Submitted:
30 April 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
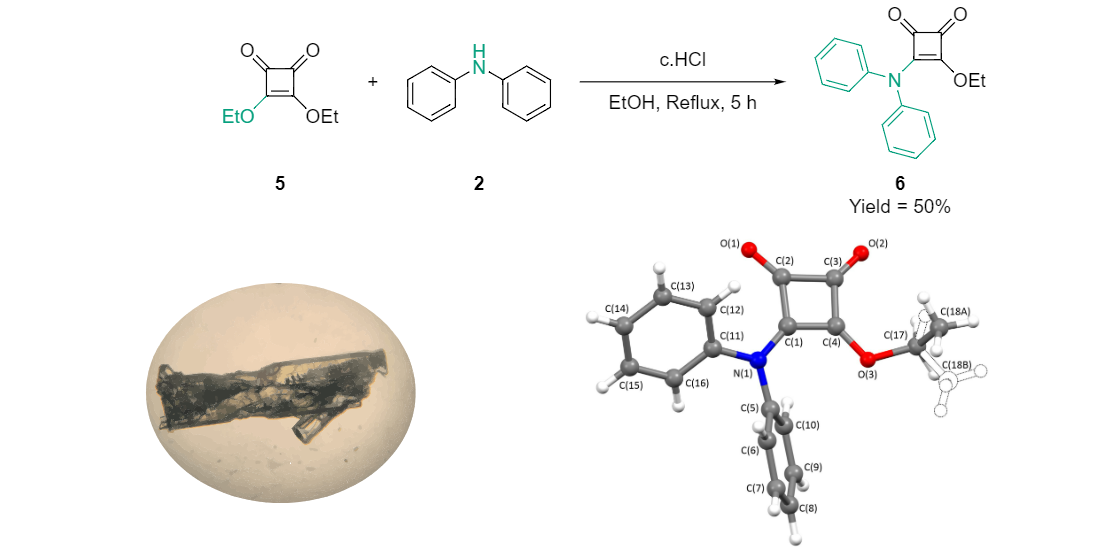
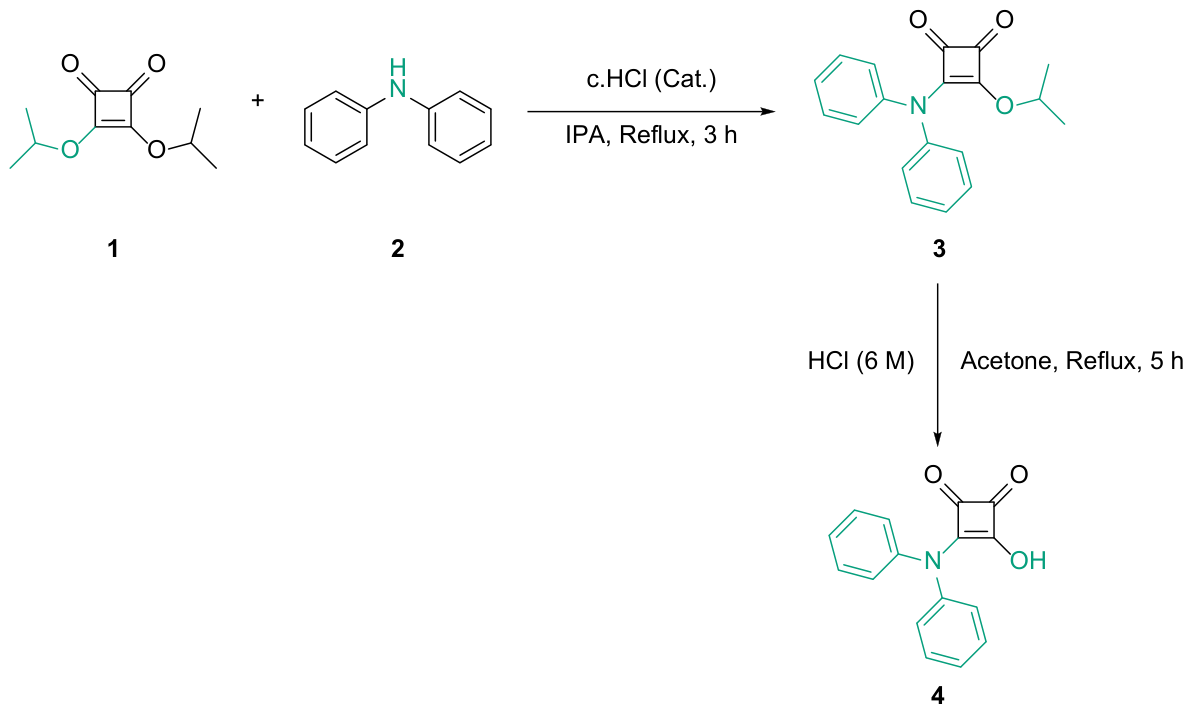
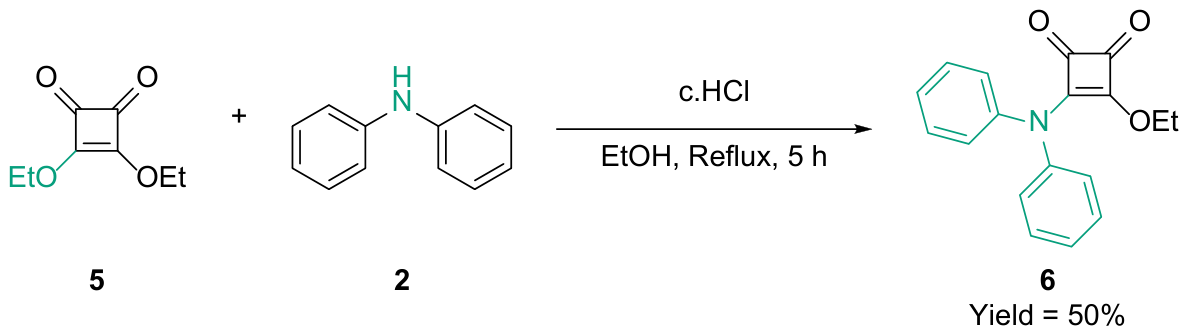
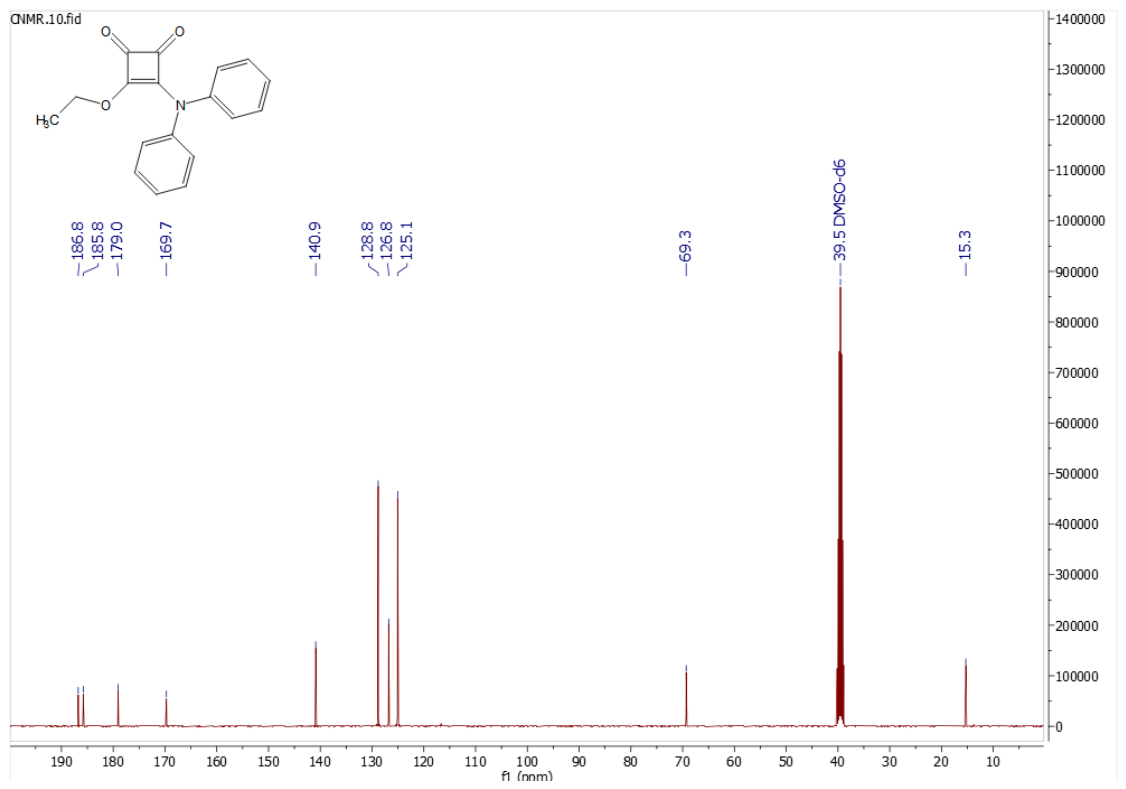
2.1. Chemical Synthesis
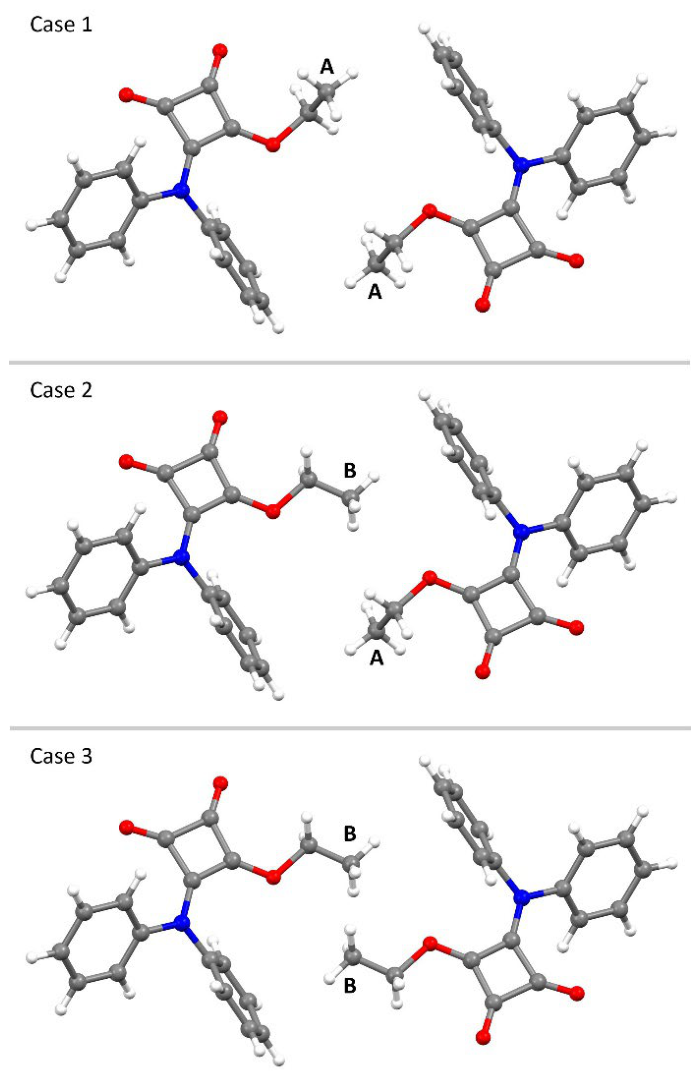
2.3. X-Ray Crystallography
3. Materials and Methods
3.1. Synthesis; General Information
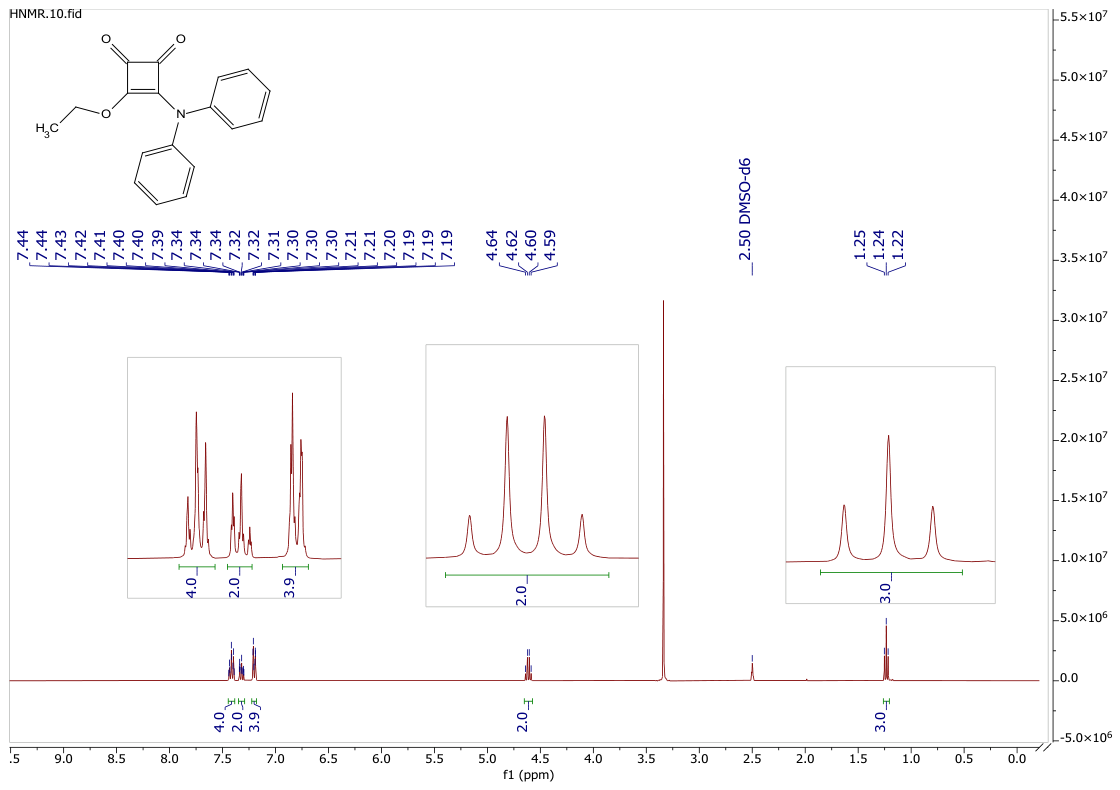
3.1. Synthesis of 3-(diphenylamino)-4-ethoxycyclobut-3-ene-1,2-dione (6)
3.3. X-Ray Crystallography of 3-(diphenylamino)-4-ethoxycyclobut-3-ene-1,2-dione (6)
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Liebeskind, L.S. Cyclobutenediones as Precursors to Quinones and Cyclopentenones. Tetrahedron 1989, 45, 3053–3060. [CrossRef]
- Liu, Y.; Lam, A.H.W.; Fowler, F.W.; Lauher, J.W. The Squaramides. A New Family of Host Molecules for Crystal Engineering. Molecular Crystals and Liquid Crystals 2002, 389, 39–46. [CrossRef]
- Storer, R.I.; Aciro, C.; Jones, L.H. Squaramides: Physical Properties, Synthesis and Applications. Chem. Soc. Rev. 2011, 40, 2330–2346. [CrossRef]
- Cohen, S.; Cohen, S.G.; Cohen, S.; Lacher, J.R.; Park, J.D.; Am Chem Soc, J.; West, R.; Niu, H.Y.; Powell, D.L.; Smutny, E.J.; et al. Preparation and Reactions of Derivatives of Squaric Acid. Alkoxy-, Hydroxy-, and Aminocyclobutenediones1. J. Am. Chem. Soc. 1966, 88, 1533–1536. [CrossRef]
- Kitson, S. Squaryl Molecular Metaphors – Application to Rational Drug Design and Imaging Agents. J. Diagn. Imaging Ther. 2017, 4, 35–75. [CrossRef]
- Liu, H.; Tomooka, C.S.; Moore, H.W. An Efficient General Synthesis of Squarate Esters. Synth. Commun. 1997, 27, 2177–2180. [CrossRef]
- Schmidt, A.H. Reaktionen von Quadratsäure Und Quadratsäure-Derivaten. Synthesis (Germany) 1980, 1980, 961–994.
- Green, B.R.; Neuse, E.W. Synthesis of 1,2-Diphenylcyclobutene-3,4-Dione. Synthesis (Stuttg). 1974, 1974, 46–47.
- Tietze, L.F.; Arlt, M.; Beller, M.; Gl üsenkamp, K. -H; Jähde, E.; Rajewsky, M.F. Anticancer Agents, 15. Squaric Acid Diethyl Ester: A New Coupling Reagent for the Formation of Drug Biopolymer Conjugates. Synthesis of Squaric Acid Ester Amides and Diamides. Chem. Ber. 1991, 124, 1215–1221. [CrossRef]
- Pirrung, M.C.; Han, H.; Chen, J. O-Alkyl Hydroxamates as Metaphors of Enzyme-Bound Enolate Intermediates in Hydroxy Acid Dehydrogenases. Inhibitors of Isopropylmalate Dehydrogenase, Isocitrate Dehydrogenase, and Tartrate Dehydrogenase 1; 1996;
- Lassalas, P.; Gay, B.; Lasfargeas, C.; James, M.J.; Tran, V.; Vijayendran, K.G.; Brunden, K.R.; Kozlowski, M.C.; Thomas, C.J.; Smith, A.B.; et al. Structure Property Relationships of Carboxylic Acid Isosteres. J. Med. Chem. 2016, 59, 3183–3203. [CrossRef]
- Long, N.; Le Gresley, A.; Solomonsz, A.; Wozniak, A.; Brough, S.; Wren, S.P. Synthesis of Squaric Acid Monoamides as Building Blocks for Drug Discovery. SynOpen 2023, 07, 401–407. [CrossRef]
- Long, N.; Le Gresley, A.; Wozniak, A.; Brough, S.; Wren, S.P. Synthesis and Evaluation of Druglike Parameters via in Silico Techniques for a Series of Heterocyclic Monosquarate-Amide Derivatives as Potential Carboxylic Acid Bioisosteres. Bioorg. Med. Chem. 2024, 98, 117565. [CrossRef]
- Long, N.; Meyer-Almes, F.-J.; Kopranovic, A.; Wren, S.P. Fair and Square: Design, Synthesis and Biological Evaluations of Squaric Acid Derivatives as Novel HDAC8 Inhibitors. ChemMedChem 2026, 21, e70270. [CrossRef]
- Wang, S.; Hall, L.; Diev, V. V.; Haiges, R.; Wei, G.; Xiao, X.; Djurovich, P.I.; Forrest, S.R.; Thompson, M.E. N,N-Di Aryl Anilinosquaraines and Their Application to Organic Photovoltaics. Chemistry of Materials 2011, 23, 4789–4798. [CrossRef]
- Alleyne, B.D.; St. Bernard, L.; Jaggernauth, H.; Hall, L.A.; Baxter, I.; White, A.J.P.; Williams, D.J. Lanthanide Complexes of 3-Diphenylamino-4-Hydroxycyclobut-3-Ene-1,2-Dione (Diphenylaminosquarate). Inorg. Chem. 1999, 38, 3774–3778. [CrossRef]
- Goh, T.; Huang, J.S.; Bielinski, E.A.; Thompson, B.A.; Tomasulo, S.; Lee, M.L.; Sfeir, M.Y.; Hazari, N.; Taylor, A.D. Coevaporated Bisquaraine Inverted Solar Cells: Enhancement Due to Energy Transfer and Open Circuit Voltage Control. ACS Photonics 2015, 2, 86–95. [CrossRef]
- Yang, L.; Zhu, Y.; Wu, J.; Hu, B.; Pang, Z.; Lu, Z.; Zhao, S.; Huang, Y. Squaraine Dyes Containing Diphenylamine Group: Effects of Different Type Structures on Material Properties and Organic Photovoltaic Performances. Dyes and Pigments 2019, 171. [CrossRef]
- Williams, A.R.; Alleyne, B.D.; Hall, L.A.; White, A.J.P.; Williams, D.J.; Thompson, L.K. Synthesis and Structures of Polymeric Mn, Co, Cu, and Zn Complexes of 3-Diphenylamino-4-Hydroxycyclobut-3-Ene-1,2-Dione (Diphenylaminosquarate) and of the Salt [Ni(H2O)6][(C6H5)2NC4O3]2·2H2O. Inorg. Chem. 2000, 39, 5265–5270. [CrossRef]
- Sheldrick, G.M. SHELXT - Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. A Found. Adv. 2015, 71, 3–8. [CrossRef]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. C Struct. Chem. 2015, 71, 3–8. [CrossRef]
- MacRae, C.F.; Sovago, I.; Cottrell, S.J.; Galek, P.T.A.; McCabe, P.; Pidcock, E.; Platings, M.; Shields, G.P.; Stevens, J.S.; Towler, M.; et al. Mercury 4.0: From Visualization to Analysis, Design and Prediction. urn:issn:1600-5767 2020, 53, 226–235. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



