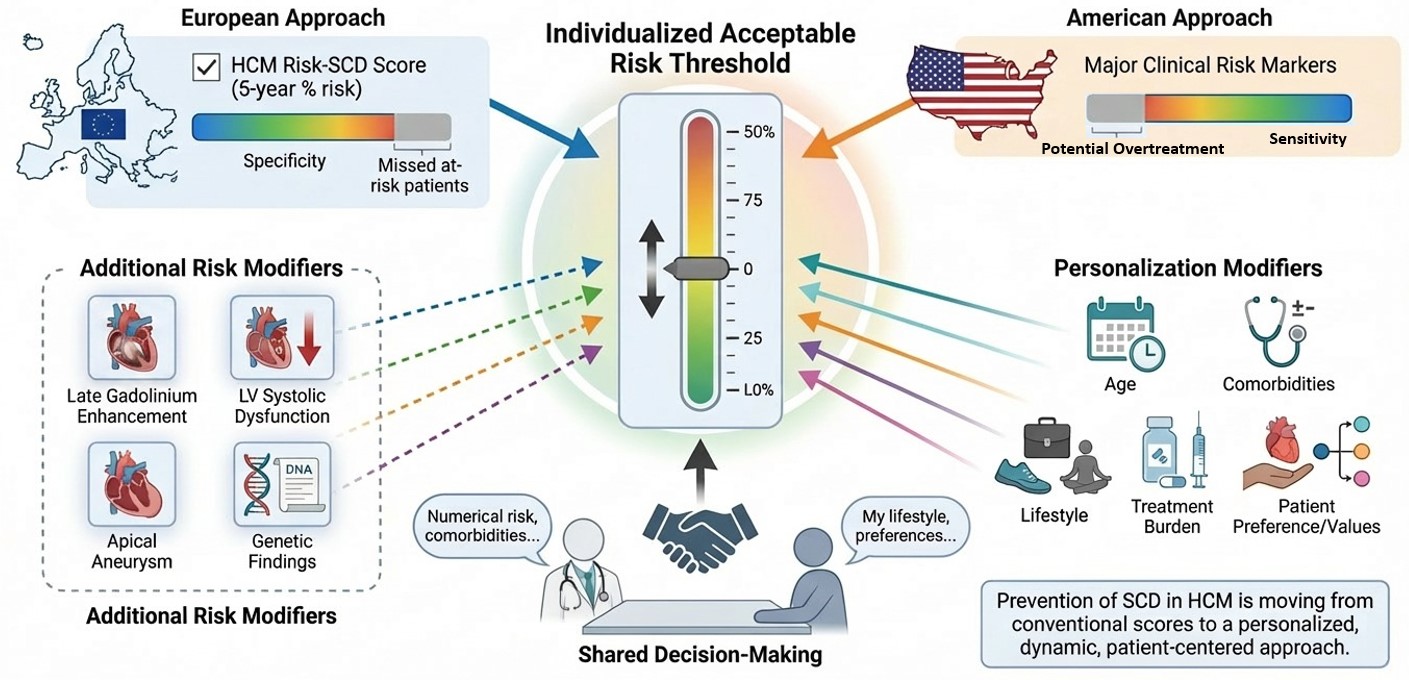
Hypertrophic Cardiomyopathy (HCM) is one of the most common inherited cardiomyopathies and remains an important cause of ventricular arrhythmias and Sudden Cardiac Death (SCD), particularly in younger individuals. Although the annual incidence of arrhythmic death is relatively low in contemporary cohorts, identifying those patients who may benefit from primary prevention with an Implantable Cardioverter-Defibrillator (ICD) remains a major clinical challenge. Current risk stratification strategies rely on two principal paradigms. The European approach is centered on the HCM Risk-SCD score, a multivariable model that provides an individualized 5-year risk estimate, whereas the American approach is mainly based on major clinical risk markers. Both strategies have important strengths and limitations, reflecting the persistent difficulty of accurately predicting arrhythmic events in such a heterogeneous disease. The HCM Risk-SCD score has demonstrated robust external validation and high specificity for identifying patients at higher risk, but it may fail to recognize some vulnerable individuals who remain below conventional treatment thresholds. For this reason, several additional risk modifiers have gained increasing relevance in contemporary practice. Among them, extensive late gadolinium enhancement, left ventricular systolic dysfunction, apical aneurysm, and clinically meaningful genetic findings may provide important incremental prognostic information beyond traditional models. Emerging disease-modifying therapies may also influence future risk assessment. In particular, Mavacamten has shown substantial reduction of left ventricular outflow tract obstruction together with favorable reverse remodeling, including changes in variables incorporated into conventional risk scores. However, whether these improvements translate into a true reduction in SCD risk remains uncertain. Importantly, the decision to implant an ICD should not depend on numerical risk alone. It should arise from a process of shared decision-making integrating estimated risk, treatment burden, competing comorbidities, age, lifestyle, and patient values. In this context, the concept of an individualized threshold of “acceptable risk” becomes central. In conclusion, prevention of SCD in HCM is moving beyond conventional scores toward a personalized and dynamic framework in which predictive tools, advanced phenotyping, evolving therapies, clinical expertise, and patient preferences are combined to guide individualized care.