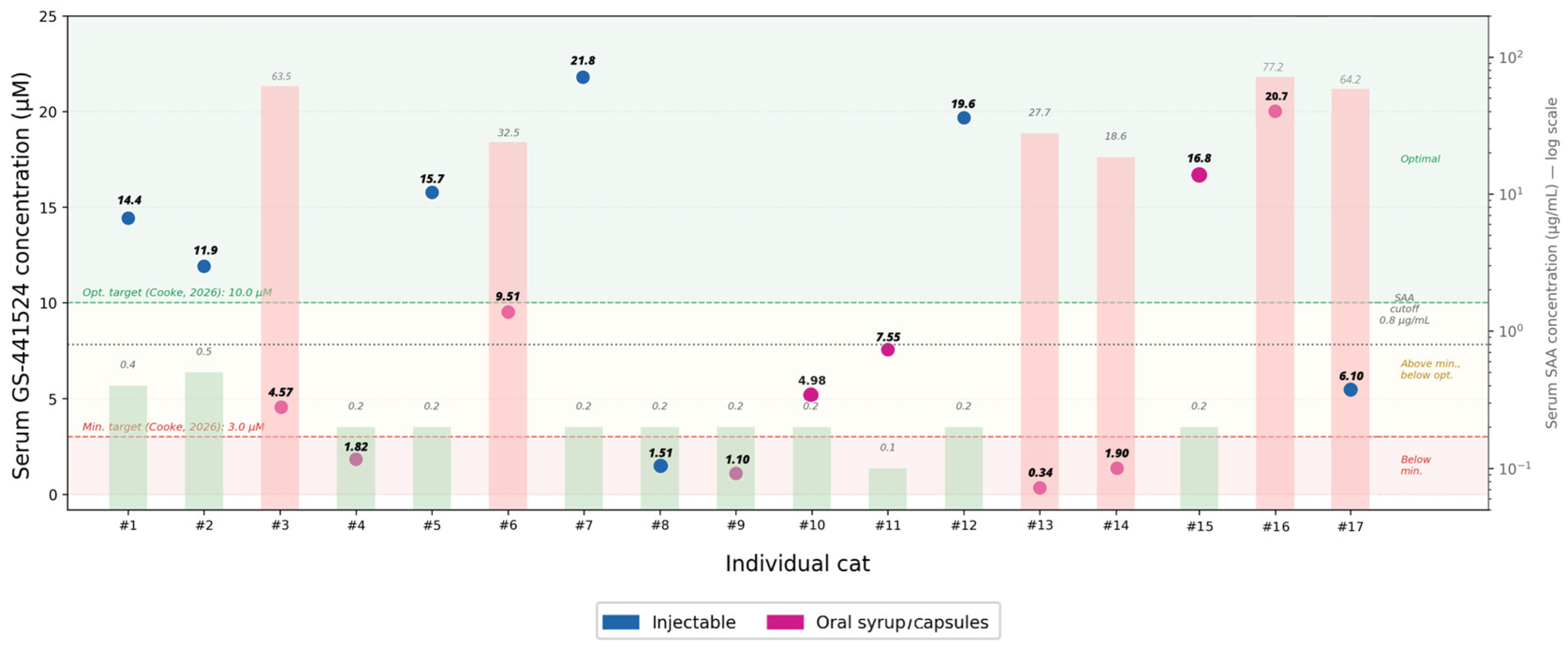
3.4. Quality Control of Compounded GS-441524 Formulations from Commercial Sources
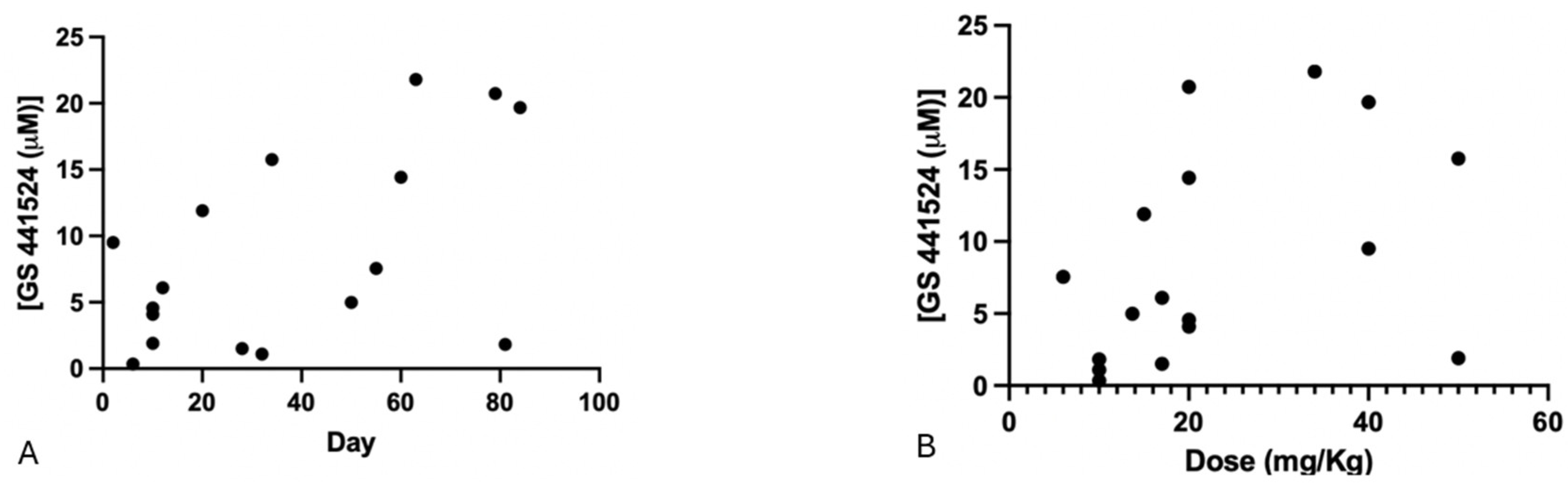
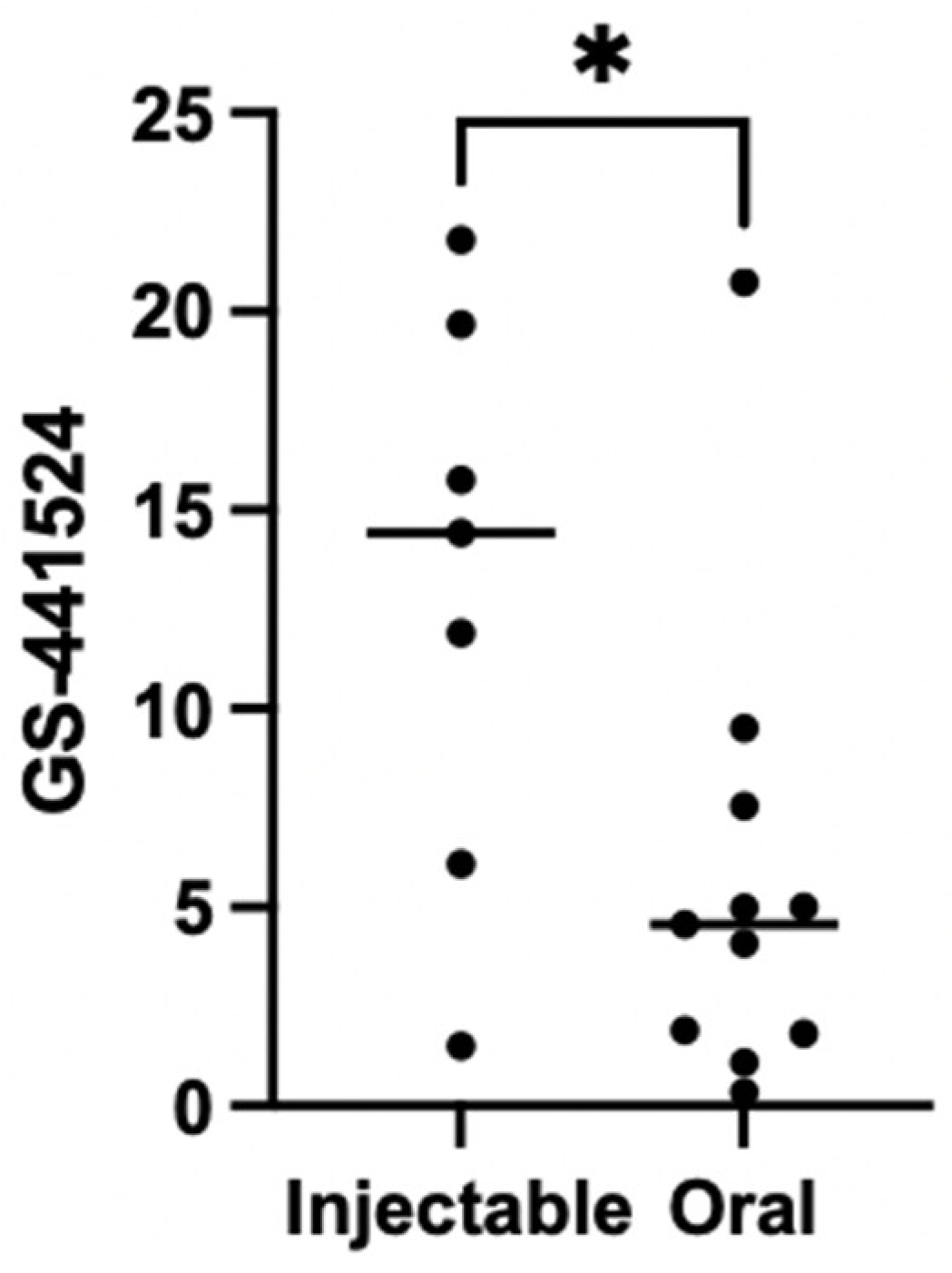
3.5. Therapeutical Drug Monitoring
4. Discussion
Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kipar, A.; Meli, M.L. Feline Infectious Peritonitis: Still an Enigma? Vet. Pathol. 2014, 51, 505–526. [Google Scholar] [CrossRef]
- Rottier, P.J.M.; Nakamura, K.; Schellen, P.; Volders, H.; Haijema, B.J. Acquisition of Macrophage Tropism during the Pathogenesis of Feline Infectious Peritonitis Is Determined by Mutations in the Feline Coronavirus Spike Protein. J. Virol. 2005, 79, 14122–14130. [Google Scholar] [CrossRef]
- Pedersen, N.C. A Review of Feline Infectious Peritonitis Virus Infection: 1963–2008. J. Feline Med. Surg. 2009, 11, 225–258. [Google Scholar] [CrossRef] [PubMed]
- Cho, A.; Saunders, O.L.; Butler, T.; Zhang, L.; Xu, J.; Vela, J.E.; Feng, J.Y.; Ray, A.S.; Kim, C.U. Synthesis and Antiviral Activity of a Series of 1’-Substituted 4-Aza-7,9-Dideazaadenosine C-Nucleosides. Bioorg Med. Chem. Lett. 2012, 22, 2705–2707. [Google Scholar] [CrossRef]
- Siegel, D.; Hui, H.C.; Doerffler, E.; Clarke, M.O.; Chun, K.; Zhang, L.; Neville, S.; Carra, E.; Lew, W.; Ross, B.; et al. Discovery and Synthesis of a Phosphoramidate Prodrug of a Pyrrolo[2,1-f][Triazin-4-Amino] Adenine C-Nucleoside (GS-5734) for the Treatment of Ebola and Emerging Viruses. J. Med. Chem. 2017, 60, 1648–1661. [Google Scholar] [CrossRef]
- Murphy, B.G.; Perron, M.; Murakami, E.; Bauer, K.; Park, Y.; Eckstrand, C.; Liepnieks, M.; Pedersen, N.C. The Nucleoside Analog GS-441524 Strongly Inhibits Feline Infectious Peritonitis (FIP) Virus in Tissue Culture and Experimental Cat Infection Studies. Vet. Microbiol. 2018, 219, 226–233. [Google Scholar] [CrossRef]
- Gokalsing, E.; Ferrolho, J.; Gibson, M.S.; Vilhena, H.; Anastácio, S. Efficacy of GS-441524 for Feline Infectious Peritonitis: A Systematic Review (2018–2024). Pathogens 2025, 14, 717. [Google Scholar] [CrossRef] [PubMed]
- Thayer, V.; Gogolski, S.; Felten, S.; Hartmann, K.; Kennedy, M.; Olah, G.A. 2022 AAFP/EveryCat Feline Infectious Peritonitis Diagnosis Guidelines. J. Feline Med. Surg. 2022, 24, 905–933. [Google Scholar] [CrossRef]
- Tasker, S.; Spiri, A.M.; Hartmann, K.; Addie, D.D.; Belák, S.; Bergmann, M.; Egberink, H.; Frymus, T.; Hofmann-Lehmann, R.; Marsilio, F.; et al. Update on Treatment of Feline Infectious Peritonitis: European Advisory Board on Cat Diseases (ABCD) Guidelines. Viruses 2026, 18, 452. [Google Scholar] [CrossRef]
- Taylor, S.S.; Coggins, S.; Barker, E.N.; Gunn-Moore, D.; Jeevaratnam, K.; Norris, J.M.; Hughes, D.; Stacey, E.; MacFarlane, L.; O’Brien, C.; et al. Retrospective Study and Outcome of 307 Cats with Feline Infectious Peritonitis Treated with Legally Sourced Veterinary Compounded Preparations of Remdesivir and GS-441524 (2020-2022). J. Feline Med. Surg. 2023, 25, 1098612X231194460. [Google Scholar] [CrossRef] [PubMed]
- Kimble, B.; Coggins, S.J.; Norris, J.M.; Thompson, M.F.; Govendir, M. Quantification of GS-441524 Concentration in Feline Plasma Using High Performance Liquid Chromatography with Fluorescence Detection. Vet. Q. 2023, 43, 1–9. [Google Scholar] [CrossRef]
- Coggins, S.J.; Govendir, M.; Norris, J.M.; Malik, R.; Hall, E.J.; Thompson, M.F.; Kimble, B. Pharmacokinetics of GS -441524 Following Intravenous Remdesivir in Six Cats and Results of Therapeutic Drug Monitoring during Treatment of Feline Infectious Peritonitis: 22 Cases (2021–2024). J. Small Anim. Pract. 2025, 66, 495–506. [Google Scholar] [CrossRef]
- Cooke, S.W.; Hammond, R.; Gunn-Moore, D.A. Therapeutic Drug Monitoring of GS-441524 in Cats with Feline Infectious Peritonitis: Pharmacokinetic Variability and Implications for Dose Optimization. Pathogens 2026, 15, 291. [Google Scholar] [CrossRef]
- Kamiyoshi, T.; Kamiyoshi, N.; Jintake, C. High-dose Induction Therapy and Treatment Termination Criteria for Feline Infectious Peritonitis with Remdesivir, GS -441524 and Adjunctive Mefloquine: 46 Cases (2023). J. Small Anim. Pract. 2025, 66, 617–626. [Google Scholar] [CrossRef]
- Fang, Z.; Zhang, H.; Guo, J.; Guo, J. Overview of Therapeutic Drug Monitoring and Clinical Practice. Talanta 2024, 266, 124996. [Google Scholar] [CrossRef] [PubMed]
- Bardhi, A.; Lanci, A.; Mannini, A.; Castagnetti, C.; Barbarossa, A. A Laboratory Protocol for Routine Therapeutic Drug Monitoring of Beta-Lactams Antimicrobials in Horses and Dogs. Antibiotics 2025, 14, 390. [Google Scholar] [CrossRef]
- Adaway, J.E.; Keevil, B.G. Therapeutic Drug Monitoring and LC–MS/MS. J. Chromatogr. B 2012, 883–884, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Veyrat, S.; Hopfgartner, G. Mass Spectrometry Based High-Throughput Bioanalysis of Low Molecular Weight Compounds: Are We Ready to Support Personalized Medicine? Anal. Bioanal. Chem. 2022, 414, 181–192. [Google Scholar] [CrossRef]
- ICH Guideline M10 on Bioanalytical Method Validation and Study Sample Analysis; International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH): Geneva, 2022.
- Gu, H.; Liu, G.; Wang, J.; Aubry, A.-F.; Arnold, M.E. Selecting the Correct Weighting Factors for Linear and Quadratic Calibration Curves with Least-Squares Regression Algorithm in Bioanalytical LC-MS/MS Assays and Impacts of Using Incorrect Weighting Factors on Curve Stability, Data Quality, and Assay Performance. Anal. Chem. 2014, 86, 8959–8966. [Google Scholar] [CrossRef]
- Harris, C.R.; Millman, K.J.; Van Der Walt, S.J.; Gommers, R.; Virtanen, P.; Cournapeau, D.; Wieser, E.; Taylor, J.; Berg, S.; Smith, N.J.; et al. Array Programming with NumPy. Nature 2020, 585, 357–362. [Google Scholar] [CrossRef] [PubMed]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental Algorithms for Scientific Computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef]
- Hunter, J.D. Matplotlib: A 2D Graphics Environment. Comput. Sci. Eng. 2007, 9, 90–95. [Google Scholar] [CrossRef]
- Matuszewski, B.K.; Constanzer, M.L.; Chavez-Eng, C.M. Strategies for the Assessment of Matrix Effect in Quantitative Bioanalytical Methods Based on HPLC−MS/MS. Anal. Chem. 2003, 75, 3019–3030. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, L.; McDougall, S.; Gnoth, M.J.; Mascher, D.; Ferrari, L.; Wheller, R.; Hawthorne, H.; Jansat, J.-M.; Faber, J.; Huber, P.; et al. The Use of Surrogate Matrices in Bioanalytical Preclinical Safety Testing Using Chromatographic Methods: A Recommendation from the European Bioanalysis Forum. Bioanalysis 2024, 16, 1199–1202. [Google Scholar] [CrossRef] [PubMed]
- Grimm, H.; Biller-Andorno, N.; Buch, T.; Dahlhoff, M.; Davies, G.; Cederroth, C.R.; Maissen, O.; Lukas, W.; Passini, E.; Törnqvist, E.; et al. Advancing the 3Rs: Innovation, Implementation, Ethics and Society. Front. Vet. Sci. 2023, 10, 1185706. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.; Awaiye, K. Use of Internal Standards in LC-MS Bioanalysis. In Handbook of LC-MS Bioanalysis; Li, W., Zhang, J., Tse, F.L.S., Eds.; Wiley, 2013; pp. 217–227. ISBN 978-1-118-15924-8. [Google Scholar]
- Avedissian, S.N.; Mu, Y.; McCarthy, C.; Bosch, R.J.; Spudich, S.; Gandhi, R.T.; McMahon, D.K.; Eron, J.J.; Mellors, J.W.; Liu, J.; et al. Pharmacokinetic Approaches to Standardize Antiviral Exposure in Cerebrospinal Fluid. Pharmacotherapy 2025, 45, 251–263. [Google Scholar] [CrossRef]
| Spike Level | Parameter | Intra-Assay | Inter-Assay | ||
|---|---|---|---|---|---|
| Day 1 | Day 2 | Day 3 | |||
| LLOQ-QC (0.1 µg/mL) |
Mean (µg/mL) | 0.09 | 0.10 | 0.10 | 0.10 |
| CV (%) | 10.9 | 9.82 | 0.00 | 7.00 | |
| Bias (%) | -12.5 | -2.50 | 0.01 | -5.00 | |
| Accuracy (%) | 87.5 | 97.5 | 100.0 | 95.0 | |
| LQC (0.3 µg/mL) |
Mean (µg/mL) | 0.29 | 0.31 | 0.30 | 0.30 |
| CV (%) | 3.04 | 3.23 | 2.94 | 2.70 | |
| Bias (%) | -2.00 | 3.30 | 1.30 | 0.90 | |
| Accuracy (%) | 98.0 | 103 | 101 | 101 | |
| MQC (25.0 µg/mL) |
Mean (µg/mL) | 22.9 | 25.1 | 25.1 | 24.4 |
| CV (%) | 1.82 | 1.49 | 0.55 | 5.30 | |
| Bias (%) | -8.50 | 0.40 | 0.40 | -2.60 | |
| Accuracy (%) | 91.5 | 100 | 100 | 97.4 | |
| HQC (50.0 µg/mL) |
Mean (µg/mL) | 47.1 | 53.9 | 47.1 | 49.4 |
| CV (%) | 3.55 | 2.05 | 3.55 | 8.10 | |
| Bias (%) | -5.90 | 7.90 | -5.90 | -1.30 | |
| Accuracy (%) | 94.1 | 107.9 | 94.1 | 98.7 | |
| Timepoint/ Condition |
LQC | MQC | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | % resp. Relative to T0 | Acceptability | Mean | SD | % resp. Relative to T0 | Acceptability | |
| T0 | 0.29 | 0.01 | — | — | 26.8 | 0.11 | — | — |
| Benchtop day 1 | 0.28 | 0.01 | -5.17% | ✓ | 27.1 | 0.04 | 0.95% | ✓ |
| Benchtop day 3 | 0.30 | 0.01 | 1.72% | ✓ | 28.1 | 1.44 | 4.72% | ✓ |
| Benchtop day 7 | 0.27 | 0.01 | -8.62% | ✓ | 25.8 | 0.42 | -3.88% | ✓ |
| Benchtop day 15 | 0.22 | 0.03 | -24.1% | ✗ | 19.1 | 0.63 | -28.6% | ✗ |
| Refrigerated day 1 | 0.29 | 0.01 | 0.00% | ✓ | 26.8 | 0.11 | 0.11% | ✓ |
| Refrigerated day 3 | 0.31 | 0.01 | 6.90% | ✓ | 29.0 | 1.03 | 8.29% | ✓ |
| Refrigerated day 7 | 0.26 | 0.01 | -10.3% | ✓ | 27.2 | 0.62 | 1.44% | ✓ |
| Refrigerated day 15 | 0.24 | 0.01 | -17.2% | ✗ | 24.4 | 0.24 | 10.9% | ✓ |
| Frozen day 1 | 0.28 | 0.01 | -3.45% | ✓ | 27.4 | 0.05 | 2.35% | ✓ |
| Frozen day 3 | 0.30 | 0.01 | 1.72% | ✓ | 28.0 | 0.25 | 4.39% | ✓ |
| Frozen day 7 | 0.29 | 0.01 | -1.72% | ✓ | 27.4 | 0.47 | 2.20% | ✓ |
| Frozen day 15 | 0.27 | 0.01 | -6.90% | ✓ | 24.5 | 0.18 | -8.45% | ✓ |
| Thermal stress day 1 | 0.29 | 0.01 | -1.72% | ✓ | 25.5 | 0.40 | -4.93% | ✓ |
| Thermal stress day 3 | 0.28 | 0.01 | -3.45% | ✓ | 27.1 | 0.33 | 1.19% | ✓ |
| Thermal stress day 7 | 0.22 | 0.02 | -25.9% | ✗ | 22.9 | 0.53 | -14.7% | ✓ |
| Thermal stress day 15 | 0.16 | 0.01 | -46.6% | ✗ | 15.2 | 0.03 | -43.4% | ✗ |
| Timepoint/ Condition |
LQC | MQC | ||||||
|---|---|---|---|---|---|---|---|---|
| Mean | SD | % resp. Relative to T0 |
Acceptability | Mean | SD | % resp. Relative to T0 |
Acceptability | |
| T0 | 0.29 | 0.00 | — | — | 27.4 | 0.34 | — | — |
| Thaw 1 | 0.28 | 0.00 | -3.40 | ✓ | 28.2 | 0.53 | 3.00 | ✓ |
| Thaw 2 | 0.27 | 0.00 | -6.90 | ✓ | 28.4 | 0.31 | 3.50 | ✓ |
| Thaw 3 | 0.28 | 0.00 | -3.40 | ✓ | 26.2 | 0.14 | -4.40 | ✓ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




