Submitted:
30 April 2026
Posted:
01 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
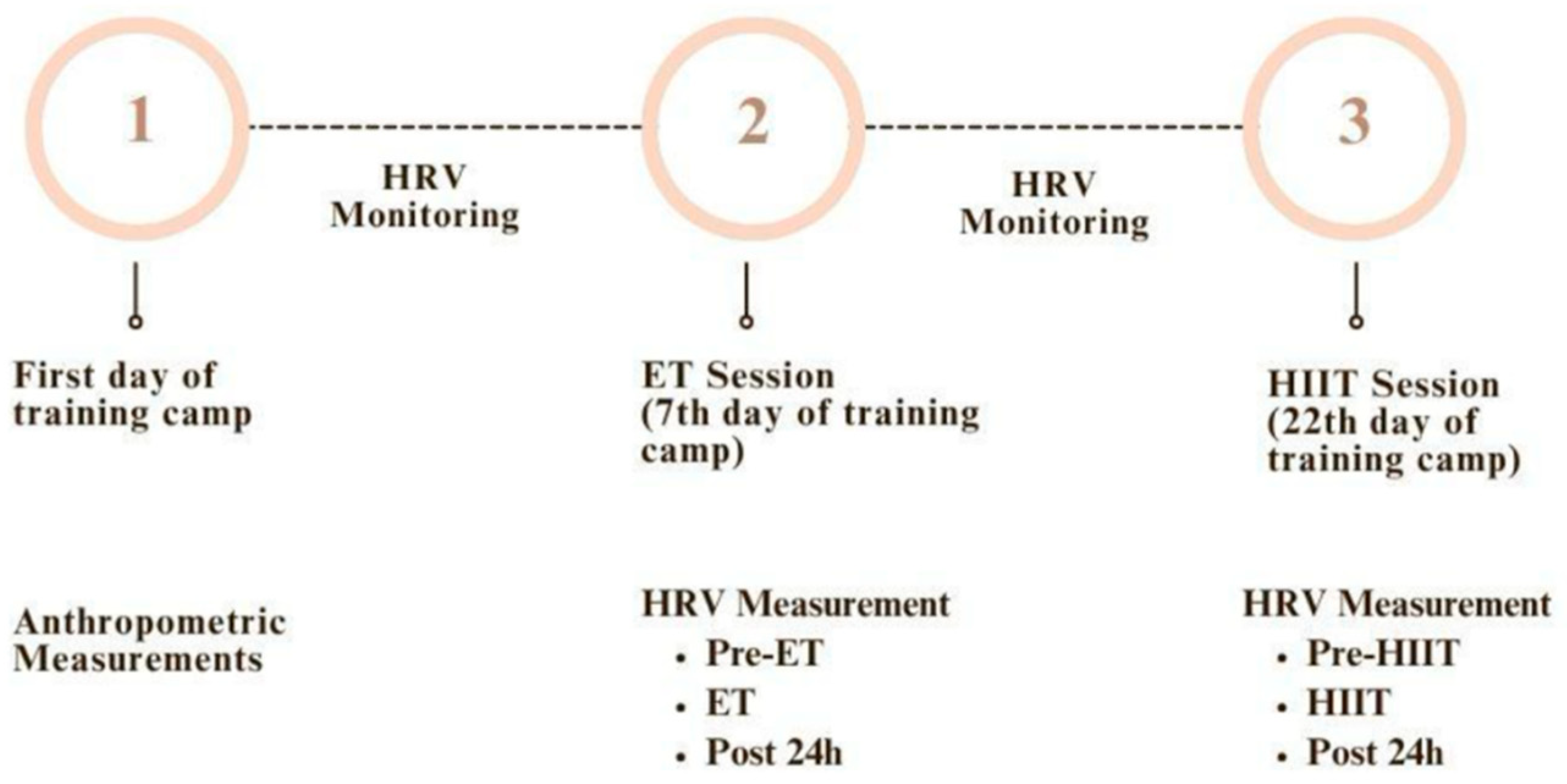
2.1. Experimental Approach to the Problem
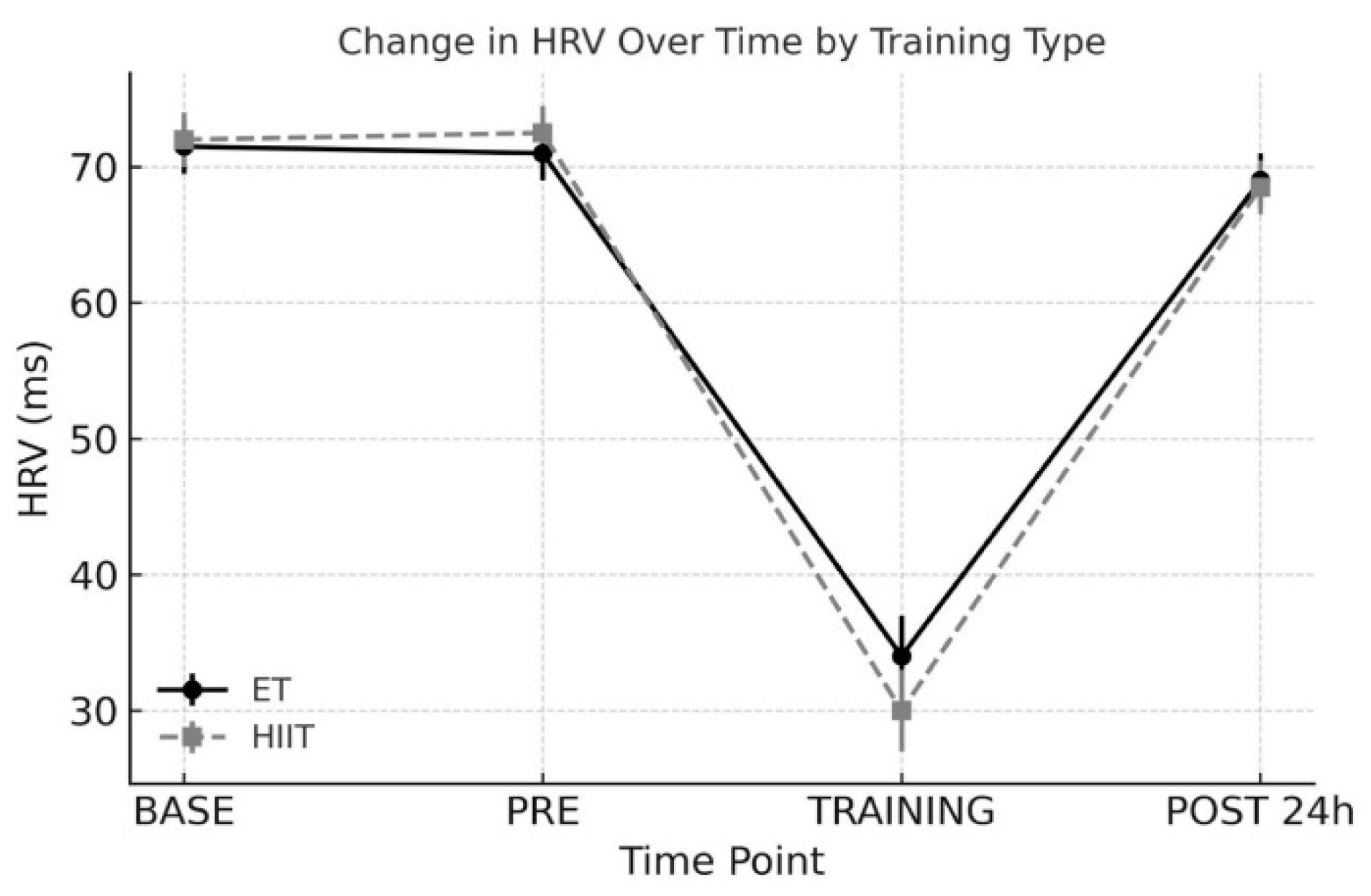
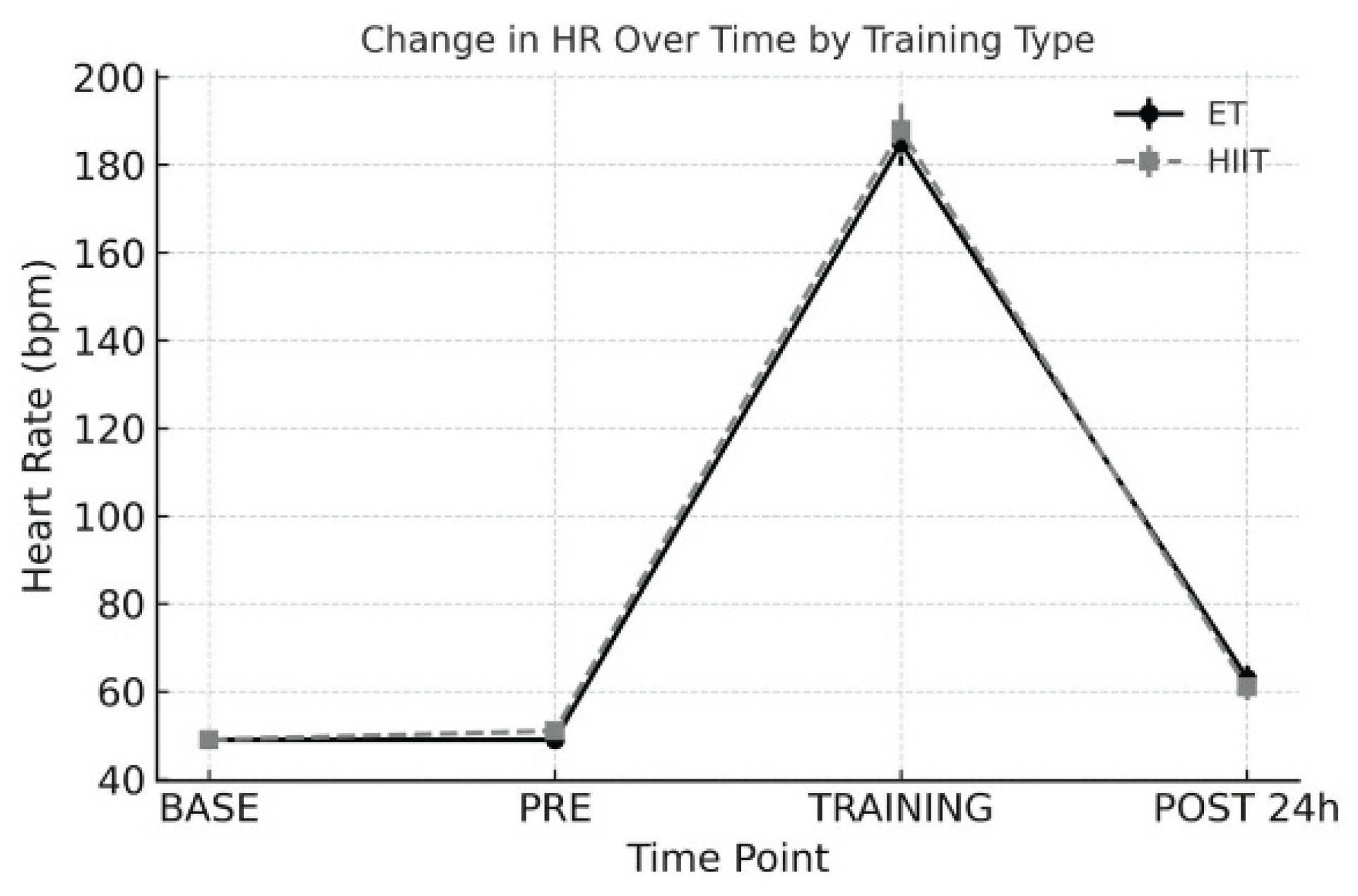
- Baseline (BASE): At least one week prior to each training protocol
- Pre-exercise (PRE): On the day of each training session, before exercise
- During Exercise (TRAINING): Average HRV during the session
- Post 24 h (POST): 24 hours after each training session
2.2. Subjects
2.3. Training Protocol
2.4. Anthropometric Measurements
2.5. Heart Rate Variability Measurements
2.6. Statistical Analyses
3. Results
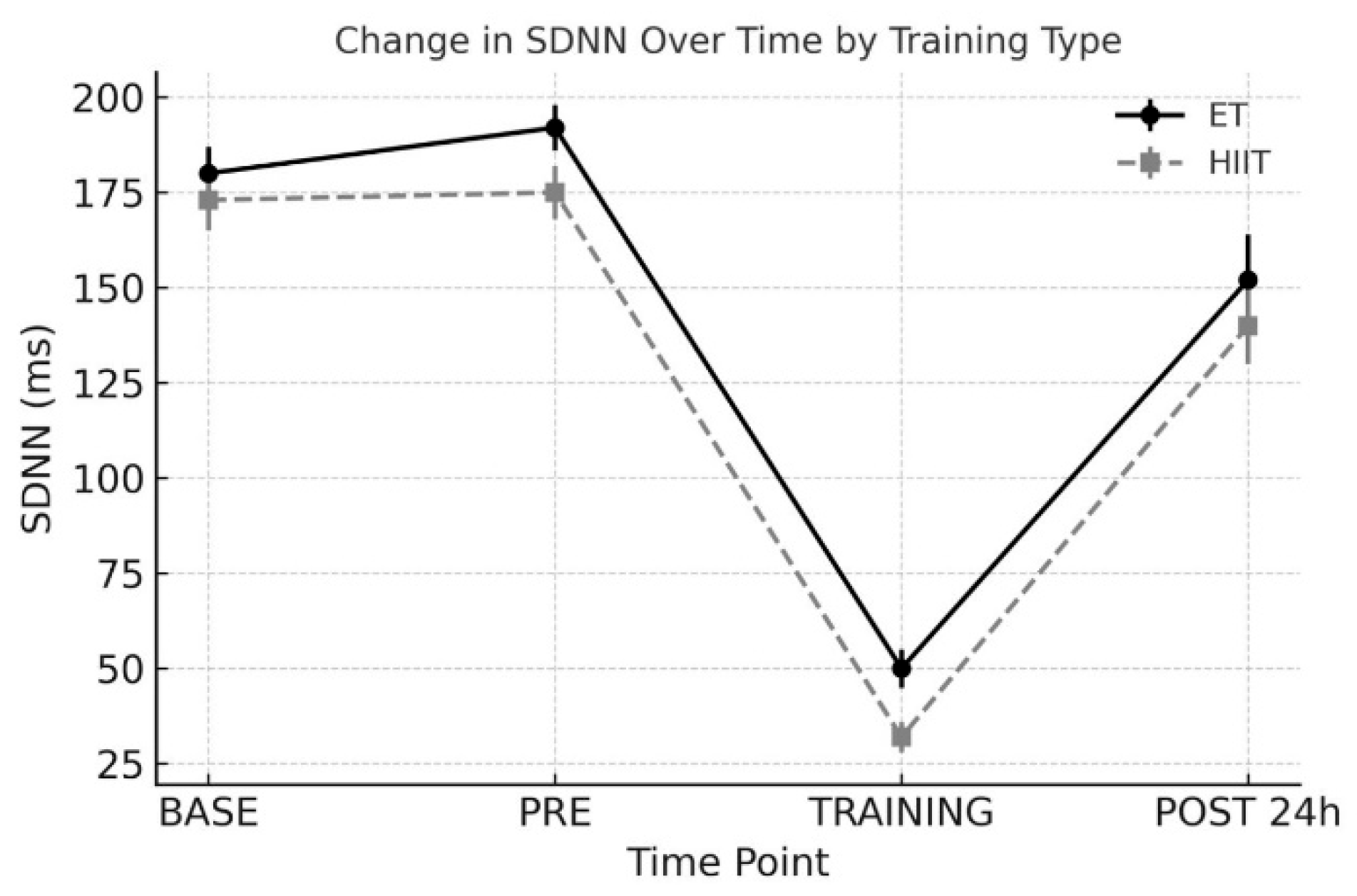
3.1. SDNN (Standard Deviation of NN Intervals)
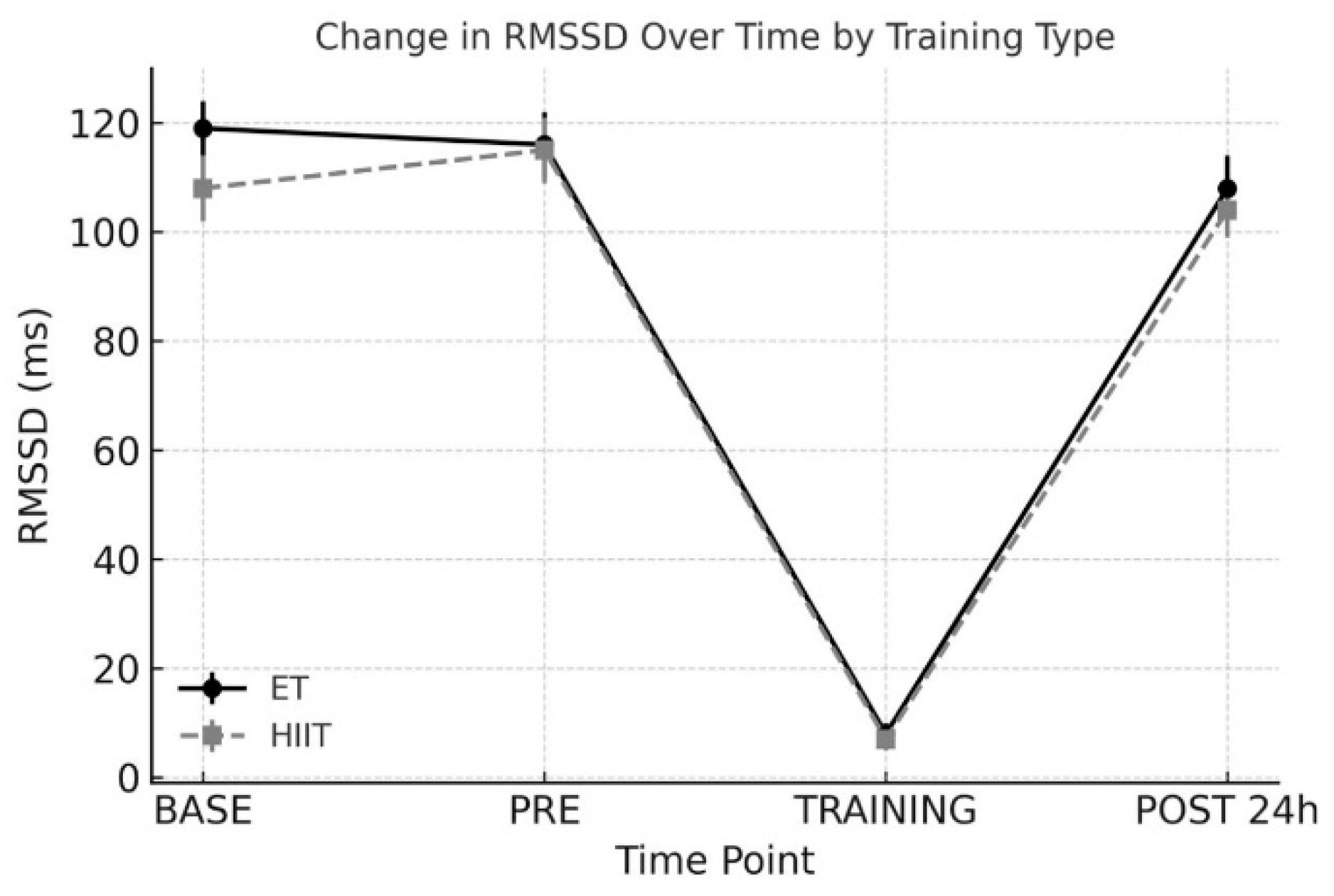
3.2. RMSSD (Root Mean Square of Successive Differences)
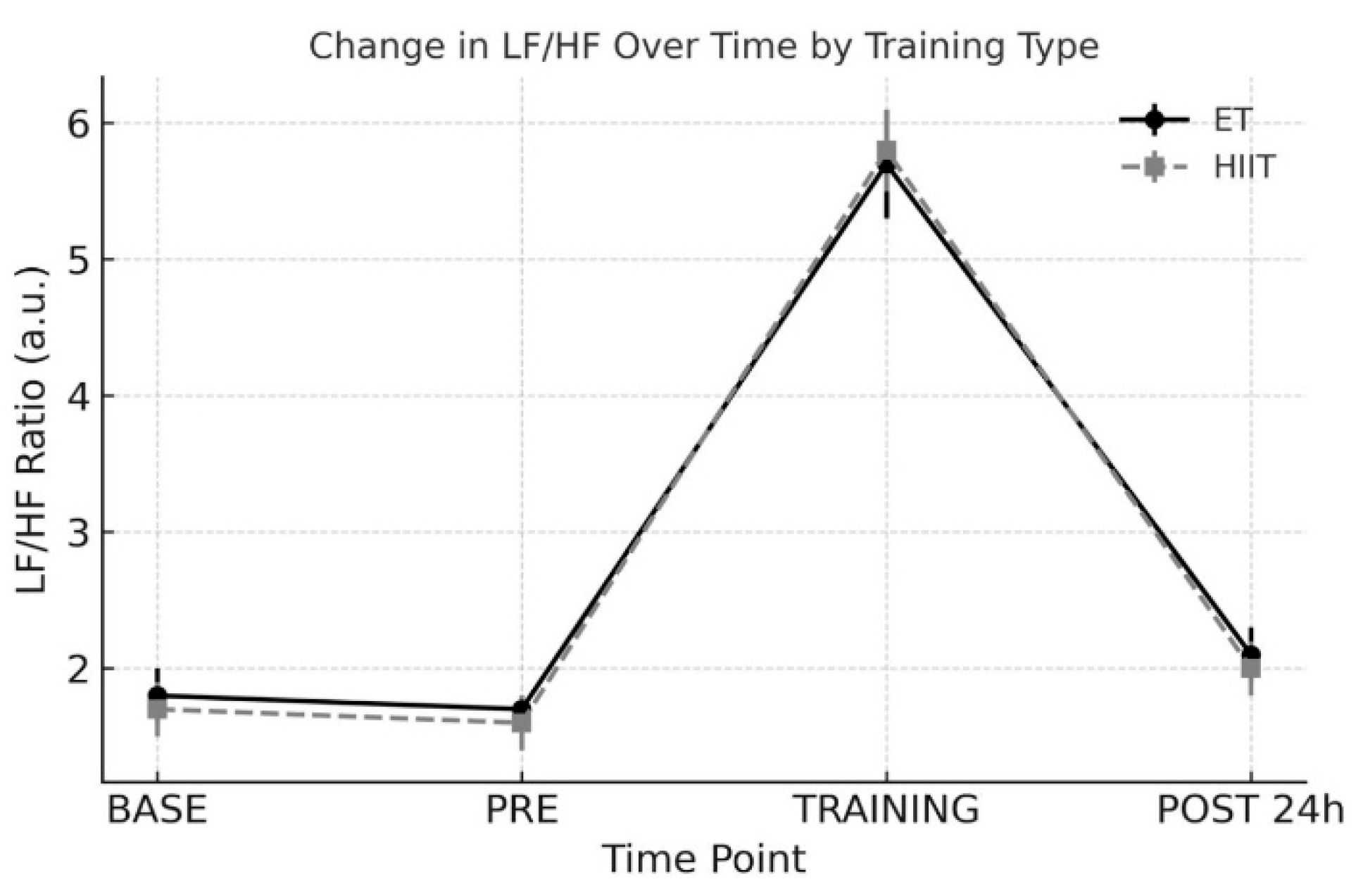
3.3. LF/HF Ratio
3.4. HRV (General Heart Rate Variability Index)
3.5. Heart Rate
4. Discussion
5. Conclusions
6. Practical Applications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sztajzel, J. Heart rate variability: A noninvasive electrocardiographic method to measure the autonomic nervous system. Swiss Med. Wkly. 2004, 134, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Borresen, J.; Lambert, M.I. Autonomic control of heart rate during and after exercise: Measurements and implications for monitoring training status. Sports Med. 2008, 38, 633–646. [Google Scholar] [CrossRef]
- Thomas, B.L.; Claassen, N.; Bhana, S.; Viljoen, M. Validity of commonly used heart rate variability markers of autonomic nervous system function. Neuropsychobiology 2019, 78, 14–26. [Google Scholar] [CrossRef]
- Gullett, N.; Mevawala, N.; Kim, S.Y.S.; Myrick, J.; Gable, P.A. Heart rate variability (HRV) as a way to understand associations between the autonomic nervous system (ANS) and affective states: A critical review of the literature. Int. J. Psychophysiol. 2023, 192, 35–42. [Google Scholar] [CrossRef]
- Choi, W.S.; Kim, K.S.; Kim, S.W.; Yi, J.J.; Kim, J.Y. Effects of Aerobic Exercise on Heart Rate Variability (HRV). Korean J. Fam. Med. 2005, 26, 561–566. [Google Scholar]
- Cornelissen, V.; Verheyden, B.; Aubert, A.E.; Fagard, R.H. Effects of aerobic training intensity on resting, exercise and post-exercise blood pressure, heart rate and heart-rate variability. J. Hum. Hypertens. 2010, 24, 175–182. [Google Scholar] [CrossRef]
- Fronchetti, L.; Aguiar, C.A.; Aguiar, A.F.; Nakamura, F.Y.; Oliveira, P.R. Effects of High-Intensity Interval Training on Heart Rate Variability During Exercise. J. Exerc. Physiol. Online 2007, 10, 1–9. [Google Scholar]
- Bentley, R.F.; Walsh, J.J.; Drouin, P.J.; Hollohan, Q.J.; Reed, J.L. Heart rate variability and recovery following maximal exercise in endurance athletes and physically active individuals. Appl. Physiol. Nutr. Metab. 2020, 45, 1138–1144. [Google Scholar] [CrossRef] [PubMed]
- Suzic Lazic, J.; Dekleva, M.; Soldatovic, I.; Leischik, R.; Mazic, S. Heart rate recovery in elite athletes: The impact of age and exercise capacity. Clin. Physiol. Funct. Imaging 2017, 37, 117–123. [Google Scholar] [CrossRef]
- Schmitt, L.; Regnard, J.; Desmarets, M.; Mauny, F.; Mourot, L.; Millet, G.P. Fatigue shifts and scatters heart rate variability in elite endurance athletes. PLoS ONE 2013, 8, e71588. [Google Scholar] [CrossRef]
- Kim, J.A.; Kang, S.W. Relationship among sleep quality, heart rate variability, fatigue, depression, and anxiety in adults. Korean J. Adult Nurs. 2017, 29, 87–97. [Google Scholar] [CrossRef]
- Javorka, M.; Zila, I.; Balharek, T.; Javorka, K. Heart rate recovery after exercise: Relations to heart rate variability and complexity. Braz. J. Med. Biol. Res. 2002, 35, 991–1000. [Google Scholar] [CrossRef]
- Bellenger, C.R.; Fuller, J.T.; Thomson, R.L.; Davison, K.; Robertson, E.Y.; Buckley, J.D. Monitoring athletic training status through autonomic heart rate regulation: A systematic review and meta-analysis. Sports Med. 2016, 46, 1461–1486. [Google Scholar] [CrossRef]
- Hautala, A.J.; Makikallio, T.H.; Kiviniemi, A.; Laukkanen, R.T.; Nissila, S.; Heikkila, J.; Huikuri, H.V. Cardiovascular autonomic function correlates with the response to aerobic training in healthy sedentary subjects. Am. J. Physiol. Heart Circ. Physiol. 2003, 285, H1747–H1752. [Google Scholar] [CrossRef]
- Stanley, J.; Peake, J.M.; Buchheit, M. Cardiac parasympathetic reactivation following exercise: Implications for training prescription. Sports Med. 2013, 43, 1259–1277. [Google Scholar] [CrossRef]
- Plews, D.J.; Laursen, P.B.; Stanley, J.; Kilding, A.E.; Buchheit, M. Training adaptation and heart rate variability in elite endurance athletes: Opening the door to effective monitoring. Sports Med. 2013, 43, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Buchheit, M. Monitoring training status with HR measures: Do all roads lead to Rome? Front. Physiol. 2014, 5, 73. [Google Scholar] [CrossRef]
- Plews, D.J.; Laursen, P.B.; Kilding, A.E.; Buchheit, M. Heart-rate variability and training-intensity distribution in elite rowers. Int. J. Sports Physiol. Perform. 2014, 9, 1026–1032. [Google Scholar] [CrossRef] [PubMed]
- Sarmiento, S.; Garcia-Manso, J.M.; Jimenez-Gutierrez, A.; Rodriguez-Matoso, D.; Calderon, J. Heart rate variability during high-intensity exercise. J. Syst. Sci. Complex. 2013, 26, 104–116. [Google Scholar] [CrossRef]
- Gronwald, T.; Hoos, O. Correlation properties of heart rate variability during endurance exercise: A systematic review. Ann. Noninvasive Electrocardiol. 2020, 25, e12697. [Google Scholar] [CrossRef] [PubMed]
- Sandercock, G.; Brodie, D. The use of heart rate variability measures to assess autonomic control during exercise. Scand. J. Med. Sci. Sports 2006, 16, 302–313. [Google Scholar] [CrossRef]
- Fu, Q.; Levine, B.D. Exercise and the autonomic nervous system. Handb. Clin. Neurol. 2013, 117, 147–160. [Google Scholar] [PubMed]
- Kaikkonen, P.; Hynynen, E.; Mann, T.; Rusko, H.; Nummela, A. Heart rate variability is related to training load variables in interval running exercises. Eur. J. Appl. Physiol. 2012, 112, 829–838. [Google Scholar] [CrossRef]
- Michael, S.; Graham, K.S.; Davis, G.M. Cardiac autonomic responses during exercise and post-exercise recovery using heart rate variability and systolic time intervals—A review. Front. Physiol. 2017, 8, 301. [Google Scholar] [CrossRef]
- Kaikkonen, P.; Nummela, A.; Rusko, H. Heart rate variability dynamics during early recovery after different endurance exercises. Eur. J. Appl. Physiol. 2007, 102, 79–86. [Google Scholar] [CrossRef]
- Kaikkonen, P.; Rusko, H.; Martinmäki, K. Post-exercise heart rate variability of endurance athletes after different high-intensity exercise interventions. Scand. J. Med. Sci. Sports 2008, 18, 511–519. [Google Scholar] [CrossRef] [PubMed]
- Kiss, O.; Sydó, N.; Vargha, P.; Vágó, H.; Csajági, E.; Belicza, É.; Merkely, B. Detailed heart rate variability analysis in athletes. Clin. Auton. Res. 2016, 26, 245–252. [Google Scholar] [CrossRef]
- Berkoff, D.J.; Cairns, C.B.; Bertocci, L.A.; Cuddy, A.J. Heart rate variability in elite American track-and-field athletes. J. Strength Cond. Res. 2007, 21, 227–231. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.



