Submitted:
01 May 2026
Posted:
05 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Anthropometric and Body Composition Measurements
2.3. Biochemical Analyses
- Bone alkaline phosphatase activity was measured using the BAP EIA kit (Quidel, Athens, OH, USA), with detection limit of 0.7 U/L, and intra- and inter-assay coefficients of variation (CVs) below 5.8% and 7.6%, respectively.
- Osteocalcin and C-terminal telopeptide of type I collagen were assessed using N-MID Osteocalcin ELISA kits and Serum CrossLaps (CTX-I) ELISA kit (IDS, Bolton, UK). The limit of detection for OC was 0.5 ng/mL, intra- and inter-assay CVs were <2.2% and <5.1% and for CTX-I was 0.02 ng/mL, <3.0% and <10.9%, respectively.
- Carboxylated and undercarboxylated osteocalcin levels were measured using ELISA kits from Takara Bio Inc. (Shiga, Japan), with intra- and inter-assay CVs below 2.4% and 4.8% for Gla-OC and <6.7% and 9.9% for Glu-OC, respectively. The limit of detection was 0.25 ng/mL for both forms of osteocalcin.
- Osteoprotegerin was determined using kits from DRG Diagnostics (Marburg, Germany) with a limit of detection of 0.03 pmol/L, intra-assay CV< 4.9% and inter-assay CV <9.0%.
- Soluble receptor activator of nuclear factor kappa-B ligand were measured using the Human sRANKL ELISA kit from SunRed Biotechnology (Shanghai, China). The detection limit was 1.56 pg/mL; intra- and inter-assay CVs were <9% and <11%, respectively.
- Sclerostin was measured using the Sclerostin HS ELISA kit from Teco Medical Group (Sissach, Switzerland) with intra- and inter-assay CVs less than 4.8% and 8.2%, respectively, and a detection limit of 0.006 ng/mL.
- Leptin levels were determined using ELISA kits from DRG Diagnostics (Marburg, Germany) with detection limit of 0.7 ng/mL, and intra- and inter-assay CVs below 5.9% and 8.6%, respectively.
- Total adiponectin and high-molecular-weight adiponectin were measured using ELISA kits from ALPCO Diagnostics (Salem, NH, USA). The limit of quantitation was 0.019 ng/mL, intra- and inter-assay CVs were <5.4% and <5.0% for total adiponectin, and <5.0% and <5.7% for HMW adiponectin, respectively.
2.4. Statistical Analyses
3. Results
3.1. Anthropometric and Body Composition Characteristics
3.2. Biochemical Markers of Bone Metabolism and Adipokines
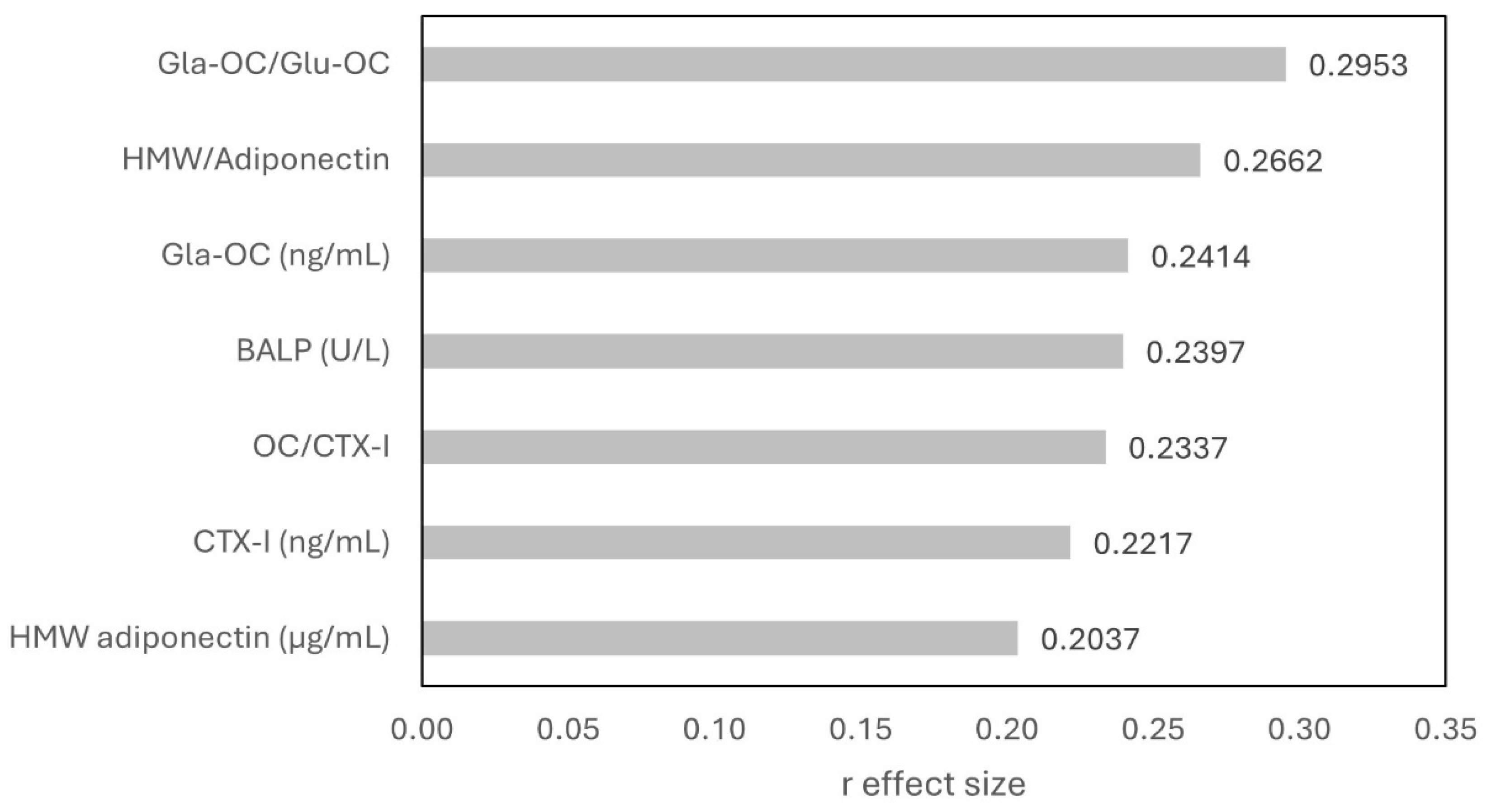
3.3. Correlation Analyses
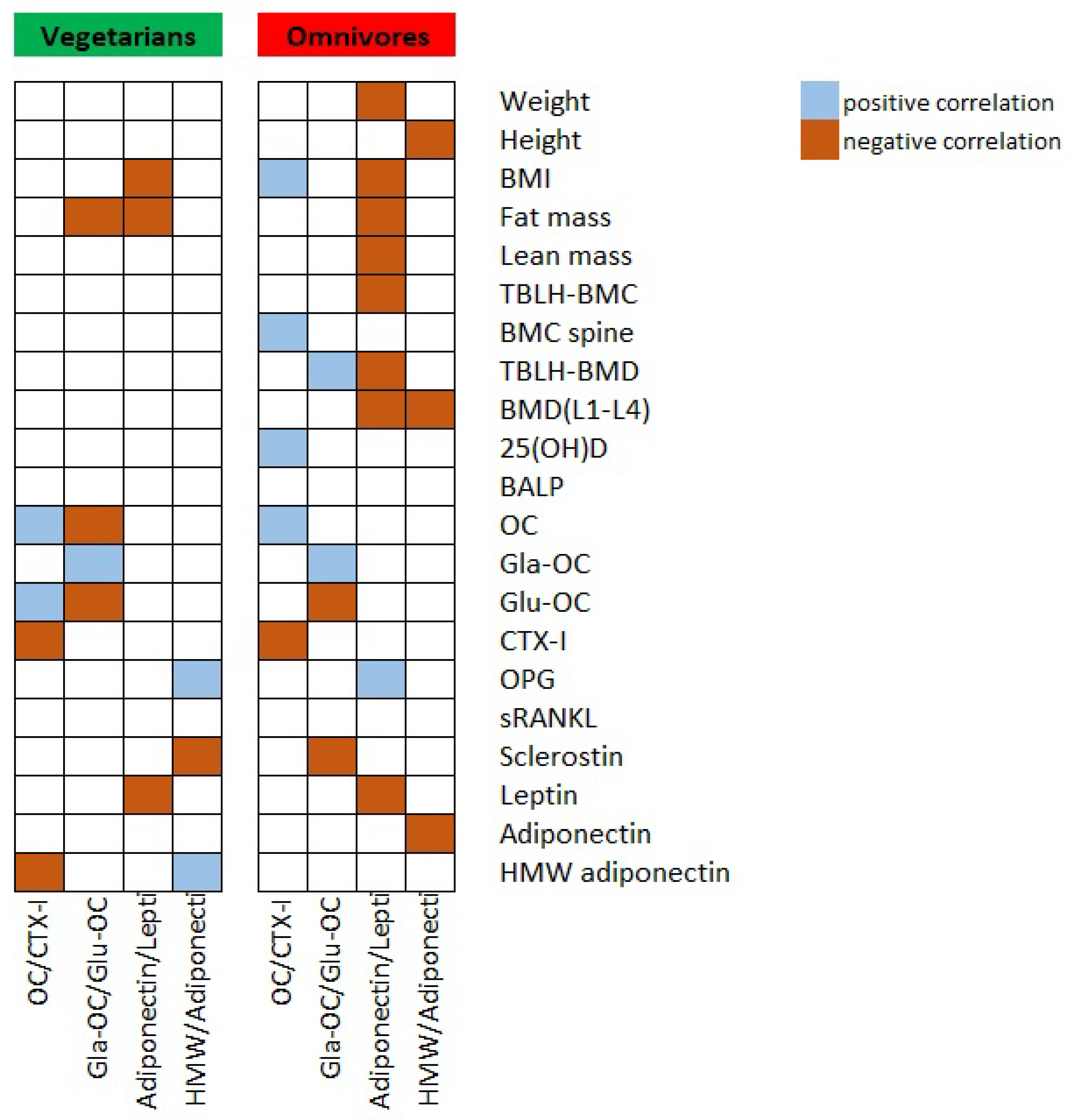
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chouraqui, J.P. Vegetarian diets and diets with restrict animal-source foods during childhood in high-income countries. Paediatr. Int. Child Health 2023, 43(4), 57-82. [CrossRef]
- Oussalah, A.; Levy, J.; Berthezene, C.; Alpers, D.H.; Gueant, J.L. Health outcomes associated with vegetarian diets: An umbrella review of systematic reviews and meta-analyses. Clin. Nutr. 2020, 39, 3283-3307. [CrossRef]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Goggi, S.; Metro, D.; Papa, M.; Sbarbati, R.; Scarino, M.L.; Pellegrini, N.; Sieri, S. A comprehensive review of healthy effects of vegetarian diets. Nutr. Metab. Cardiovasc. Dis. 2023, 33(7), 1308-1315. [CrossRef]
- Schurmann, S.; Kersting, M.; Alexy, U. Vegetarian diets in children: A systematic review. Eur. J. Nutr. 2017, 56, 1797-1817. [CrossRef]
- Henikova, M.; Ouradova, A.; Seling, E.; Tichanek, F.; Polakovicova, P.; Hrncirova, D.; Dlouhy, P.; Svetnicka, M.; El-Lababidi, E.; Potockova, J.; Kuhn, T.; Cahova, M.; Gojda, J. Dietary intake, nutritional status, and health outcomes among vegan, vegetarian, and omnivorous Czech families. Commun. Med. (Lond). 2025, 5(1), 538. [CrossRef]
- Liu, X.; Wu, Y.; Bennett, S.; Zou, J.; Xu, J.; Zhang, L. The effects of different dietary patterns on bone health. Nutrients 2024, 16, 2289. [CrossRef]
- McDonnell, J.M.; Darwish, S.; Bugtler, J.S.; Buckley, C.T. The role of adipokines in spinal disease: A narrative review. JOR Spine 2025, 8, e70083. [CrossRef]
- Mangion, D.; Pace, N.P.; Formosa, M.M. The relationship between adipokine levels and bone mass - A systematic review. Endocrinol. Diab. Metab. 2023, 6, e408. [CrossRef]
- Leszczyńska, D.; Szatko, A.; Toboła, A.; Karoń, K.; Misiorowski, W.; Glinicki, P.; Zgliszczyński, W. Novel aspects of biochemical assessment of bone remodeling and mineralization. Front. Endocrinol. (Lausanne) 2025, 16, 1702413. [CrossRef]
- Smith, C.; Lin, X.; Parker, L.; Yeap, B.B.; Hayes, A.; Levinger, I. The role of bone in energy metabolism: A focus on osteocalcin. Bone 2024, 188, 117238. [CrossRef]
- Udagawa, N.; Koide, M.; Nakamura, M.; Nakamichi, Y.; Yamashita, T.; Uehara, S.; Kobayashi, Y.; Furuya, Y.; Yasuda, H.; Fukuda, C.; Tsuda, E. Osteoclast differentiation by RANKL and OPG signaling pathways. J. Bone Miner. Metab. 2021, 39(1), 19-26. [CrossRef]
- Chen, L.; Gao, G.; Shen, L.; Yue, H.; Zhang, G.; Zhang, Z. Serum sclerostin and its association with bone turnover marker in metabolic bone diseases. Dis. Markers 2022, 2022, 7902046. [CrossRef]
- Chung, H.S.; Choi, K.M. Organokines in disease. Adv. Clin. Chem. 2020, 94:261-321.
- Iguacel, I.; Miguel-Berges, M.L.; Gomez-Bruton, A.; Moreno, L.A.; Julian, C. Veganism, vegetarianism, bone mineral density, and fracture risk: a systematic review and meta-analysis. Nutr. Rev. 2019, 77(1), 1-18. [CrossRef]
- Tong, T.Y.N.; Appleby, P.N.; Armstrong, M.E.G.; Fensom, G.K.; Knuppel, A.; Papiern, K.; Perez-Cornago, A.; Travie, R.C.; Key, T.J. Vegetarian and vegan diets and risks of total and site-specific fractures: results from the prospective EPIC-Oxford study. BMC Med. 2020, 23, 353.
- Webster, J.; Greenwood, D.C.; Cade, J.E. Risk of fracture in meat-eaters, pescarians, and vegetarians: a prospective cohort study of 413,914 UK Biobank participants. BMC Med. 2023, 21(1), 278. [CrossRef]
- Kraselnik, A. Risk of bone fracture on vegetarian and vegan diets. Curr. Nutr. Rep. 2024, 13, 331-339. [CrossRef]
- Ambroszkiewicz, J.; Chełchowska, M.; Szamotulska, K.; Rowicka, G.; Klemarczyk, W.; Strucińska, M.; Gajewska, J. The assessment of bone regulatory pathways, bone turnover, and bone mineral density in vegetarian and omnivorous children. Nutrients 2018, 10(2), 183. [CrossRef]
- Itkonen, S.T.; Hovinen, T.; Kettunen, E.; Freese, R.; Tilli, V.; Cashman, K.D.; Erkkola, M.; Suomalainen, A.; Korkalo, L. Bone and mineral metabolism in 2-7-year-old Finnish children and their caregivers following vegan, vegetarian, and omnivorous diets. Eur. J. Nutr. 2025, 64(6), 276. [CrossRef]
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian diets. J. Acad. Nutr. Diet. 2016, 116, 1970-1980. [CrossRef]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Papa, M.; Pellegrini, N.; Sbarbati, R.; Scarino, M.L.; Siani, V.; Sieri, S. Position paper on vegetarian diets from the working group of the Italian Society of Human Nutrition. Nutr. Metab. Cardiovasc. Dis. 2017, 27(12), 1037-1052. [CrossRef]
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellof, M.; Embleton, N.; Fidler Mis, N.; Hojsak, I.; Hulst, J.M.; Indrio, F.; Lapillonne, A.; Molgaard, C. Complementary feeding: A Position Paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPAGAN) Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 64(1), 119-132.
- Rudloff, S.; Buhrer, C.; Jochum, F.; Kauth, T.; Kersting, M.; Korner, A.; Koletzko, B.; Mihatsch, W.; Prell, C.; Reinehr, T.; Zimmer, K.P. Vegetarian diets in childhood and adolescence: Position paper of the nutrition Committee, German Society for Paediatric and Adolescent Medicine (DGKJ). Mol. Cell Pediatr. 2019, 6(1), 4.
- Hansch, L.; Fischer, M.; Langler, A.; Michalsen, A.; Weder, S.; Keller, M.; Alexy, U.; Perrar, I. Dietary patterns among children and adolescents in Germany consuming vegetarian, vegan or omnivore diets: results of the VeChi Youth Study. Eur. J. Nutr. 2024, 63, 3161-3174. [CrossRef]
- Jarosz, M.; Rychlik, E.; Stoś, K.; Charzewska, J. Normy żywienia dla populacji polskiej i ich zastosowania; Jarosz, M., Ed.; NIZP-PZH: Warsaw, Poland, 2020; pp. 26–148.
- Ambroszkiewicz, J.; Klemarczyk, W.; Mazur, J.; Gajewska, J.; Rowicka, G.; Strucińska M, Chełchowska M. Serum hepcidin and soluble transferrin receptor in the assessment of iron metabolism in children on a vegetarian diet. Biol. Trace Elem. Res. 2017, 180, 182–190. [CrossRef]
- Płudowski, P.; Kos-Kudła, B.; Walczak, M.; Fal, A.; Zozulińska-Ziółkiewicz, D.; Sieroszewski, P.; Peregud-Pogorzelski, J.; Lauterbach, R.; Targowski, T.; Lewiński, A.; et al. Guidelines for Preventing and Treating Vitamin D Deficiency: A 2023 Update in Poland. Nutrients 2023, 15, 695. [CrossRef]
- Kułaga, Z.; Różdżyńska-Świątkowska, A.; Grajda, A.; Gurzkowska, B.; Wojtyło, M.; Góźdź, M.; Światek-Leśniak, A.; Litwin, M. Percentile charts for growth and nutritional status assessment in Polish children and adolescents from birth to 18 year of age. Stand. Med. 2015, 12, 119–135.
- Kalkwarf, H.J.; Abrams, S.A.; DiMeglio, L. A.; Koo, W. W. K.; Specke, B. L.; Weiler, H, et al. Bone densitometry in infants and young children: the 2023 ISCD Pediatric Official Positions. J. Clin. Densitom. 2014, 17(2), 243-257. [CrossRef]
- McVey, M. K.; Geraghty, A. A.; O`Brien, E.C.; McKenna, M. J.; Kilbane, M. T.; Crowley, R. K.; Twomey, P. J.; McAuliffe, F. M. The impact of diet, body composition, and physical activity on child bone mineral density at five years age-findings from the ROLO Kids Study. Eur. J. Pediatr. 2020, 179(1), 121-131. [CrossRef]
- Zhang, L.; Li, H.; Zhang, Y.; Kong, Z.; Zhang, T.; Zhang, Z. Association of body compositions and bone mineral density in Chinese children and adolescents: Compositional data analysis. Biomed. Res. Int. 2021, 2021, 1904343. [CrossRef]
- Ambroszkiewicz J.; Chełchowska, M.; Szamotulska, K.; Rowicka, G.; Klemarczyk, W.; Strucińska, M.; Gajewska, J. Bone status and adipokine levels in children on vegetarian and omnivorous diets. Clin. Nutr. 2019, 38(2), 730-737. [CrossRef]
- Ambroszkiewicz, J.; Gajewska, J.; Szamotulska, K.; Rowicka, G.; Klemarczyk, W.; Strucińska, M.; Chełchowska, M. Comparative analysis of myokines and bone metabolism markers in prepubertal vegetarian and omnivorous children. Nutrients 2024, 16(13), 2009. [CrossRef]
- Meyer, R.; Protudjer J.L. Plant-based diets and child growth. Curr. Opin. Clin. Nutr. Metab. Care 2025, 28(3), 274-283. [CrossRef]
- Reis, D.; Schwermer, M.; Nowak, L.; Naami, N.; Zuzak, T.J.; Langler, A. Vegetarian diet and dietary intake, health, and nutritional status in infants, children, and adolescents: A systematic review. Nutrients 2025, 17, 2181. [CrossRef]
- Kiely, M.E. Risks and benefits of vegan and vegetarian diets in children. Proc. Nutr. Soc. 2021, 80(2), 159-164. [CrossRef]
- Vaquero, M. P.; Garcia-Maldonado, E.; Gallego-Narbon, A.; Zapatera, B.; Alcorta, A.; Martin-Suarez, M. Iron deficiency is associated with elevated parathormone levels, low vitamin D status, and risk of bone loss in omnivores and plant-based diet consumers. Int. J. Mol. Sci. 2024, 25(19), 10290. [CrossRef]
- Goldman, D.M.; Warbeck, C.B.; Barbaro, R.; Khambatta, C.; Nagra, M. Assessing the roles of retinol, vitamin K2, carnitine, and creatinine in plant-based diets: A narrative review of nutritional adequacy and health implications. Nutrients 2025, 17(3), 525. [CrossRef]
- Michalopoulou, O.; Kostoglou-Athanassiou, I.; Troupis, G.; Mastorakos, G. The intimate relationship between adipose tissue, fertility, and bone. J. Frailty Sarcopenia Falls 2026, 11(1), 80-90. [CrossRef]
- Hsu, C-N.; Kao, C-H.; Yang, C-H.; Cheng, M-T.; Hsu, Y-P.; Hong, S-G.; Yao, C-L.; Chen, Y-H. Leptin promotes the expression of pro-inflammatory mediator genes but does not alter osteoclastogenesis and early stage differentiation osteoblasts. J. Physiol. Investig. 2024, 67(6), 355-363. [CrossRef]
- Soininen, S.; Sidoroff, V.; Lindi, V.; Mahonen, A.; Kroger, L.; Kroger, H.; Jaaskelainen, J.; Atalay, M.; Laaksonen, D.E.; Laitinen, T.; Lakka, T.A. Body fat mass, lean body mass and associated biomarkers as determinants of bone mineral density in children 6-8 years of age – The Physical Activity and Nutrition in Children (PANIC) study. Bone 2018, 108, 106-114.
- Ambroszkiewicz, J.; Chełchowska, M.; Mazur, J.; Rowicka, G.; Gajewska, J. Relationships between body weight status and serum levels of adipokine, myokine and bone metabolism parameters in healthy normal weight and thin children. J. Clin. Med. 2022, 11(14), 4013. [CrossRef]
- Khwanchuea, R.; Punsawad, C. Correlations among whole-body fat, bone, and biomarkers in boys and girls with obesity: a cross-sectional study. Ann. Pediatr. Endocrinol. Metab. 2026, 31(1), 55-65. [CrossRef]
- Legroux-Gerot, I.; Vignau, J.; Viltart, O.; Hardouin, P.; Chauveau, C.; Cortet, B. Adipokines and bone status in a cohort of anorexic patients. Joint Bone Spine 2019, 86(1), 95-101. [CrossRef]
- Bertels, J.C.; Koehnken Sawall, J.; Dulmovits, B.; Liu, X.; Phan, A.; Ji, X.; Song, F.; Thom, C.; Long, F. IGF-1 from bone marrow Adipoq-lineage cells stimulates endocortical bone formation in mature female mice. JBMR Plus 2026, 10(4), ziag024. [CrossRef]
- China, S.P.; Sanyal, S.; Chattopadhyay, N. Adiponectin signaling and its role in bone metabolism. Cytokine 2018, 112, 116-131. [CrossRef]
| Vegetarians (n = 50) | Omnivores (n = 40) | p | |
|---|---|---|---|
| Weight (kg) | 19.1 ± 2.1 | 19.7 ± 3.4 | 0.791 |
| Height (cm) | 114.9 ± 4.5 | 115.3 ± 5.6 | 0.569 |
| BMI (kg/m2) | 14.4 ± 1.1 | 14.7 ± 1.6 | 0.448 |
| BMI Z-score | -0.529 ± 0.888 | -0.345 ± 1.064 | 0.436 |
| Fat mass (%) | 17.6 (15.0–21.5) | 21.4 (16.5–24.2) | 0.133 |
| Fat mass (kg) | 3.18 (2.67–3.76) | 3.35 (2.79–4.45) | 0.294 |
| Lean mass (kg) | 14.30 ± 1.89 | 14.98 ± 2.86 | 0.318 |
| TBLH-BMC (g) | 607 ± 115 | 621 ± 154 | 0.789 |
| BMC spine (g) | 49.8 ± 10.9 | 52.7 ± 11.8 | 0.338 |
| TBLH-BMD (g/cm2) | 0.586 ± 0.050 | 0.607 ± 0.055 | 0.078 |
| TBLH-BMD Z-score | -0.382 ± 0.852 | -0.318 ± 0.676 | 0.758 |
| BMD (L1-L4) (g/cm2) | 0.573 ± 0.068 | 0.593 ± 0.052 | 0.259 |
| BMD (L1-L4) Z-score | -0.740 ± 0.942 | -0.605 ± 0.593 | 0.302 |
| Vegetarians | Omnivores | p | |
|---|---|---|---|
| Bone metabolism markers | |||
| 25(OH)D (ng/mL) | 27.3 ± 9.7 | 29.1 ± 6.8 | 0.408 |
| BALP (U/L) | 130.5 (83.4–160.6) | 112.6 (90.6–128.3) | 0.023 |
| OC (ng/mL) | 73.6 (56.4–94.2) | 70.2 (62.7–90.7) | 0.955 |
| Gla-OC (ng/mL) | 33.9 (28.6–38.6) | 30.2 (23.0–35.9) | 0.022 |
| Glu-OC (ng/mL) | 24.7 (15.5–35.9) | 30.1 (24.6–38.1) | 0.070 |
| CTX-I (ng/mL) | 1.947 ± 0.495 | 1.695 ± 0.580 | 0.035 |
| OPG (pmol/L) | 4.57 ± 0.92 | 4.62 ± 0.93 | 0.890 |
| sRANKL (ng/mL) | 2036 (692–3726) | 1729 (1176–3111) | 0.782 |
| Sclerostin (ng/mL) | 0.424 ± 0.123 | 0.436 ± 0.096 | 0.470 |
| OC/CTX-I | 37.6 (27.4–51.6) | 46.2 (37.9–55.9) | 0.027 |
| Gla-OC/Glu-OC | 1.37 (0.87–2.45) | 1.01 (0.74–1.32) | 0.005 |
| OPG/sRANKL | 0.002 (0.001–0.004) | 0.003 (0.001–0.005) | 0.881 |
| Adipokines | |||
| Leptin (ng/mL) | 1.40 (0.82–1.85) | 1.54 (0.77–3.15) | 0.342 |
| Adiponectin (µg/mL) | 9.16 ± 2.26 | 9.66 ± 2.87 | 0.567 |
| HMW adiponectin (µg/mL) | 5.79 ± 1.75 | 6.48 ± 1.90 | 0.050 |
| Adiponectin/leptin | 7.14 (4.08–10.18) | 6.71 (3.07–10.38) | 0.470 |
| HMW/Adiponectin | 63.0 ± 8.9 | 67.8 ± 8.8 | 0.012 |
| Vegetarians | Omnivores | |||||||
|---|---|---|---|---|---|---|---|---|
| TBLH-BMC | BMC spine | TBLH-BMD | BMD (L1–L4) | TBLH-BMC | BMC spine | TBLH BMD | BMD (L1–L4) | |
| Weight | 0.730 0.000 |
0.775 0.000 |
0.549 0.000 |
0.584 0.000 |
0.703 0.000 |
0.742 0.000 |
0.504 0.001 |
0.579 0.000 |
| Height | 0.676 0.000 |
0.691 0.000 |
0.602 0.000 |
0.490 0.000 |
0.561 0.000 |
0.646 0.000 |
0.384 0.014 |
0.452 0.004 |
| BMI | 0.344 0.014 |
0.436 0.002 |
0.463 0.001 |
0.314 0.026 |
0.673 0.000 |
0.569 0.000 |
0.489 0.001 |
0.422 0.007 |
| Fat mass | 0.163 0.257 |
0.368 0.009 |
0.342 0.015 |
0.419 0.002 |
0.373 0.019 |
0.387 0.015 |
0.339 0.035 |
0.366 0.022 |
| Lean mass | 0.757 0.000 |
0.606 0.000 |
0.710 0.000 |
0.334 0.018 |
0.652 0.000 |
0.611 0.000 |
0.621 0.000 |
0.500 0.001 |
| 25(OH)D | 0.092 0.525 |
0.054 0.616 |
0.052 0.625 |
0.155 0.284 |
-0.119 0.463 |
-0.090 0.583 |
0.083 0.611 |
-0.096 0.583 |
| BALP | 0.252 0.078 |
0.312 0.027 |
0.255 0.074 |
0.164 0.256 |
0.186 0.252 |
0.012 0.940 |
0.048 0.768 |
0.018 0.914 |
| OC | 0.086 0.553 |
0.116 0.421 |
0.097 0.501 |
0.333 0.018 |
0.026 0.873 |
0.126 0.438 |
0.054 0.739 |
0.259 0.111 |
| Gla-OC | 0.172 0.234 |
0.176 0.221 |
0.165 0.252 |
0.130 0.369 |
0.145 0.371 |
0.013 0.936 |
0.070 0.668 |
0.076 0.644 |
| Glu-OC | 0.007 0.960 |
0.040 0.785 |
0.011 0.941 |
0.300 0.034 |
-0.144 0.375 |
0.008 0.962 |
-0.218 0.176 |
-0.061 0.710 |
| CTX-I | 0.198 0.169 |
0.367 0.009 |
0.320 0.024 |
0.234 0.102 |
-0.169 0.296 |
-0.240 0.136 |
-0.215 0.183 |
-0.041 0.805 |
| OPG | 0.118 0.415 |
0.109 0.451 |
0.174 0.227 |
0.135 0.350 |
-0.367 0.020 |
-0.374 0.017 |
-0.381 0.015 |
-0.215 0.183 |
| sRANKL | 0.044 0.762 |
0.159 0.271 |
0.109 0.450 |
0.235 0.350 |
-0.232 0.150 |
-0.280 0.081 |
-0.038 0.814 |
-0.023 0.888 |
| Sclerostin | 0.147 0.308 |
0.106 0.463 |
0.128 0.374 |
0.313 0.027 |
0.065 0.692 |
0.030 0.854 |
-0.079 0.628 |
0.151 0.359 |
| Leptin | -0.007 0.962 |
-0.033 0.822 |
-0.094 0.517 |
-0.054 0.712 |
0.618 0.000 |
0.564 0.000 |
0.535 0.000 |
0.326 0.043 |
| Total adiponectin | -0.029 0.841 |
0.148 0.304 |
0.053 0.716 |
-0.003 0.981 |
0.181 0.265 |
0.279 0.081 |
0.238 0.139 |
0.532 0.000 |
| HMW adiponectin | 0.020 0.890 |
0.151 0.294 |
0.075 0.603 |
-0.067 0.644 |
0.099 0.542 |
0.240 0.135 |
0.353 0.025 |
0.362 0.023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




