Submitted:
01 May 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
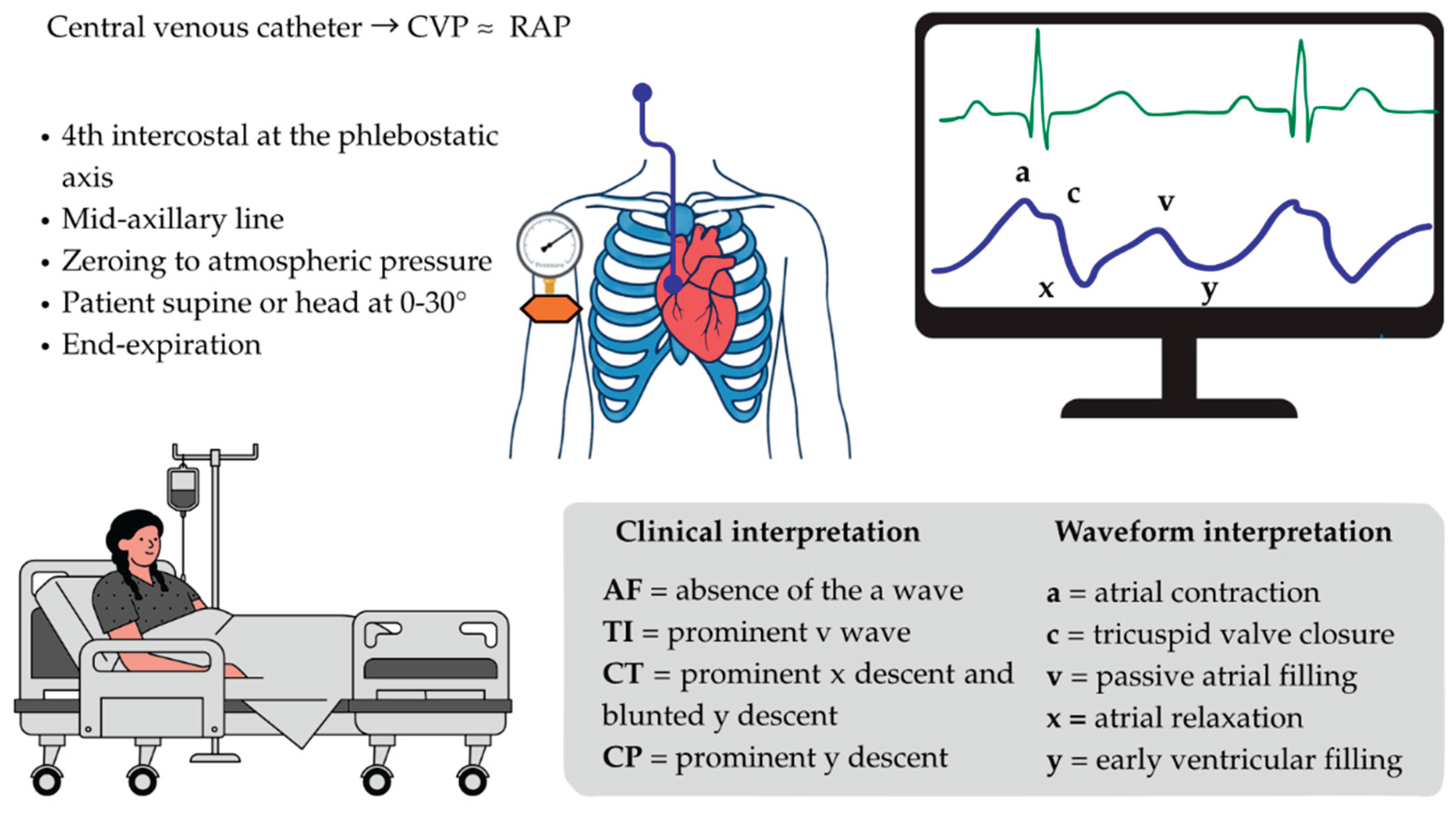
2. CVP Measurement and Interpretation
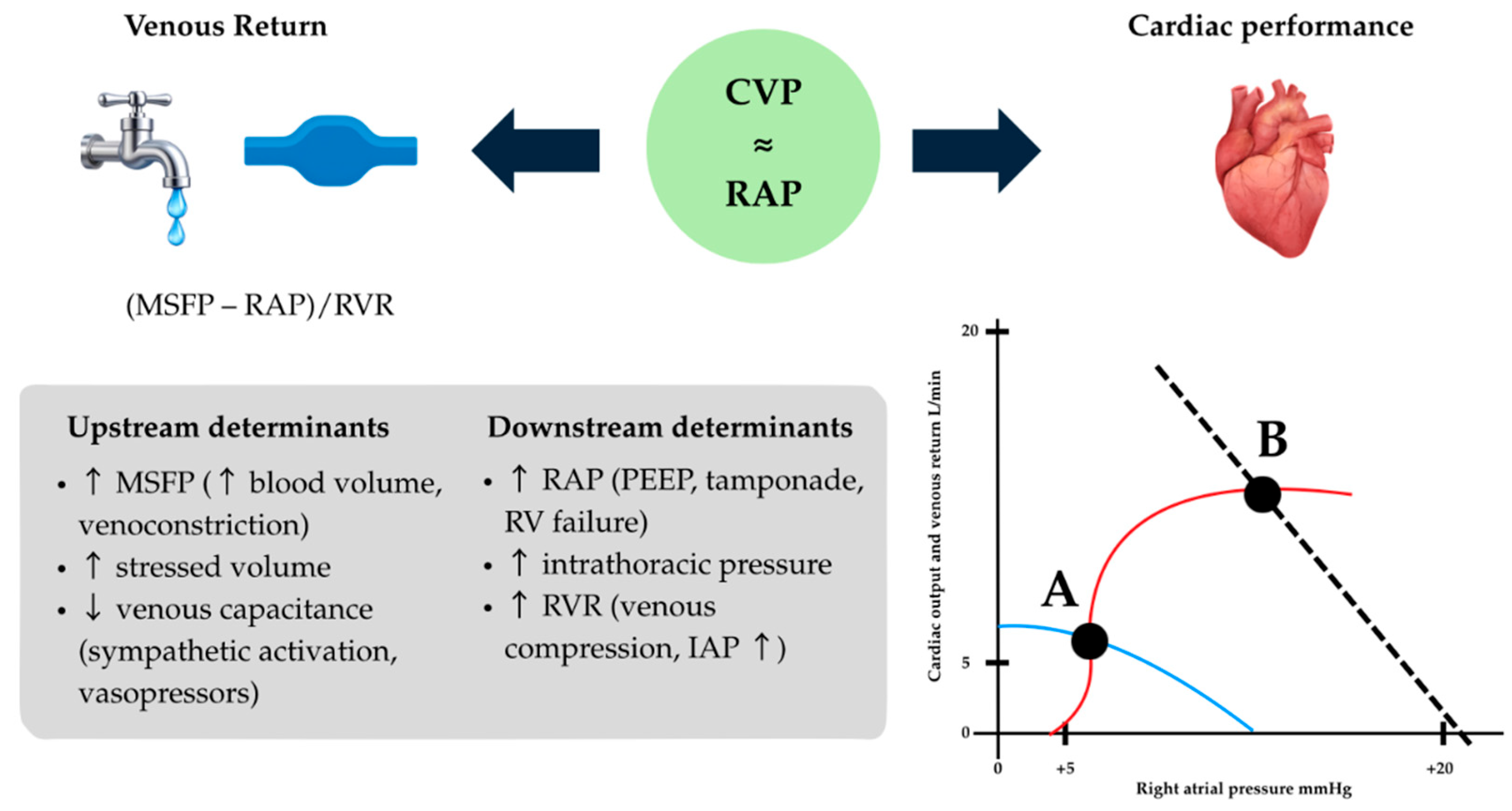
3. Historical Perspective and Physiological Basis of CVP
4. The Changing Role of CVP in Sepsis Resuscitation Guidelines
5. Divergences from Evidence
5.1. Reasons of CVP Persistence in Clinical Practice
5.2. Physiological Interpretation
6. Modern CVP Uses
6.1. Venous Congestion and Organ Perfusion
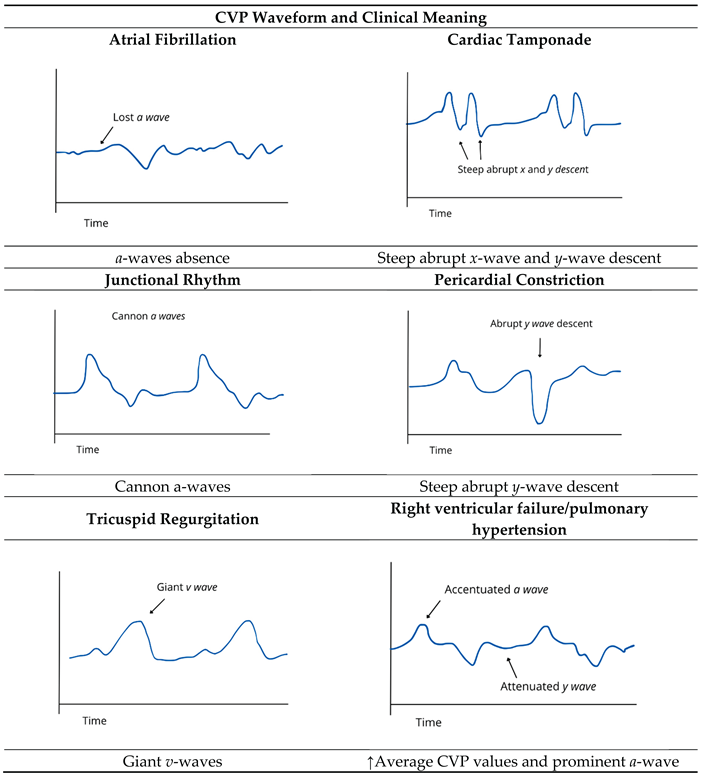
6.2. Waveform Morphology and Right Heart Pathophysiology
6.3. Role During Fluid Challenge
6.4. Emerging and Expanding Applications
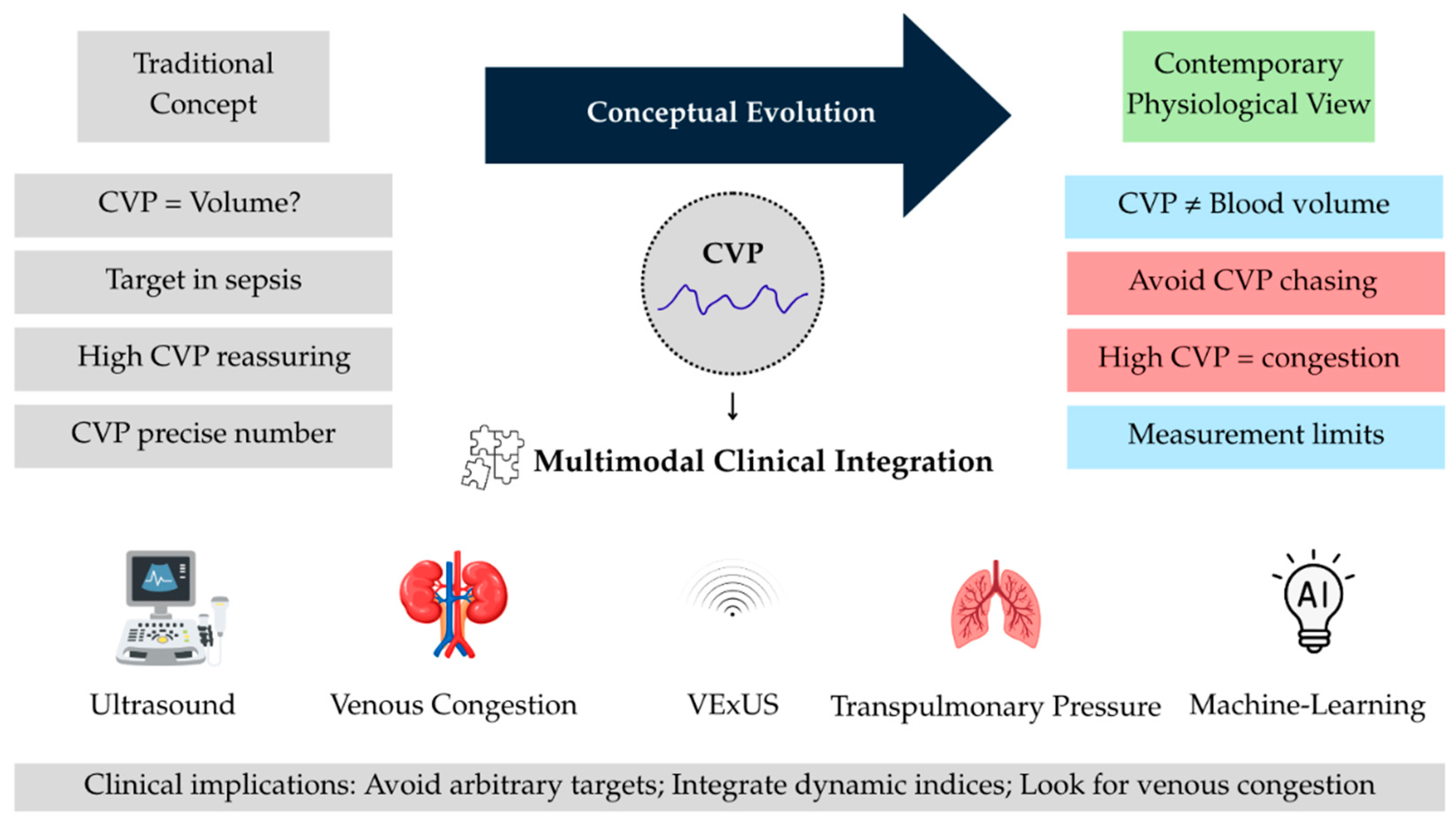
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AF | Atrial Fibrillation |
| ARDS | Acute Respiratory Distress Syndrome |
| CO | Cardiac Output |
| CP | Constrictive Pericarditis |
| CVC | Central Venous Catheter |
| CVP | Central Venous Pressure |
| EGDT | Early Goal-Directed Therapy |
| FR | Fluid Responsiveness |
| MAP | Mean Arterial Pressure |
| MPP | Mean Perfusion Pressure |
| MSFP | Mean Systemic Filling Pressure |
| PAOP | Pulmonary Artery Occlusion Pressure |
| PEEP | Positive End-Expiratory Pressure |
| PPV | Pulse Pressure Variation |
| PT | Pericardial Tamponade |
| RAP | Right Atrial Pressure |
| RV | Right Ventricle |
| RVR | Resistance to Venous Return |
| ScvO₂ | Central Venous Oxygen Saturation |
| SVC | Superior Vena Cava |
| SV | Stroke Volume |
| SVV | Stroke Volume Variation |
| TPP | Transpulmonary Pressure |
| VExUS | Venous Excess Ultrasound Score |
| VILI | Ventilator-Induced Lung Injury |
References
- Magder, S. Right Atrial Pressure in the Critically Ill. Chest 2017, 151, 908–916. [CrossRef]
- Lloyd-Donald, P.; Fujino, M.; Waldman, B.; Miles, L.F. Measurement and Interpretation of Central Venous Pressure: A Narrative Review. Anaesthesia 2025, 80, 1093–1102. [CrossRef]
- De Backer, D.; Vincent, J.-L. Should We Measure the Central Venous Pressure to Guide Fluid Management? Ten Answers to 10 Questions. Crit Care 2018, 22, 43. [CrossRef]
- Magder, S. Central Venous Pressure: A Useful but Not so Simple Measurement: Critical Care Medicine 2006, 34, 2224–2227. [CrossRef]
- De Backer, D.; Aissaoui, N.; Cecconi, M.; Chew, M.S.; Denault, A.; Hajjar, L.; Hernandez, G.; Messina, A.; Myatra, S.N.; Ostermann, M.; et al. How Can Assessing Hemodynamics Help to Assess Volume Status? Intensive Care Med 2022, 48, 1482–1494. [CrossRef]
- Zampieri, F.G.; Bagshaw, S.M.; Semler, M.W. Fluid Therapy for Critically Ill Adults With Sepsis: A Review. JAMA 2023, 329, 1967. [CrossRef]
- Prezioso, C.; Trotta, R.; Cavallo, E.; Fusina, F.; Malpetti, E.; Albani, F.; Caserta, R.; Rosano, A.; Natalini, G. Central Venous Pressure and Dynamic Indices to Assess Fluid Appropriateness in Critically Ill Patients: A Pilot Study. PLoS ONE 2023, 18, e0285935. [CrossRef]
- Sondergaard, S.; Parkin, G.; Aneman, A. Central Venous Pressure: We Need to Bring Clinical Use into Physiological Context. Acta Anaesthesiol Scand 2015, 59, 552–560. [CrossRef]
- Magder, S. Central Venous Pressure Monitoring: Current Opinion in Critical Care 2006, 12, 219–227. [CrossRef]
- Magder, S. Understanding Central Venous Pressure: Not a Preload Index? Current Opinion in Critical Care 2015, 21, 369–375. [CrossRef]
- Pinsky, M.R. The Right Ventricle: Interaction with the Pulmonary Circulation. Crit Care 2016, 20, 266. [CrossRef]
- Umbrello, M.; Cereghini, S.; Muttini, S. Respiratory Variations of Central Venous Pressure as Indices of Pleural Pressure Swings: A Narrative Review. Diagnostics (Basel) 2023, 13, 1022. [CrossRef]
- Lassola, S.; Miori, S.; Sanna, A.; Menegoni, I.; De Rosa, S.; Bellani, G.; Umbrello, M. Assessment of Inspiratory Effort in Spontaneously Breathing COVID-19 ARDS Patients Undergoing Helmet CPAP: A Comparison between Esophageal, Transdiaphragmatic and Central Venous Pressure Swing. Diagnostics (Basel) 2023, 13, 1965. [CrossRef]
- Heerdt, P.M.; Kheyfets, V.O.; Oakland, H.T.; Joseph, P.; Singh, I. Right Ventricular Pressure Waveform Analysis-Clinical Relevance and Future Directions. J Cardiothorac Vasc Anesth 2024, 38, 2433–2445. [CrossRef]
- Cecconi, M.; De Backer, D.; Antonelli, M.; Beale, R.; Bakker, J.; Hofer, C.; Jaeschke, R.; Mebazaa, A.; Pinsky, M.R.; Teboul, J.L.; et al. Consensus on Circulatory Shock and Hemodynamic Monitoring. Task Force of the European Society of Intensive Care Medicine. Intensive Care Med 2014, 40, 1795–1815. [CrossRef]
- Berlin, D.A.; Bakker, J. Starling Curves and Central Venous Pressure. Crit Care 2015, 19, 55. [CrossRef]
- Gelman, S.; Warner, D.S.; Warner, M.A. Venous Function and Central Venous Pressure: A Physiologic Story. Anesthesiology 2008, 108, 735–748. [CrossRef]
- Guyton, A.C.; Jones, C.E. Central Venous Pressure: Physiological Significance and Clinical Implications. American Heart Journal 1973, 86, 431–437. [CrossRef]
- Ball, C.; Westhorpe, R.N. Central Venous Pressure Monitoring. Anaesth Intensive Care 2009, 37, 689–689. [CrossRef]
- Kelman, G.R. Interpretation of CVP Measurements. Anaesthesia 1971, 26, 209–215. [CrossRef]
- Marik, P.E.; Cavallazzi, R. Does the Central Venous Pressure Predict Fluid Responsiveness? An Updated Meta-Analysis and a Plea for Some Common Sense*: Critical Care Medicine 2013, 41, 1774–1781. [CrossRef]
- Marik, P.E. Surviving Sepsis: Going beyond the Guidelines. Ann. Intensive Care 2011, 1, 17. [CrossRef]
- Marik, P.E.; Baram, M.; Vahid, B. Does Central Venous Pressure Predict Fluid Responsiveness?*: A Systematic Review of the Literature and the Tale of Seven Mares. Chest 2008, 134, 172–178. [CrossRef]
- Osman, D.; Ridel, C.; Ray, P.; Monnet, X.; Anguel, N.; Richard, C.; Teboul, J.-L. Cardiac Filling Pressures Are Not Appropriate to Predict Hemodynamic Response to Volume Challenge*: Critical Care Medicine 2007, 35, 64–68. [CrossRef]
- Angappan, S.; Parida, S.; Vasudevan, A.; Badhe, A. The Comparison of Stroke Volume Variation with Central Venous Pressure in Predicting Fluid Responsiveness in Septic Patients with Acute Circulatory Failure. Indian Journal of Critical Care Medicine 2015, 19, 394–400. [CrossRef]
- Bentzer, P.; Griesdale, D.E.; Boyd, J.; MacLean, K.; Sirounis, D.; Ayas, N.T. Will This Hemodynamically Unstable Patient Respond to a Bolus of Intravenous Fluids? JAMA 2016, 316, 1298. [CrossRef]
- Chaves, R.C.D.F.; Barbas, C.S.V.; Queiroz, V.N.F.; Serpa Neto, A.; Deliberato, R.O.; Pereira, A.J.; Timenetsky, K.T.; Silva Júnior, J.M.; Takaoka, F.; De Backer, D.; et al. Assessment of Fluid Responsiveness Using Pulse Pressure Variation, Stroke Volume Variation, Plethysmographic Variability Index, Central Venous Pressure, and Inferior Vena Cava Variation in Patients Undergoing Mechanical Ventilation: A Systematic Review and Meta-Analysis. Crit Care 2024, 28, 289. [CrossRef]
- Michard, F.; Chemla, D.; Teboul, J.-L. Applicability of Pulse Pressure Variation: How Many Shades of Grey? Crit Care 2015, 19, 144. [CrossRef]
- Gambardella, I.; Gaudino, M.; Ronco, C.; Lau, C.; Ivascu, N.; Girardi, L.N. Congestive Kidney Failure in Cardiac Surgery: The Relationship between Central Venous Pressure and Acute Kidney Injury. Interact CardioVasc Thorac Surg 2016, 23, 800–805. [CrossRef]
- Rajendram, R.; Prowle, J.R. Venous Congestion: Are We Adding Insult to Kidney Injury in Sepsis? Crit Care 2014, 18, 104. [CrossRef]
- Rivers, E.; Nguyen, B.; Havstad, S.; Ressler, J.; Muzzin, A.; Knoblich, B.; Peterson, E.; Tomlanovich, M. Early Goal-Directed Therapy in the Treatment of Severe Sepsis and Septic Shock. N Engl J Med 2001, 345, 1368–1377. [CrossRef]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock. Critical Care Medicine 2017, 45, 486–552. [CrossRef]
- The ARISE Investigators and the ANZICS Clinical Trials Group Goal-Directed Resuscitation for Patients with Early Septic Shock. N Engl J Med 2014, 371, 1496–1506. [CrossRef]
- Mouncey, P.R.; Osborn, T.M.; Power, G.S.; Harrison, D.A.; Sadique, M.Z.; Grieve, R.D.; Jahan, R.; Harvey, S.E.; Bell, D.; Bion, J.F.; et al. Trial of Early, Goal-Directed Resuscitation for Septic Shock. N Engl J Med 2015, 372, 1301–1311. [CrossRef]
- The ProCESS Investigators A Randomized Trial of Protocol-Based Care for Early Septic Shock. N Engl J Med 2014, 370, 1683–1693. [CrossRef]
- Dellinger, R.P.; Schorr, C.A.; Levy, M.M. A Users’ Guide to the 2016 Surviving Sepsis Guidelines. Intensive Care Med 2017, 43, 299–303. [CrossRef]
- Rhodes, A.; Evans, L.E.; Alhazzani, W.; Levy, M.M.; Antonelli, M.; Ferrer, R.; Kumar, A.; Sevransky, J.E.; Sprung, C.L.; Nunnally, M.E.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 2017, 43, 304–377. [CrossRef]
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; Mcintyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Intensive Care Med 2021, 47, 1181–1247. [CrossRef]
- Prescott, H.C.; Antonelli, M.; Alhazzani, W.; Møller, M.H.; Alshamsi, F.; Azevedo, L.C.P.; Belley-Cote, E.; De Waele, J.; Derde, L.; Dionne, J.C.; et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2026. Critical Care Medicine 2026. [CrossRef]
- on behalf of the FENICE Investigators and the ESICM Trial Group; Cecconi, M.; Hofer, C.; Teboul, J.-L.; Pettila, V.; Wilkman, E.; Molnar, Z.; Della Rocca, G.; Aldecoa, C.; Artigas, A.; et al. Fluid Challenges in Intensive Care: The FENICE Study: A Global Inception Cohort Study. Intensive Care Med 2015, 41, 1529–1537. [CrossRef]
- Michard, F.; Teboul, J.-L. Predicting Fluid Responsiveness in ICU Patients. Chest 2002, 121, 2000–2008. [CrossRef]
- Lansdorp, B.; Lemson, J.; Van Putten, M.J.A.M.; De Keijzer, A.; Van Der Hoeven, J.G.; Pickkers, P. Dynamic Indices Do Not Predict Volume Responsiveness in Routine Clinical Practice. British Journal of Anaesthesia 2012, 108, 395–401. [CrossRef]
- La Via, L.; Vasile, F.; Perna, F.; Zawadka, M. Prediction of Fluid Responsiveness in Critical Care: Current Evidence and Future Perspective. Trends in Anaesthesia and Critical Care 2024, 54, 101316. [CrossRef]
- Chen, C.-Y.; Zhou, Y.; Wang, P.; Qi, E.-Y.; Gu, W.-J. Elevated Central Venous Pressure Is Associated with Increased Mortality and Acute Kidney Injury in Critically Ill Patients: A Meta-Analysis. Crit Care 2020, 24, 80. [CrossRef]
- De Backer, D.; Cecconi, M.; Chew, M.S.; Hajjar, L.; Monnet, X.; Ospina-Tascón, G.A.; Ostermann, M.; Pinsky, M.R.; Vincent, J.-L. A Plea for Personalization of the Hemodynamic Management of Septic Shock. Crit Care 2022, 26, 372. [CrossRef]
- Mullens, W.; Abrahams, Z.; Francis, G.S.; Sokos, G.; Taylor, D.O.; Starling, R.C.; Young, J.B.; Tang, W.H.W. Importance of Venous Congestion for Worsening of Renal Function in Advanced Decompensated Heart Failure. Journal of the American College of Cardiology 2009, 53, 589–596. [CrossRef]
- Damman, K.; Navis, G.; Smilde, T.D.J.; Voors, A.A.; Van Der Bij, W.; Van Veldhuisen, D.J.; Hillege, H.L. Decreased Cardiac Output, Venous Congestion and the Association with Renal Impairment in Patients with Cardiac Dysfunction. European Journal of Heart Failure 2007, 9, 872–878. [CrossRef]
- Lim, S.Y.; Kim, S. Pathophysiology of Cardiorenal Syndrome and Use of Diuretics and Ultrafiltration as Volume Control. Korean Circ J 2021, 51, 656. [CrossRef]
- Redant, S.; Honoré, P.M.; De Bels, D. Fifty Shades of Central Venous Pressure in the Cardiorenal Syndrome. J Transl Int Med 2020, 8, 1–2. [CrossRef]
- Khwannimit, B.; Koonrangsesomboon, W. Impact of Positive Fluid Balance on Mortality and Length of Stay in Septic Shock Patients. Indian Journal of Critical Care Medicine 2015, 19, 708–713. [CrossRef]
- Beaubien-Souligny, W.; Rola, P.; Haycock, K.; Bouchard, J.; Lamarche, Y.; Spiegel, R.; Denault, A.Y. Quantifying Systemic Congestion with Point-Of-Care Ultrasound: Development of the Venous Excess Ultrasound Grading System. Ultrasound J 2020, 12, 16. [CrossRef]
- Longino, A.A.; Martin, K.C.; Leyba, K.R.; McCormack, L.; Siegel, G.; Sharma, V.M.; Riscinti, M.; Lopez, C.O.; Douglas, I.S.; Gill, E.A. Reliability and Reproducibility of the Venous Excess Ultrasound (VExUS) Score, a Multi-Site Prospective Study: Validating a Novel Ultrasound Technique for Comprehensive Assessment of Venous Congestion. Crit Care 2024, 28, 197. [CrossRef]
- Cecconi, M.; Aya, H.D. Central Venous Pressure Cannot Predict Fluid-Responsiveness. Evid Based Med 2014, 19, 63–63. [CrossRef]
- Monnet, X.; Marik, P.E.; Teboul, J.-L. Prediction of Fluid Responsiveness: An Update. Ann. Intensive Care 2016, 6, 111. [CrossRef]
- Muñoz, F.; Born, P.; Bruna, M.; Ulloa, R.; González, C.; Philp, V.; Mondaca, R.; Blanco, J.P.; Valenzuela, E.D.; Retamal, J.; et al. Coexistence of a Fluid Responsive State and Venous Congestion Signals in Critically Ill Patients: A Multicenter Observational Proof-of-Concept Study. Crit Care 2024, 28, 52. [CrossRef]
- Marini, J.J.; Gattinoni, L. Ventilatory Management of Acute Respiratory Distress Syndrome: A Consensus of Two: Critical Care Medicine 2004, 32, 250–255. [CrossRef]
- Beitler, J.R.; Malhotra, A.; Thompson, B.T. Ventilator-Induced Lung Injury. Clinics in Chest Medicine 2016, 37, 633–646. [CrossRef]
- Chen, L.; Xia, H.-F.; Shang, Y.; Yao, S.-L. Molecular Mechanisms of Ventilator-Induced Lung Injury. Chinese Medical Journal 2018, 131, 1225–1231. [CrossRef]
- Sarge, T.; Talmor, D. Targeting Transpulmonary Pressure to Prevent Ventilator Induced Lung Injury. Minerva Anestesiol 2009, 75, 293–299.
- Talmor, D.; Sarge, T.; Malhotra, A.; O’Donnell, C.R.; Ritz, R.; Lisbon, A.; Novack, V.; Loring, S.H. Mechanical Ventilation Guided by Esophageal Pressure in Acute Lung Injury. N Engl J Med 2008, 359, 2095–2104. [CrossRef]
- Beitler, J.R.; Sarge, T.; Banner-Goodspeed, V.M.; Gong, M.N.; Cook, D.; Novack, V.; Loring, S.H.; Talmor, D.; for the EPVent-2 Study Group Effect of Titrating Positive End-Expiratory Pressure (PEEP) With an Esophageal Pressure–Guided Strategy vs an Empirical High PEEP-F io2 Strategy on Death and Days Free From Mechanical Ventilation Among Patients With Acute Respiratory Distress Syndrome: A Randomized Clinical Trial. JAMA 2019, 321, 846. [CrossRef]
- Kyogoku, M.; Takeuchi, M.; Inata, Y.; Okuda, N.; Shimizu, Y.; Hatachi, T.; Moon, K.; Tachibana, K. A Novel Method for Transpulmonary Pressure Estimation Using Fluctuation of Central Venous Pressure. J Clin Monit Comput 2020, 34, 725–731. [CrossRef]
- Franchi, F.; Detti, E.; Fogagnolo, A.; Spadaro, S.; Cevenini, G.; Cataldo, G.; Addabbo, T.; Biuzzi, C.; Marianello, D.; Volta, C.A.; et al. Estimation of the Transpulmonary Pressure from the Central Venous Pressure in Mechanically Ventilated Patients. J Clin Monit Comput 2024, 38, 847–858. [CrossRef]
- Avanzolini, G.; Barbini, P.; Cappello, A.; Cevenini, G. Real-Time Tracking of Parameters of Lung Mechanics: Emphasis on Algorithm Tuning. J. Biomed. Eng. 1990, 12, 489–495. [CrossRef]
- Addabbo, T.; Cataldo, G.; Cevenini, G.; Fort, A.; Franchi, F.; Moretti, R.; Mugnaini, M.; Scolletta, S.; Vignoli, V. A Measurement System to Estimate the Pleural Pressure From the CVP for Respiratory System Monitoring. IEEE Transactions on Instrumentation and Measurement 2019, 68, 2469–2478. [CrossRef]
- Okuda, N.; Kyogoku, M.; Inata, Y.; Isaka, K.; Moon, K.; Hatachi, T.; Shimizu, Y.; Takeuchi, M. Estimation of Change in Pleural Pressure in Assisted and Unassisted Spontaneous Breathing Pediatric Patients Using Fluctuation of Central Venous Pressure: A Preliminary Study. PLoS One 2021, 16, e0247360. [CrossRef]
- Kyogoku, M.; Mizuguchi, S.; Miyasho, T.; Endo, Y.; Inata, Y.; Tachibana, K.; Fujino, Y.; Yamashita, K.; Takeuchi, M. Estimating the Change in Pleural Pressure Using the Change in Central Venous Pressure in Various Clinical Scenarios: A Pig Model Study. Intensive Care Med Exp 2024, 12, 4. [CrossRef]
- Ogliari, G.; Mazzuca Mari, L.; Crisci, S.; Pradissitto, G.; Fusina, F.; Albani, F.; Morandi, A.; Bertelli, M.; Natalini, G. Relationship between Esophageal and Central Venous Pressure Variation during Respiratory Activity: The Best Choice among Four Different Methods to Measure the Amplitude of Central Venous Pressure Changes. J Clin Monit Comput 2026. [CrossRef]
- Longino, A.A.; Martin, K.C.; Douglas, I.S. Monitoring the Venous Circulation: Novel Techniques and Applications. Current Opinion in Critical Care 2024, 30, 260–267. [CrossRef]
 |
| Category | Specific Cause | Mechanism of Inaccuracy | Clinical Implication |
|---|---|---|---|
| Respiratory Factors | High PEEP/mechanical ventilation | Increase intrathoracic pressure → artificially elevates CVP | Overstimation of preload and volume status |
| Spontaneous breathing efforts | Negative intrathoracic pressure swings → decreases CVP | Underestimation of filling pressures | |
| Dynamic hyperinflation / auto-PEEP | Sustained elevation of intrathoracic pressure | Persistent CVP overestimation | |
| Poor timing of measurement (not at end-expiration) | Respiratory variations distort true value | Misleading single-point measurements | |
|
Cardiac Factors |
Tricuspid regurgitation | Systolic backflow → large v-waves, elevated mean CVP | Overestimation of right atrial pressure |
| Right ventricular dysfunction | Elevated RV filling pressures | High CVP unrelated to volume status | |
| Cardiac tamponade | Impaired filling → elevated and equalized pressures | High CVP despite low preload | |
| Constrictive pericarditis | Impaired diastolic filling | Elevated CVP with abnormal waveform | |
| Atrial fibrillation | Loss of a-wave | Difficult waveform interpretation | |
| Atrioventricular dissociation | Cannon a-waves | Intermittent CVP spikes | |
| Vascular Factors | Reduced venous compliance (↑ sympathetic tone) | Same volume → higher pressure | CVP overestimates volume |
| Increased intra-abdominal pressure | Impairs venous return → elevates CVP | False impression of volume overload | |
| Venous obstruction (e.g., SVC syndrome, thrombosis) | Impaired drainage → elevated upstream pressure | CVP not reflecting right atrial pressure | |
| Volume Status & Hemodynamics | Hypervolemia | True increase in venous pressure | May reflect congestion rather than preload reserve |
| Hypovolemia with high intrathoracic pressure | Opposing effects distort CVP | Unreliable assessment of volume status | |
| Redistribution of blood (venoconstriction) | Centralization of volume | Elevated CVP without true volume increase | |
| Technical Factors | Incorrect transducer leveling | Reference point error | Systematic over- or underestimation |
| Failure to zero to atmospheric pressure | Calibration error | Inaccurate absolute values | |
| Catheter malposition | Non-central measurement | Invalid CVP reading | |
| Air bubbles / clot in line | Signal damping or artifact | Distorted waveform and values | |
| External compression of catheter | Artificial pressure elevation | False high CVP | |
| Interpretation Errors | Use of single absolute value | Ignores dynamic and contextual factors | Misleading clinical decisions |
| Ignoring waveform morphology | Loss of diagnostic information | Missed cardiac pathology | |
| Using CVP to predict fluid responsiveness | Poor correlation with preload reserve | Inappropriate fluid administration |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




