Submitted:
01 May 2026
Posted:
04 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
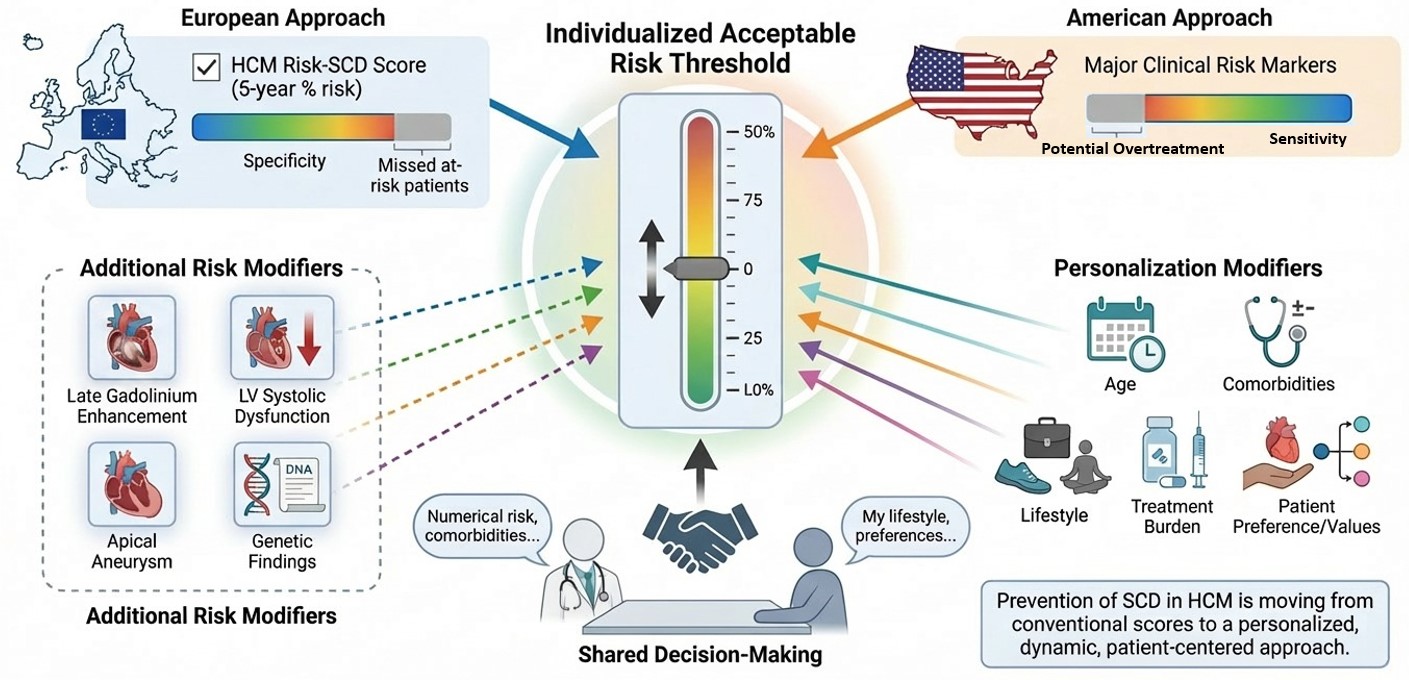
2. Defining the Threshold of “Acceptable Risk”
3. Two Different Perspectives: United States vs Europe
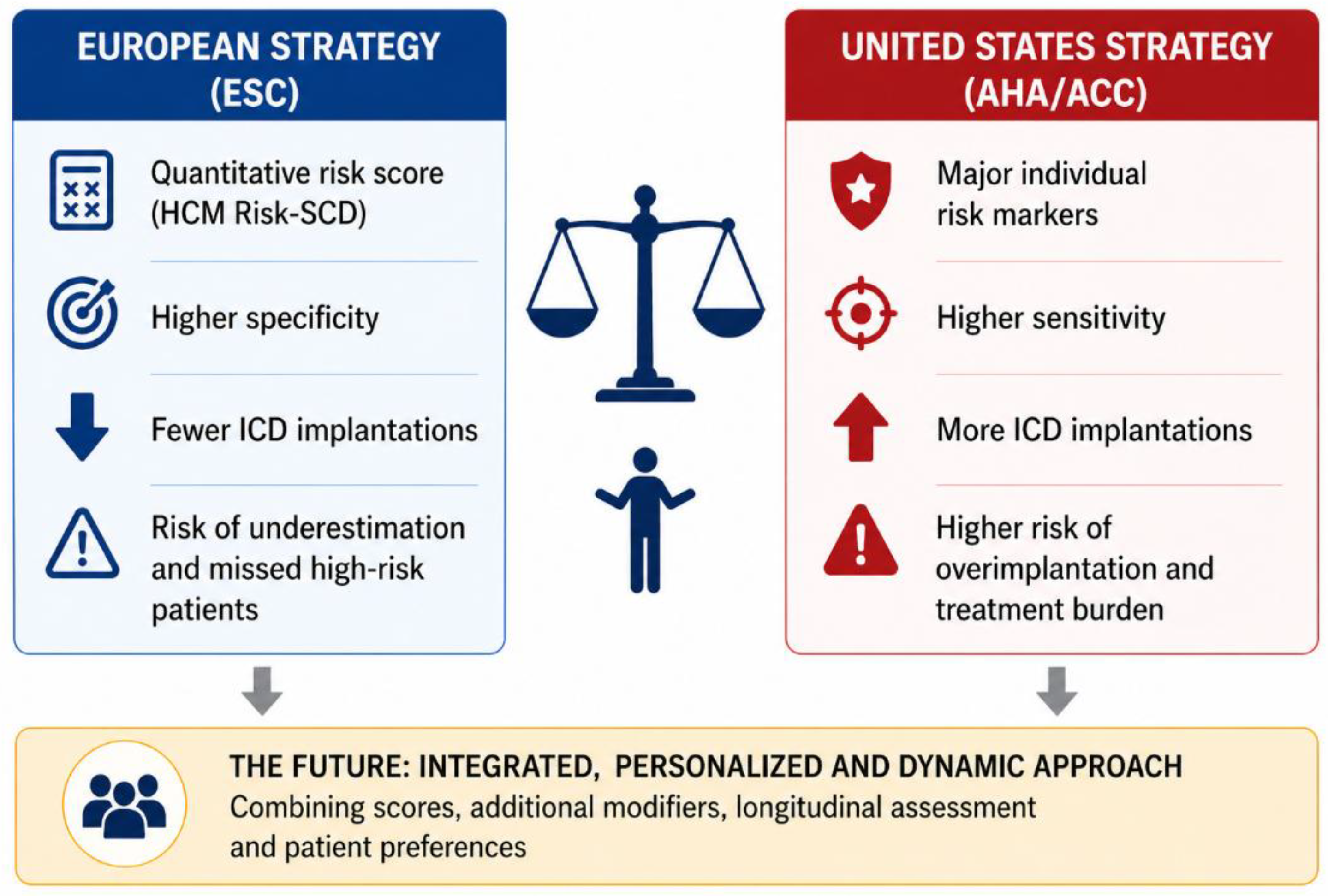
4. HCM Risk-SCD Score: Strengths, Limitations, and Clinical Interpretation
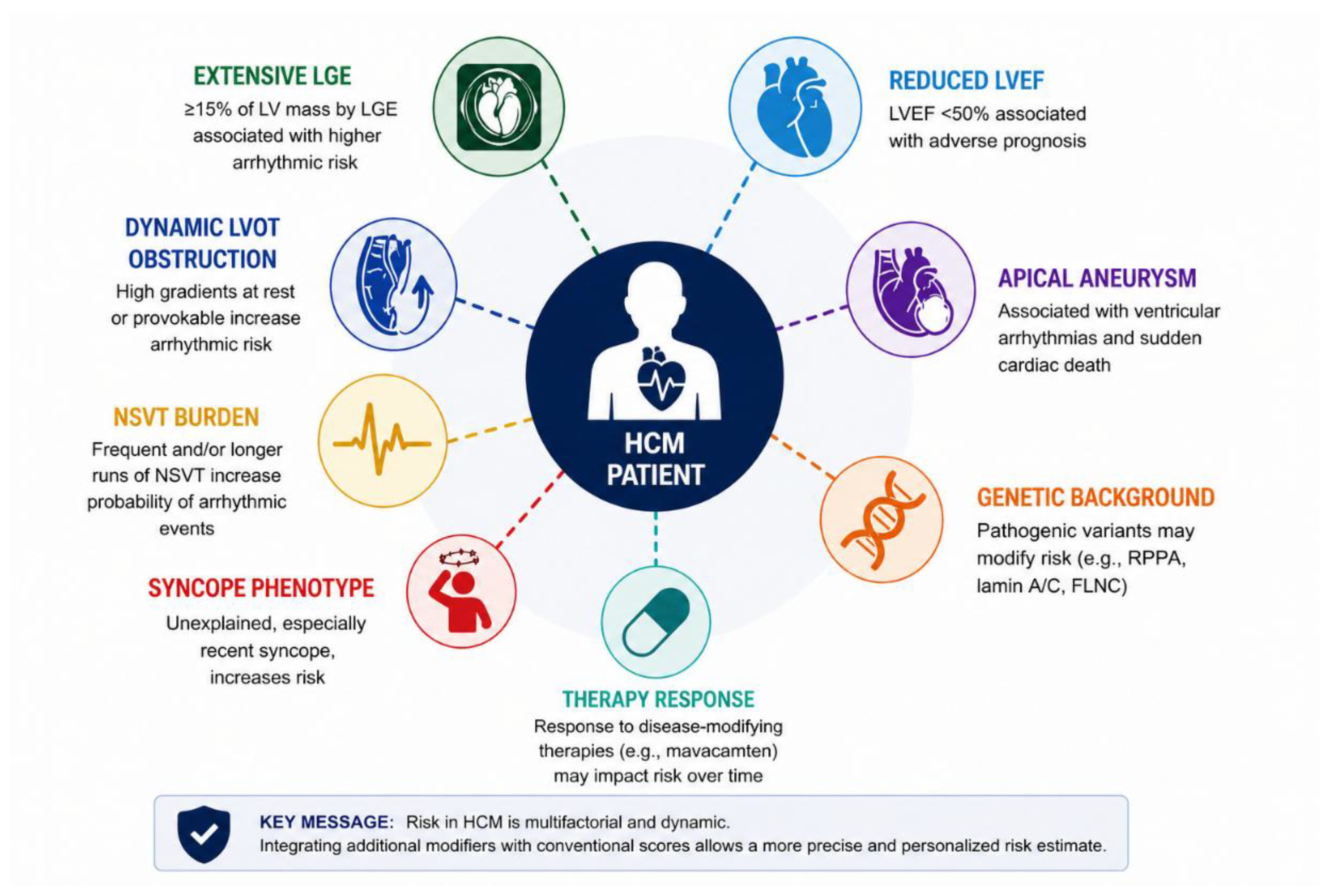
5. Additional Risk Modifiers Beyond the Score
5.1. Apical Aneurism
5.2. Late Gadolinium Enhancement
5.3. Left Ventricular Systolic Dysfunction
5.4. Abnormal Blood Pressure Response to Exercise
5.5. Genetics
5.6. The Emerging Role of Mavacamten
6. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACC | American College of Cardiology |
| AHA | American Heart Association |
| CMR | Cardiac Magnetic Resonance |
| ESC | European Society of Cardiology |
| HCM | Hypertrophic Cardiomyopathy |
| HF | Heart Failure |
| HR | Hazard Ratio |
| ICD | Implantable Cardioverter-Defibrillator |
| LA | Left Atrium / Left Atrial |
| LGE | Late Gadolinium Enhancement |
| LV | Left Ventricle / Left Ventricular |
| LVEF | Left Ventricular Ejection Fraction |
| LVOT | Left Ventricular Outflow Tract |
| LVOTO | Left Ventricular Outflow Tract Obstruction |
| NSVT | Non-Sustained Ventricular Tachycardia |
| ROC | Receiver Operating Characteristic |
| SCD | Sudden Cardiac Death |
| SD | Standard Deviation |
| SHaRe | Sarcomeric Human Cardiomyopathy Registry |
| VF | Ventricular Fibrillation |
| VT | Ventricular Tachycardia |
References
- Maron BJ. Clinical Course and Management of Hypertrophic Cardiomyopathy. N Engl J Med. 2018;379(7):655-668. [CrossRef]
- Marian AJ, Braunwald E. Hypertrophic Cardiomyopathy: Genetics, Pathogenesis, Clinical Manifestations, Diagnosis, and Therapy. Circ Res. 2017;121(7):749-770. [CrossRef]
- Burke MA, Cook SA, Seidman JG, Seidman CE. Clinical and Mechanistic Insights Into the Genetics of Cardiomyopathy. J Am Coll Cardiol. 2016;68(25):2871-2886. [CrossRef]
- Semsarian C, Ingles J, Maron MS, Maron BJ. New perspectives on the prevalence of hypertrophic cardiomyopathy. J Am Coll Cardiol. 2015;65(12):1249-1254. [CrossRef]
- Arbelo E, Protonotarios A, Gimeno JR, et al. 2023 ESC Guidelines for the management of cardiomyopathies. Eur Heart J. 2023;44(37):3503-3626. [CrossRef]
- Ommen SR, Ho CY, Asif IM, et al. 2024 AHA/ACC/AMSSM/HRS/PACES/SCMR Guideline for the Management of Hypertrophic Cardiomyopathy: A Report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2024;149(23):e1239-e1311. [CrossRef]
- Lorenzini M, Anastasiou Z, O’Mahony C, et al. Mortality Among Referral Patients With Hypertrophic Cardiomyopathy vs the General European Population. JAMA Cardiol. 2020;5(1):73-80. [CrossRef]
- Norrish G, Chubb H, Field E, et al. Clinical outcomes and programming strategies of implantable cardioverter-defibrillator devices in paediatric hypertrophic cardiomyopathy: a UK National Cohort Study. Europace. 2021;23(3):400-408. [CrossRef]
- O’Mahony C, Jichi F, Pavlou M, et al. A novel clinical risk prediction model for sudden cardiac death in hypertrophic cardiomyopathy (HCM risk-SCD). Eur Heart J. 2014;35(30):2010-2020. [CrossRef]
- Maron, M.S.; Rowin, E.J.; Wessler, B.S.; Mooney, P.J.; Fatima, A.; Patel, P.; Koethe, B.C.; Romashko, M.; Link, M.S.; Maron, B.J. Enhanced American College of Cardiology/American Heart Association Strategy for Prevention of Sudden Cardiac Death in High-Risk Patients with Hypertrophic Cardiomyopathy. JAMA Cardiol. 2019, 4, 644–657.
- Zegkos, T.; Tziomalos, G.; Parcharidou, D.; Ntelios, D.; Papanastasiou, C.A.; Karagiannidis, E.; Gossios, T.; Rouskas, P.; Katranas, S.; Paraskevaidis, S.; et al. Validation of the New American College of Cardiology/American Heart Association Guidelines for the Risk Stratification of Sudden Cardiac Death in a Large Mediterranean Cohort with Hypertrophic Cardiomyopathy. Hellenic J. Cardiol. 2022, 63, 15–21.
- Leong, K.M.W.; Chow, J.-J.; Ng, F.S.; Falaschetti, E.; Qureshi, N.; Koa-Wing, M.; Linton, N.W.F.; Whinnett, Z.I.; Lefroy, D.C.; Davies, D.W.; et al. Comparison of the Prognostic Usefulness of the European Society of Cardiology and American Heart Association/American College of Cardiology Foundation Risk Stratification Systems for Patients with Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2018, 121, 349–355.
- Nauffal V, Marstrand P, Han L, et al. Worldwide differences in primary prevention implantable cardioverter defibrillator utilization and outcomes in hypertrophic cardiomyopathy. Eur Heart J. 2021;42(38):3932-3944. [CrossRef]
- O’Mahony C, Jichi F, Ommen SR, et al. International External Validation Study of the 2014 European Society of Cardiology Guidelines on Sudden Cardiac Death Prevention in Hypertrophic Cardiomyopathy (EVIDENCE-HCM). Circulation. 2018;137(10):1015-1023. [CrossRef]
- O’Mahony C, Akhtar MM, Anastasiou Z, et al. Effectiveness of the 2014 European Society of Cardiology guideline on sudden cardiac death in hypertrophic cardiomyopathy: a systematic review and meta-analysis. Heart. 2019;105(8):623-631. [CrossRef]
- Maron BJ, Casey SA, Chan RH, Garberich RF, Rowin EJ, Maron MS. Independent Assessment of the European Society of Cardiology Sudden Death Risk Model for Hypertrophic Cardiomyopathy. Am J Cardiol. 2015;116(5):757-764. [CrossRef]
- Spirito, P.; Autore, C.; Rapezzi, C.; Bernabò, P.; Badagliacca, R.; Maron, M.S.; Bongioanni, S.; Coccolo, F.; Estes, N.A.M.; Barillà, C.S.; et al. Syncope and Risk of Sudden Death in Hypertrophic Cardiomyopathy. Circulation 2009, 119, 1703–1710.
- Mascia G, Crotti L, Groppelli A, et al. Syncope in hypertrophic cardiomyopathy (part I): An updated systematic review and meta-analysis. Int J Cardiol. 2022;357:88-94. [CrossRef]
- Brignole M, Cecchi F, Anastasakis A, et al. Syncope in hypertrophic cardiomyopathy (part II): An expert consensus statement on the diagnosis and management. Int J Cardiol. 2023;370:330-337. [CrossRef]
- Monserrat L, Elliott PM, Gimeno JR, Sharma S, Penas-Lado M, McKenna WJ. Non-sustained ventricular tachycardia in hypertrophic cardiomyopathy: an independent marker of sudden death risk in young patients. J Am Coll Cardiol. 2003;42(5):873-879. [CrossRef]
- Viswanathan K, Suszko AM, DAS M, et al. Rapid Device-Detected Nonsustained Ventricular Tachycardia in the Risk Stratification of Hypertrophic Cardiomyopathy. Pacing Clin Electrophysiol. 2016;39(7):642-651. [CrossRef]
- Spirito P, Bellone P, Harris KM, Bernabo P, Bruzzi P, Maron BJ. Magnitude of left ventricular hypertrophy and risk of sudden death in hypertrophic cardiomyopathy. N Engl J Med. 2000;342(24):1778-1785. [CrossRef]
- Elliott PM, Gimeno JR, Tomé MT, et al. Left ventricular outflow tract obstruction and sudden death risk in patients with hypertrophic cardiomyopathy. Eur Heart J. 2006;27(16):1933-1941. [CrossRef]
- Rowin, E.J.; Maron, B.J.; Haas, T.S.; Garberich, R.F.; Wang, W.; Link, M.S.; Maron, M.S. Hypertrophic Cardiomyopathy with Left Ventricular Apical Aneurysm. J. Am. Coll. Cardiol.
- Neubauer, S.; Kolm, P.; Ho, C.Y.; Kwong, R.Y.; Desai, M.Y.; Dolman, S.F.; Appelbaum, E.; Desvigne-Nickens, P.; DiMarco, J.P.; Friedrich, M.G.; et al. Distinct Subgroups in Hypertrophic Cardiomyopathy in the NHLBI HCM Registry. J. Am. Coll. Cardiol. 2019, 74, 2333–2345.
- Maron, M.S.; Finley, J.J.; Bos, J.M.; Hauser, T.H.; Manning, W.J.; Haas, T.S.; Lesser, J.R.; Udelson, J.E.; Ackerman, M.J.; Maron, B.J. Prevalence, Clinical Significance, and Natural History of Left Ventricular Apical Aneurysms in Hypertrophic Cardiomyopathy. Circulation 2008, 118, 1541–1549.
- Ichida M, Nishimura Y, Kario K. Clinical significance of left ventricular apical aneurysms in hypertrophic cardiomyopathy patients: the role of diagnostic electrocardiography. J Cardiol. 2014;64(4):265-272. [CrossRef]
- Alfonso, F.; Frenneaux, M.P.; McKenna, W.J. Clinical Sustained Uniform Ventricular Tachycardia in Hypertrophic Cardiomyopathy: Association with Left Ventricular Apical Aneurysm. Heart 1989, 61, 178–181.
- Papanastasiou, C.A.; Zegkos, T.; Karamitsos, T.D.; Rowin, E.J.; Maron, M.S.; Parcharidou, D.; Kokkinidis, D.G.; Karvounis, H.; Rimoldi, O.; Maron, B.J.; et al. Prognostic Role of Left Ventricular Apical Aneurysm in Hypertrophic Cardiomyopathy: A Systematic Review and Meta-Analysis. Int. J. Cardiol.
- Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2022;43(40):3997-4126. [CrossRef]
- Lorenzini M, Elliott PM. Do apical aneurysms predict sudden cardiac death in hypertrophic cardiomyopathy?. Eur Heart J. 2023;44(17):1519-1521. [CrossRef]
- Rowin EJ, Maron BJ, Haas TS, Garberich RF, Wang W, Link MS, et al. Hypertrophic cardiomyopathy with left ventricular apical aneurysm: implications for risk stratification and management. J Am Coll Cardiol 2017;69:761–77.
- Kamp NJ, Chery G, Kosinski AS, et al. Risk stratification using late gadolinium enhancement on cardiac magnetic resonance imaging in patients with hypertrophic cardiomyopathy: A systematic review and meta-analysis. Prog Cardiovasc Dis. 2021;66:10-16. [CrossRef]
- Raiker, N.; Vullaganti, S.; Collins, J.D.; Allen, B.D.; Choudhury, L. Myocardial Tissue Characterization by Gadolinium-Enhanced Cardiac Magnetic Resonance Imaging for Risk Stratification of Adverse Events in Hypertrophic Cardiomyopathy. Int. J. Cardiovasc. Imaging 2020, 36, 1147–1156.
- Kariki, O.; Antoniou, C.-K.; Mavrogeni, S.; Gatzoulis, K.A. Updating the Risk Stratification for Sudden Cardiac Death in Cardiomyopathies: The Evolving Role of Cardiac Magnetic Resonance Imaging. An Approach for the Electrophysiologist. Diagnostics 2020, 10, 541.
- Maron, M.S. Clinical Utility of Cardiovascular Magnetic Resonance in Hypertrophic Cardiomyopathy. J. Cardiovasc. Magn. Reson. 2012, 14, 7.
- Flett AS, Hasleton J, Cook C, et al. Evaluation of techniques for the quantification of myocardial scar of differing etiology using cardiac magnetic resonance. JACC Cardiovasc Imaging. 2011;4(2):150-156. [CrossRef]
- Kiaos A, Daskalopoulos GN, Kamperidis V, Ziakas A, Efthimiadis G, Karamitsos TD. Quantitative Late Gadolinium Enhancement Cardiac Magnetic Resonance and Sudden Death in Hypertrophic Cardiomyopathy: A Meta-Analysis. JACC Cardiovasc Imaging. 2024;17(5):489-497. [CrossRef]
- Habib, M.; Adler, A.; Fardfini, K.; Hoss, S.; Hanneman, K.; Rowin, E.J.; Maron, M.S.; Maron, B.J.; Rakowski, H.; Chan, R.H. Progression of Myocardial Fibrosis in Hypertrophic Cardiomyopathy: A Cardiac Magnetic Resonance Study. JACC Cardiovasc. Imaging 2021, 14, 947–958.
- Klopotowski, M.; Kukula, K.; Malek, L.A.; Spiewak, M.; Polanska-Skrzypczyk, M.; Jamiolkowski, J.; Dabrowski, M.; Baranowski, R.; Klisiewicz, A.; Kusmierczyk, M.; et al. The Value of Cardiac Magnetic Resonance and Distribution of Late Gadolinium Enhancement for Risk Stratification of Sudden Cardiac Death in Patients with Hypertrophic Cardiomyopathy. J. Cardiol. 2016, 68, 49–56.
- Bravo, P.E.; Luo, H.-C.; Pozios, I.; Zimmerman, S.L.; Corona-Villalobos, C.P.; Sorensen, L.; Kamel, I.R.; Bluemke, D.A.; Wahl, R.L.; Abraham, M.R.; et al. Late Gadolinium Enhancement Confined to the Right Ventricular Insertion Points in Hypertrophic Cardiomyopathy: An Intermediate Stage Phenotype? Eur. Heart J. Cardiovasc. Imaging 2016, 17, 293–300.
- Rowin EJ, Maron BJ, Carrick RT, et al. Outcomes in Patients With Hypertrophic Cardiomyopathy and Left Ventricular Systolic Dysfunction. J Am Coll Cardiol. 2020;75(24):3033-3043. [CrossRef]
- Wasserstrum, Y.; Larrañaga-Moreira, J.M.; Martinez-Veira, C.; Itelman, E.; Lotan, D.; Sabbag, A.; Kuperstein, R.; Peled, Y.; Freimark, D.; Barriales-Villa, R.; et al. Hypokinetic Hypertrophic Cardiomyopathy: Clinical Phenotype, Genetics, and Prognosis. ESC Heart Fail. 2022, 9, 2301–2312.
- Marstrand, P.; Han, L.; Day, S.M.; Olivotto, I.; Ashley, E.A.; Michels, M.; Pereira, A.C.; Wittekind, S.G.; Helms, A.; Saberi, S.; et al. Hypertrophic Cardiomyopathy with Left Ventricular Systolic Dysfunction. Circulation 2020, 141, 1371–1383.
- Choi, Y.-J.; Kim, H.-K.; Hwang, I.-C.; Park, C.S.; Rhee, T.-M.; Lee, H.-J.; Park, J.-B.; Yoon, Y.E.; Lee, S.-P.; Cho, G.-Y.; et al. Prognosis of Patients with Hypertrophic Cardiomyopathy and Low-Normal Left Ventricular Ejection Fraction. Heart 2023, 109, 771–778.
- Olivotto, I.; Maron, B.J.; Appelbaum, E.; Harrigan, C.J.; Salton, C.; Gibson, C.M.; Udelson, J.E.; O’Donnell, C.; Lesser, J.R.; Manning, W.J.; et al. Spectrum and Clinical Significance of Systolic Function and Myocardial Fibrosis Assessed by Cardiovascular Magnetic Resonance in Hypertrophic Cardiomyopathy. Am. J. Cardiol. 2010, 106, 261–267.
- Gimeno JR, Tome-Esteban M, Lofiego C, Hurtado J, Pantazis A, Mist B, et al. Exercise-induced ventricular arrhythmias and risk of sudden cardiac death in patients with hypertrophic cardiomyopathy. Eur Heart J 2009;30:2599–2605.
- Elliott PM, Poloniecki J, Dickie S, Sharma S, Monserrat L, Varnava A, et al. Sudden death in hypertrophic cardiomyopathy: identification of high risk patients. J Am Coll Cardiol 2000;36:2212–2218.
- Sadoul N, Prasad K, Elliott PM, Bannerjee S, Frenneaux MP, McKenna WJ. Prospective prognostic assessment of blood pressure response during exercise in patients with hypertrophic cardiomyopathy. Circulation 1997;96:2987–2991. [CrossRef]
- Frenneaux MP, Counihan PJ, Caforio AL, Chikamori T, McKenna WJ. Abnormal blood pressure response during exercise in hypertrophic cardiomyopathy. Circulation. 1990;82(6):1995-2002. [CrossRef]
- Counihan PJ, Frenneaux MP, Webb DJ, McKenna WJ. Abnormal vascular responses to supine exercise in hypertrophic cardiomyopathy. Circulation 1991;84:686–696.
- Smith ED, Tome J, McGrath R, Kumar S, Concannon M, Day SM, et al. Exercise hemodynamics in hypertrophic cardiomyopathy identify risk of incident heart failure but not ventricular arrhythmias or sudden cardiac death. Int J Cardiol 2019;274:226–231.
- Norrish G, Cantarutti N, Pissaridou E, Ridout DA, Limongelli G, Elliott PM, et al. Risk factors for sudden cardiac death in childhood hypertrophic cardiomyopathy: a systematic review and meta-analysis. Eur J Prev Cardiol 2017;24:1220–1230.
- Ho CY, Day SM, Ashley EA, et al. Genotype and Lifetime Burden of Disease in Hypertrophic Cardiomyopathy: Insights from the Sarcomeric Human Cardiomyopathy Registry (SHaRe). Circulation. 2018;138(14):1387-1398. [CrossRef]
- van Velzen HG, Vriesendorp PA, Oldenburg RA, et al. Value of Genetic Testing for the Prediction of Long-Term Outcome in Patients With Hypertrophic Cardiomyopathy. Am J Cardiol. 2016;118(6):881-887. [CrossRef]
- Neubauer S, Kolm P, Ho CY, et al. Distinct Subgroups in Hypertrophic Cardiomyopathy in the NHLBI HCM Registry. J Am Coll Cardiol. 2019;74(19):2333-2345. [CrossRef]
- García-Giustiniani D, Arad M, Ortíz-Genga M, et al. Phenotype and prognostic correlations of the converter region mutations affecting the β myosin heavy chain. Heart. 2015;101(13):1047-1053. [CrossRef]
- Varnava AM, Elliott PM, Baboonian C, Davison F, Davies MJ, McKenna WJ. Hypertrophic cardiomyopathy: histopathological features of sudden death in cardiac troponin T disease. Circulation. 2001;104(12):1380-1384. [CrossRef]
- Jeschke B, Uhl K, Weist B, Schroder D, Meitinger T, Dohlemann C, et al. A high risk phenotype of hypertrophic cardiomyopathy associated with a compound genotype of two mutated beta-myosin heavy chain genes. Hum Genet 1998;102:299–304.
- Zheng, S.L.; Jurgens, S.J.; McGurk, K.A.; Xu, X.; Grace, C.; Theotokis, P.I.; Buchan, R.J.; Francis, C.; de Marvao, A.; Curran, L.; et al. Evaluation of Polygenic Scores for Hypertrophic Cardiomyopathy in the General Population and across Clinical Settings. Nat. Genet. 2025, 57, 563–571.
- Olivotto I, Oreziak A, Barriales-Villa R, et al. Mavacamten for treatment of symptomatic obstructive hypertrophic cardiomyopathy (EXPLORER-HCM): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2020;396(10253):759-769. [CrossRef]
- Alfarano M, Ciccarelli F, Marchionni G, et al. Hypertrophic Cardiomyopathy and Phenocopies: New Therapies for Old Diseases-Current Evidence and Future Perspectives. J Clin Med. 2025;14(12):4228. Published 2025 Jun 13. [CrossRef]
- Saberi S, Cardim N, Yamani M, et al. Mavacamten Favorably Impacts Cardiac Structure in Obstructive Hypertrophic Cardiomyopathy: EXPLORER-HCM Cardiac Magnetic Resonance Substudy Analysis. Circulation. 2021;143(6):606-608. [CrossRef]
- Hegde SM, Lester SJ, Solomon SD, et al. Effect of Mavacamten on Echocardiographic Features in Symptomatic Patients With Obstructive Hypertrophic Cardiomyopathy. J Am Coll Cardiol. 2021;78(25):2518-2532. [CrossRef]
- Desai MY, Okushi Y, Wolski K, et al. Long-Term Favorable Cardiac Remodeling in Obstructive Hypertrophic Cardiomyopathy Patients Treated With Mavacamten for Up to 128 Weeks: Insights From the VALOR-HCM Trial. JACC Cardiovasc Imaging. 2025;18(12):1300-1311. [CrossRef]
- Marchionni G, Alfarano M, Ballatore F, Costantino J, Chimenti C. Complete left bundle branch block resolution during treatment with Mavacamten: a case report of a first-in-class scenario. Eur Heart J Case Rep. 2025;9(12):ytaf592. Published 2025 Nov 20. [CrossRef]
- J Costantino, G Marchionni, F Ballatore, M Alfarano, C D Vizza, C Chimenti, Effect of Mavacamten on reducing sudden cardiac death risk in patients with Obstructive Hypertrophic Cardiomyopathy: an evaluation using the HCM-SCD Risk Score, EP Europace, Volume 27, Issue Supplement_1, May 2025, euaf085.717,.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



