Submitted:
30 April 2026
Posted:
01 May 2026
You are already at the latest version
Abstract
Keywords:
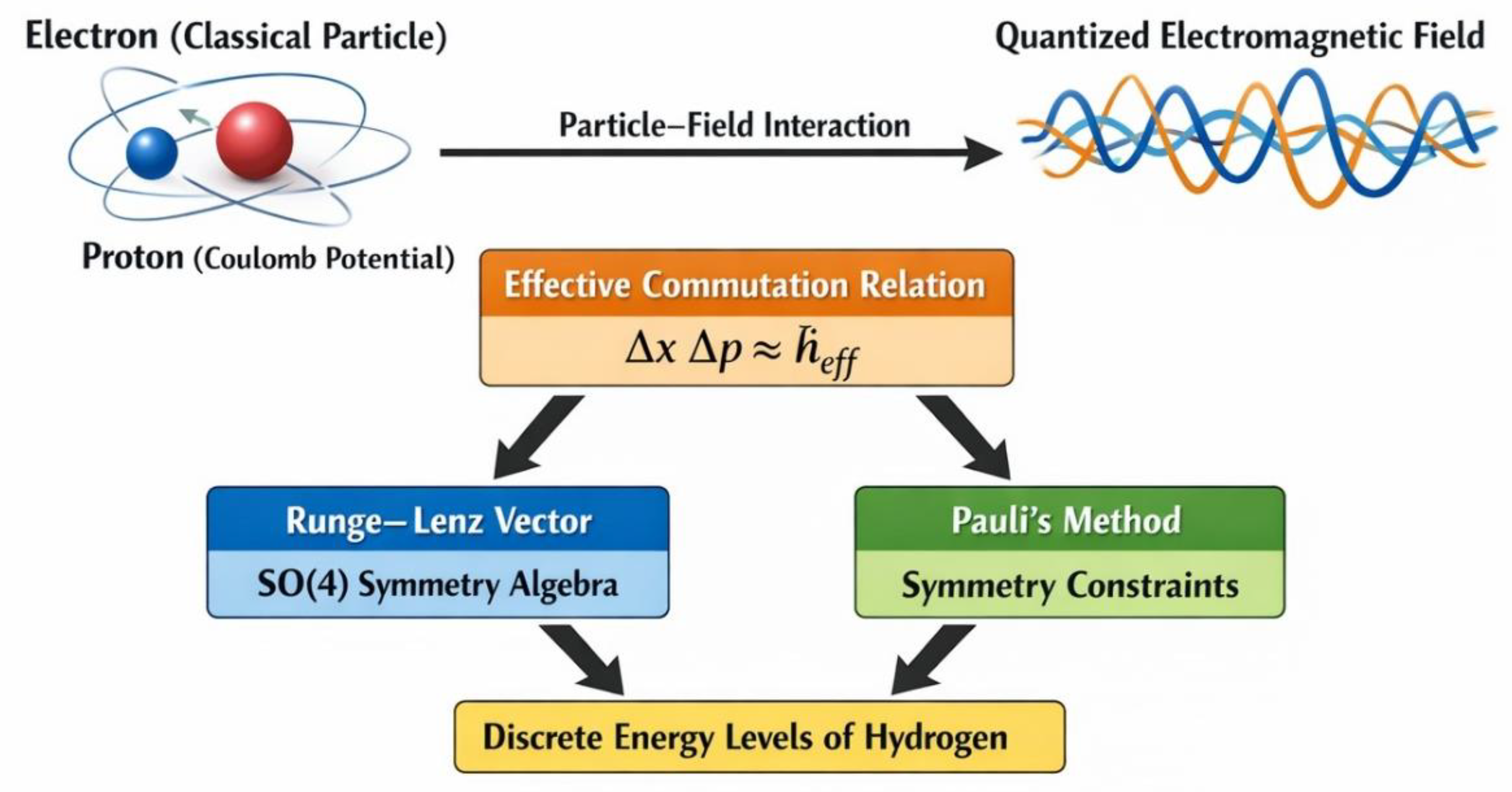
1. Introduction
2. Theoretical Framework
2.1. Semi-Quantum Hamiltonian for the Hydrogen System
2.2. Runge–Lenz Symmetry of the Coulomb System
2.3. Algebra of Conserved Quantities
2.4. Emergent Quantum Algebra from Particle–Field Interaction
2.5. Hydrogen Energy Spectrum
3. Physical Interpretation of Emergent Atomic Quantization
4. Discussion, Conclusions, and Outlook
Outlook
Conflict of Interest Statement
Funding
Data Availability Statement
Appendix A. Runge–Lenz Algebra and SO(4) Symmetry of the Coulomb System
Appendix A.1. Conserved Quantities of the Coulomb System
- 1)
- Angular momentum
- 2)
- Runge–Lenz vector
Appendix A.2. Poisson Bracket Algebra
Appendix A.3. Emergent Quantum Commutation Structure
Appendix A.4. SO(4) Symmetry and Energy Quantization
Appendix A.5. Role of Particle–Field Interaction
Appendix B. How Classical Electrons Acquire Matter-Wave Duality via Quantized Field Interactions
Appendix B.1. Classical Electron Dynamics via Poisson Brackets
Appendix B.2. Quantized Electromagnetic Field and Minimal Coupling
Appendix B.2.1 Quantized Vector Potential in Coulomb Gauge
Appendix B.2.2. Minimal Coupling and the Interaction Hamiltonian
Appendix B.3. Stochastic Momentum Transfer from the Quantized Field
Appendix B.3.1. Transverse Equation of Motion
Appendix B.3.2. Effective Stochastic Force from Field Modes
Appendix B.3.3. Accumulated Transverse Displacement
Appendix B.4.1. Fokker–Planck Equation for Momentum Diffusion
Appendix B.4.2. Hydrodynamic Form: Continuity and Momentum Equations
- 1)
- Continuity equation:
- 2)
- Momentum balance (neglecting external potential):
Appendix B.4.3. Emergence of Quantum Potential
Appendix B.4.4. Madelung Transformation and Schrödinger Equation
References
- Dirac, P.A.M. The Principles of Quantum Mechanics.; Oxford University Press: Oxford, 1930. [Google Scholar]
- de Broglie, L. Recherches sur la théorie des quanta. Ann. De Phys. 1925, 10, 22–128. [Google Scholar] [CrossRef]
- Schrödinger, E. Quantisierung als Eigenwertproblem. Ann. Der Phys. 1926, 79, 361–376. [Google Scholar] [CrossRef]
- Heisenberg, W. Über quantentheoretische Umdeutung kinematischer und mechanischer Beziehungen. Z. Für Phys. 1925, 33, 879–893. [Google Scholar] [CrossRef]
- Dirac, P.A.M. The Fundamental Equations of Quantum Mechanics. Proc. R. Soc. A 1925, 109, 642–653. [Google Scholar] [CrossRef]
- Bohr, N. The Quantum Postulate and the Recent Development of Atomic Theory. Nature 1928, 121, 580–590. [Google Scholar] [CrossRef]
- Feynman, R.P.; Leighton, R.B.; Sands, M. The Feynman Lectures on Physics, Vol. III: Quantum Mechanics.; Addison-Wesley: Reading, MA, 1965. [Google Scholar]
- Tonomura, A.; Endo, J.; Matsuda, T.; Kawasaki, T.; Ezawa, H. Demonstration of Single-Electron Buildup of an Interference Pattern. Am. J. Phys. 1989, 57, 117–120. [Google Scholar] [CrossRef]
- von Neumann, J. Mathematical Foundations of Quantum Mechanics.; Princeton University Press: Princeton, 1932. [Google Scholar]
- Everett, H. Relative State Formulation of Quantum Mechanics. Rev. Mod. Phys. 1957, 29, 454–462. [Google Scholar] [CrossRef]
- Balmer, J.J. Notiz über die Spectrallinien des Wasserstoffs. Ann. Der Phys. Und Chem. 1885, 25, 80–87. [Google Scholar] [CrossRef]
- Griffiths, D.J. Introduction to Quantum Mechanics., 2nd ed.; Pearson Prentice Hall: Upper Saddle River, NJ, 2005. [Google Scholar]
- Bohr, N. On the Constitution of Atoms and Molecules. Philos. Mag. 1913, 26, 1–25. [Google Scholar] [CrossRef]
- Runge, C. Vektoranalysis und höhere Mechanik. Z. Für Math. Und Phys. 1903, 48, 441–451. [Google Scholar]
- Pauli, W. Über das Wasserstoffspektrum vom Standpunkt der neuen Quantenmechanik. Z. Für Phys. 1926, 36, 336–363. [Google Scholar] [CrossRef]
- Fock, V. Zur Theorie des Wasserstoffatoms. Z. Für Phys. 1935, 98, 145–154. [Google Scholar] [CrossRef]
- Cohen-Tannoudji, C.; Dupont-Roc, J.; Grynberg, G. Photons and Atoms: Introduction to Quantum Electrodynamics.; Wiley-VCH: New York, 1989. [Google Scholar]
- Sakurai, J.J.; Napolitano, J. Modern Quantum Mechanics., 2nd ed.; Cambridge University Press: Cambridge, 2017. [Google Scholar]
- Goldstein, H.; Poole, C.; Safko, J. Classical Mechanics., 3rd ed.; Addison-Wesley: San Francisco, 2002. [Google Scholar]
- Landau, L.D.; Lifshitz, E.M. Mechanics., 3rd ed.; Butterworth-Heinemann: Oxford, 1976. [Google Scholar]
- Greiner, W.; Müller, B. Quantum Mechanics: Symmetries.; Springer-Verlag: Berlin, 1994. [Google Scholar]
| Feature | Standard Schrödinger Approach | Semi-Quantum Particle–Field Framework (This Work) |
|---|---|---|
| Nature of electron | Intrinsically quantum particle described by a wavefunction | Classical particles with definite position and momentum |
| Fundamental dynamical equation | Schrödinger equation | Classical Hamiltonian dynamics coupled to a quantized electromagnetic field |
| Origin of quantization | Boundary conditions imposed on wavefunction solutions | Emergent commutation structure induced by particle–field interaction |
| Mathematical framework | Hilbert space and operator algebra are assumed from the outset | Classical phase-space dynamics combined with second-quantized field modes |
| Role of electromagnetic field | Typically treated as a classical Coulomb potential | Explicitly quantized field interacting dynamically with the particle |
| Origin of hydrogen spectrum | Eigenvalues of the Schrödinger equation | Runge–Lenz symmetry combined with emergent commutator structure |
| Interpretation of wavefunction | Fundamental description of particle state | Effective ensemble-level description arising from particle–field dynamics |
| Mechanism for discrete energy levels | Standing-wave solutions of the electron wavefunction | Symmetry constraints of the Coulomb system acting on a dynamically quantized phase space |
| Conceptual interpretation | Quantum behavior is intrinsic to matter | Quantum behavior emerges from the interaction between classical particles and quantized fields |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




