Submitted:
02 May 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Description, Guidelines, and Epidemiology
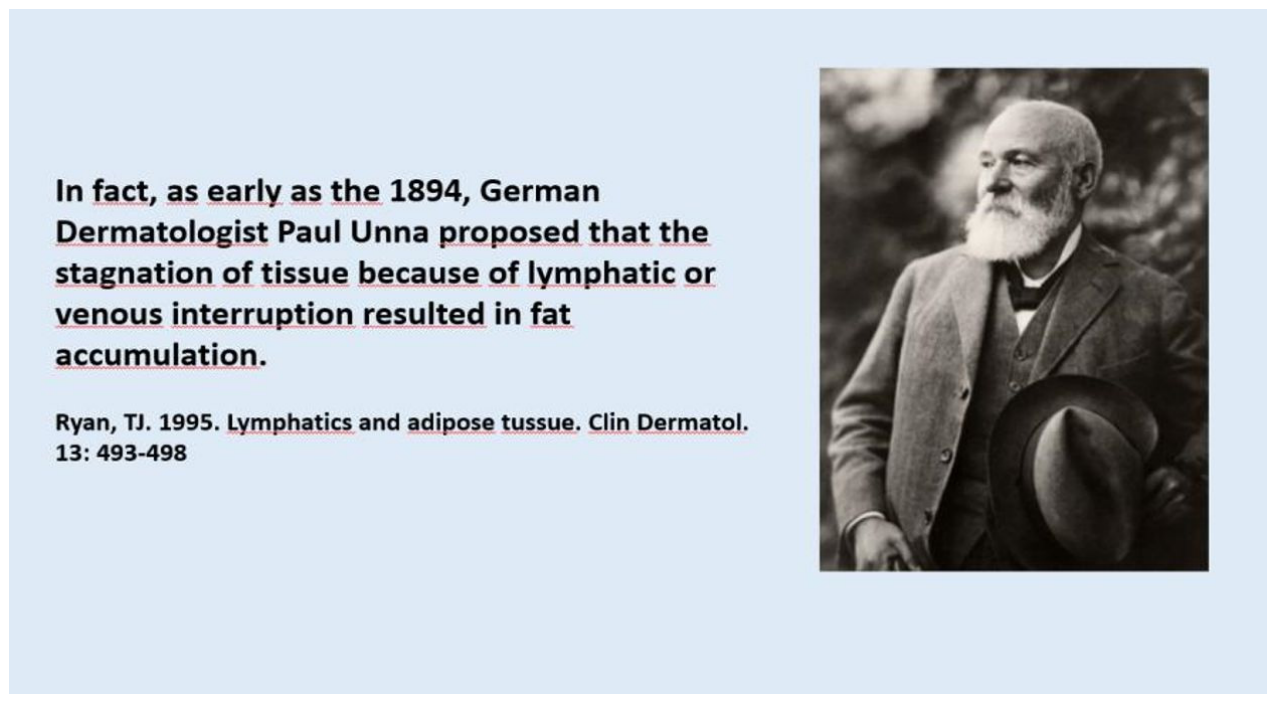
2. Historical Development and Early Concepts
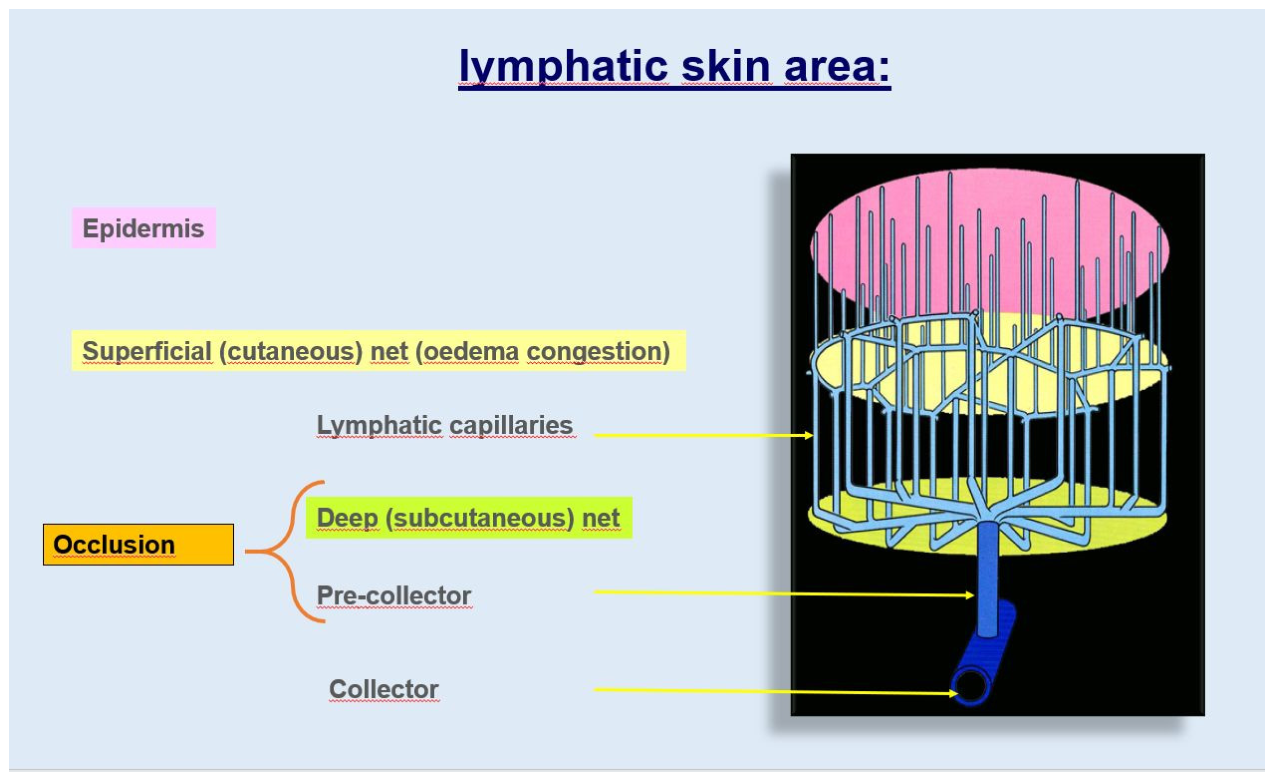
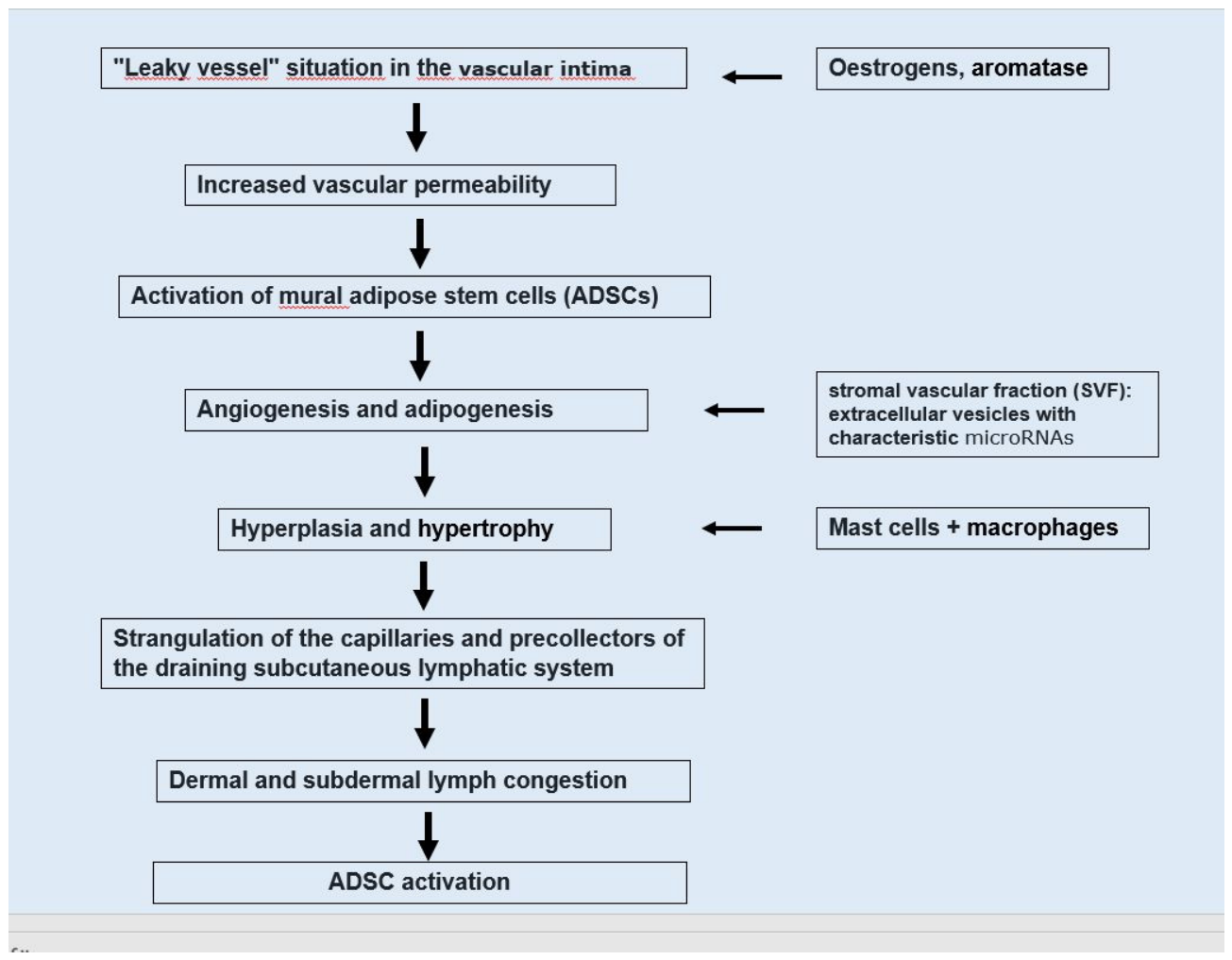
3. Anatomical Basics and Pathophysiological Mechanisms (Stem Cell Regeneration and Degeneration)
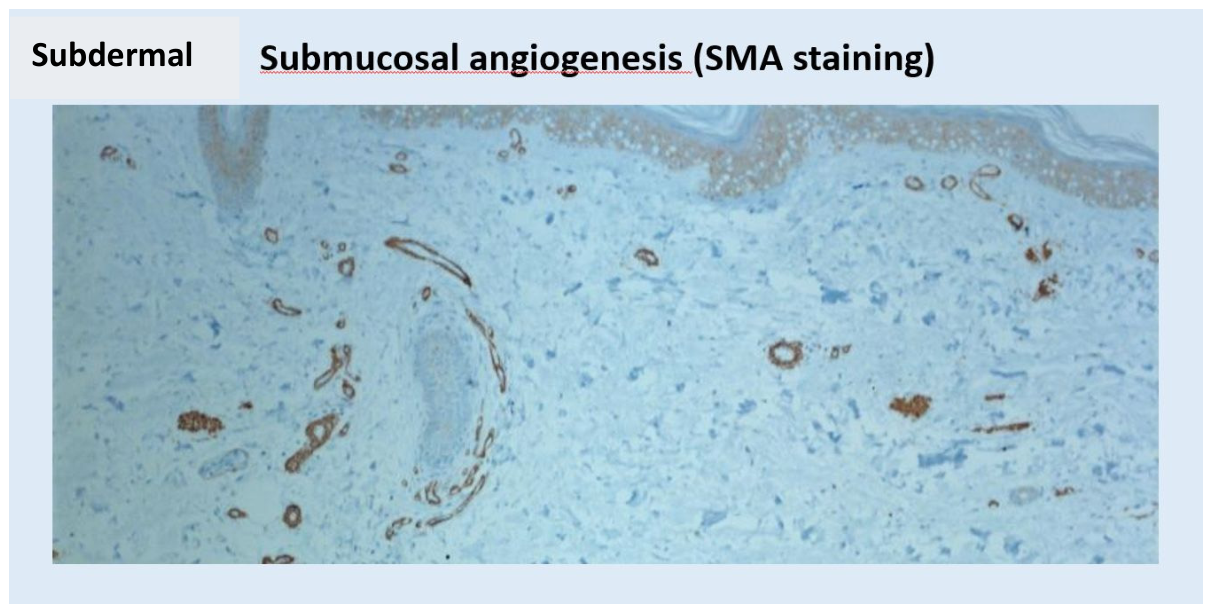
- Spontaneous skin bleeding and bleeding tendency (suffusions) due to fragile neocapillaries in the context of increased angiogenesis and adipogenesis (own histo-system staining) (Figure 3).
- 2.
- Negative pitting in dermal-subdermal oedema (13).
- 3.
- Pain/pressure/tension and heaviness caused by inflammatory activation of phagocytic cell elements (macrophages, mast cells) - (10). (Figure 4)
- 4.
- Negative influence on basal metabolic rate in the majority of cases: activation of storage function or metabolism with secondary fat accumulation (hypertrophy) in the abdominal area (100%) and additional metabolic accumulation in areas of mixed fat (buttocks, thighs, and upper arms) (30, 33). Interestingly, this metabolic storage function associated with lipedema does not lead to metabolic diseases such as type 2 diabetes mellitus (16, 33). This correlation was already described around 100 years ago by internists experienced in clinical observation as "healthy fat people."
- 5.
- Triggerable pressure pain due to fascial congestion. This results on the one hand from the subcutaneous lipedema compartment (adipofascial congestion) and on the other hand from a mostly visceroabdominal lymphatic drainage disorder in the context of metabolic fat accumulation (myofascial congestion) (17, 35).
- 6.
- The genetically determined increase in aromatase activity also plays a significant clinical role in lipedema flare-ups during menopause. (15).
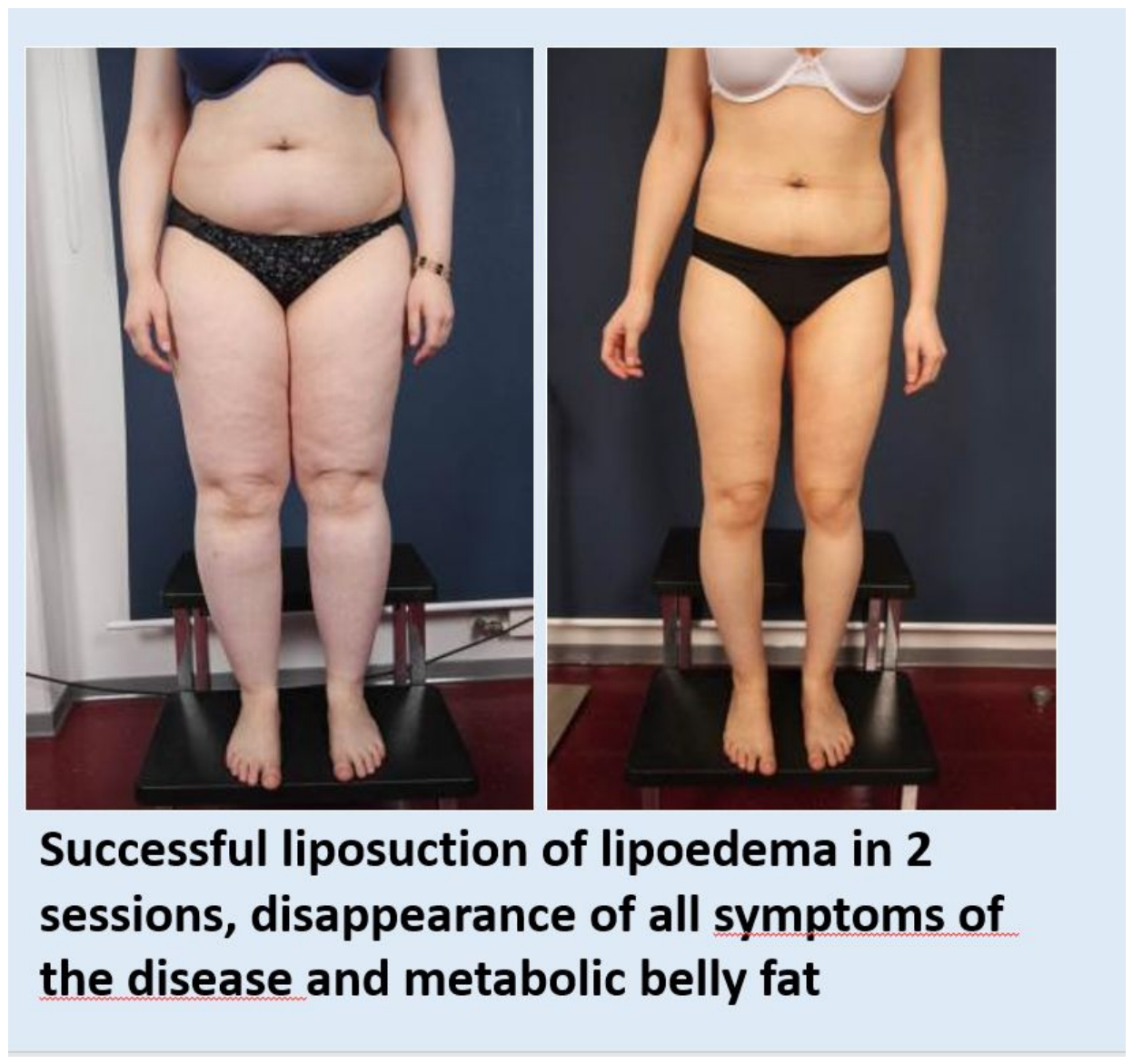
4. Confirmation of Pathophysiological Processes After Therapeutic Liposuction
5. Discussion
6. Clinical Implications
7. Limitations of the Proposed Model
8. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hucho T. Lipedema pain—the neglected symptom. Dermatologie (Heidelb). 2023;74(8):575-579. [CrossRef]
- Chakraborty A, Crescenzi R, Usman TA, Reyna AJ, Garza ME, Al-Ghadban S, Herbst KL, Donahue PMC, Rutkowski JM. Indications of peripheral pain, dermal hypersensitivity, and neurogenic inflammation in patients with lipedema. Int J Mol Sci. 2022;23(18):10313. [CrossRef]
- Faerber G, Cornely M, Daubert C, Erbacher G, Fink J, Hirsch T, Mendoza E, Miller A, Rabe E, Rapprich S, Reich-Schupke S, Stücker M, Brenner E. S2k guideline lipedema. J Dtsch Dermatol Ges. 2024;22(9):1303-1315. [CrossRef]
- Rapprich S, Baum S, Kaak I, Kottmann T, Podda M. Treatment of lipoedema using liposuction. Phlebologie. 2015;44(3):121-132. [CrossRef]
- Spalding KL, Arner E, Westermark PO, Bernard S, Buchholz BA, Bergmann O, Blomqvist L, Hoffstedt J, Näslund E, Britton T, Concha H, Hassan M, Rydén M, Frisén J, Arner P. Dynamics of fat cell turnover in humans. Nature. 2008;453(7196):783-787. [CrossRef]
- Priglinger E, Wurzer C, Steffenhagen C, Maier J, Hofer V, Peterbauer A, Nuernberger S, Redl H, Wolbank S, Sandhofer M. The adipose tissue-derived stromal vascular fraction cells from lipedema patients: Are they different? Cytotherapy. 2017;19(7):849-860. [CrossRef]
- Bauer J. Über Fettansammlungen. Klin Wochenschr. 1922;1:1077-1083.
- Günther H. Die Lipomatosis und ihre klinischen Formen. In: Arbeiten aus der medizinischen Klinik Leipzig. Jena: G. Fischer; 1920. Heft 5.
- Priglinger E, Strohmeier K, Weigl M, Lindner C, Auer D, Gimona M, Barsch M, Jacak J, Redl H, Grillari J, Sandhofer M, Hackl M, Wolbank S. SVF-derived extracellular vesicles carry characteristic miRNAs in lipedema. Sci Rep. 2020;10(1):7211. [CrossRef]
- Al-Ghadban S, Cromer W, Allen M, Ussery C, Badowski M, Harris D, Herbst KL. Dilated blood and lymphatic microvessels, angiogenesis, increased macrophages, and adipocyte hypertrophy in lipedema thigh skin and fat tissue. J Obes. 2019;2019:8747461. [CrossRef]
- Sandhofer M, Schauer P, Anderhuber F. Haut-Unterhaut-Fett-Faszien. J Ästhet Chir. 2015;8(4):151-156. [CrossRef]
- Suga H, Araki J, Aoi N, Kato H, Higashino T, Yoshimura K. Adipose tissue remodeling in lipedema: adipocyte death and concurrent regeneration. J Cutan Pathol. 2009;36(12):1293-1298. [CrossRef]
- Crescenzi R, Donahue PMC, Petersen KJ, Garza M, Patel N, Lee C, Beckman JA, Donahue MJ. Upper and lower extremity measurement of tissue sodium and fat content in patients with lipedema. Obesity (Silver Spring). 2020;28(5):907-915. [CrossRef]
- Amann-Vesti BR, Franzeck UK, Bollinger A. Microlymphatic aneurysms in patients with lipedema. Lymphology. 2001;34(4):170-175.
- Strohmeier K, Hofmann M, Jacak J, Narzt MS, Wahlmueller M, Mairhofer M, Schaedl B, Holnthoner W, Barsch M, Sandhofer M, Wolbank S, Priglinger E. Multi-level analysis of adipose tissue reveals the relevance of perivascular subpopulations and an increased endothelial permeability in early-stage lipedema. Biomedicines. 2022;10(5):1163. [CrossRef]
- Blüher M. Metabolically healthy obesity. Endocr Rev. 2020;41(3):bnaa004. [CrossRef]
- Pilsl U, Anderhuber F. Anatomie des Lymphsystems. J Ästhet Chir. 2019;12(1):51-58. [CrossRef]
- Simpson ER. Sources of estrogen and their importance. J Steroid Biochem Mol Biol. 2003;86(3-5):225-230. [CrossRef]
- Cleland WH, Mendelson CR, Simpson ER. Effects of aging and obesity on aromatase activity of human adipose cells. J Clin Endocrinol Metab. 1985;60(1):174-177. [CrossRef]
- Nelson LR, Bulun SE. Estrogen production and action. J Am Acad Dermatol. 2001;45(3 Suppl):S116-S124. [CrossRef]
- Davis KE, Neinast MD, Sun K, Skiles WM, Bills JD, Zehr JA, Zeve D, Hahner LD, Cox DW, Gent LM, Xu Y, Wang ZV, Khan SA, Clegg DJ. The sexually dimorphic role of adipose and adipocyte estrogen receptors in modulating adipose tissue expansion, inflammation, and fibrosis. Mol Metab. 2013;2(3):227-242. [CrossRef]
- Karastergiou K, Smith SR, Greenberg AS, Fried SK. Sex differences in human adipose tissues—the biology of pear shape. Biol Sex Differ. 2012;3(1):13. [CrossRef]
- Straub LG, Funcke JB, Joffin N, Joung C, Al-Ghadban S, Zhao S, Zhu Q, Kruglikov IL, Zhu Y, Langlais PR, Gordillo R, Herbst KL, Scherer PE. Defining lipedema’s molecular hallmarks by multi-omics approach for disease prediction in women. Metabolism. 2025;168:156191. [CrossRef]
- Sanchez-De la Torre Y, Wadeea R, Rosas V, Herbst KL. Lipedema: friend and foe. Horm Mol Biol Clin Investig. 2018;33(1). [CrossRef]
- Bilancini S, Lucchi M, Tucci S, Eleuteri P. Functional lymphatic alterations in patients suffering from lipedema. Angiology. 1995;46(4):333-339. [CrossRef]
- Katzer K, Hill JL, McIver KB, Foster MT. Lipedema and the potential role of estrogen in excessive adipose tissue accumulation. Int J Mol Sci. 2021;22(21):11720. [CrossRef]
- Wright T, Babula M, Schwartz J, Wright C, Danesh N, Herbst K. Lipedema reduction surgery improves pain, mobility, physical function, and quality of life: Case series report. Plast Reconstr Surg Glob Open. 2023;11(11):e5436. [CrossRef]
- Sandhofer M, Hofer V, Sandhofer M, Sonani M, Moosbauer W, Barsch M. High volume liposuction in tumescence anesthesia in lipedema patients: A retrospective analysis. J Drugs Dermatol. 2021;20(3):326-334. [CrossRef]
- Baumgartner A, Hueppe M, Meier-Vollrath I, Schmeller W. Improvements in patients with lipedema 4, 8 and 12 years after liposuction. Phlebology. 2021;36(2):152-159. [CrossRef]
- Sandhofer M, Schauer P, Anderhuber F. The aesthetic upper arm. I. On the anatomy and classification of the lipodysmorphic upper arm. Kosmet Med. 2013;34(2):56-61.
- Sandhofer M, Hanke CW, Habbema L, Podda M, Rapprich S, Schmeller W, Herbst K, Anderhuber F, Pilsl U, Sattler G, Sandhofer M, Moosbauer W, Sattler S, Schauer P, Faulhaber J, Maier S, Barsch M, Mindt S, Halk AB. Prevention of progression of lipedema with liposuction using tumescent local anesthesia: Results of an international consensus conference. Dermatol Surg. 2020;46(2):220-228. [CrossRef]
- Stöberl Ch, Partsch H, Urbanek A. Indirekte Lymphographie bei Lipödem. In: Földi E, Mannheimer E, Hrsg. Ödem. Erlangen: Perimed; 1986:129-132.
- Sandhofer M, Sandhofer M, Schauer P, Anderhuber F. Das Lipödem: Anatomische Studie, Diagnostik und perioperatives Umfeld. J Ästhet Chir. 2017;10(2):61-70. [CrossRef]
- Norr KF, Schulman BA. The second time around: Parity and birth experience. J Obstet Gynecol Neonatal Nurs. 1980;9(3):167-171. [CrossRef]
- Stecco C, Driscoll M, Huijing P, Schleip R, eds. Fascia: The Tensional Network of the Human Body. The science and clinical applications in manual and movement therapy. 2nd ed. London: Elsevier; 2021. ISBN 9780702071836.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



