Submitted:
30 April 2026
Posted:
02 May 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Epidemiological Trends of Early-Onset Colorectal Cancer
Metastatic Patterns and Multi-Organ Dissemination
| Study | Year | Study Design | Population | Key Findings on Metastasis |
|---|---|---|---|---|
| Akimoto et al. | 2021 | Review | Global epidemiology | EOCRC incidence increasing worldwide; advanced stage more common |
| Patel & Ahnen | 2021 | Review | Young CRC patients | Higher frequency of metastatic disease at presentation |
| Medici et al. | 2023 | Review | Metastatic EOCRC | Younger patients more likely to present with synchronous metastasis |
| Hua et al. | 2023 | Systematic review | Risk factors EOCRC | Lifestyle and metabolic factors contribute to aggressive tumour biology |
| Jeri-Yabar et al. | 2024 | SEER analysis | Metastatic EOCRC | Multi-organ metastasis associated with poorer survival |
| Siegel et al. | 2024 | Epidemiological study | US cancer registry | Increasing stage IV CRC among individuals <50 years |
| Sung et al. | 2024 | Global cancer statistics | Global population | Rising incidence of CRC in younger adults |
| Brown & Solomon | 2023 | Review | Metastatic CRC | Liver and lung remain dominant metastatic sites |
| Loree & Kopetz | 2023 | Review | Molecular CRC | Distinct genomic landscape in EOCRC |
| Kasi et al. | 2020 | Population study | Young CRC | Younger patients present with more advanced disease |
| Characteristic | Early-Onset Colorectal Cancer (<50 Years) | Late-Onset Colorectal Cancer (≥50 Years) | Clinical Implication |
|---|---|---|---|
| Stage at diagnosis | More frequently diagnosed at advanced stages (III–IV) | Higher proportion detected at earlier stages due to screening | Delayed diagnosis contributes to higher metastatic burden in EOCRC |
| Frequency of synchronous metastasis | Higher prevalence of synchronous metastases at initial presentation | Lower proportion of synchronous metastatic disease | More aggressive initial clinical presentation in EOCRC |
| Liver metastasis | Most common site; often occurs with additional sites | Most common site; frequently isolated | EOCRC more likely to present with combined metastatic involvement |
| Lung metastasis | More frequently observed, especially in rectal tumours | Common but less frequently multi-organ | Suggests potential differences in dissemination pathways |
| Peritoneal metastasis | Relatively higher incidence, especially in mucinous tumours | Less common | Associated with poorer prognosis and complex surgical management |
| Multi-organ metastasis | Higher likelihood of ≥2 distant organs involved | Less frequent | Indicates more aggressive tumour biology |
| Distant lymph node metastasis | More commonly reported | Less frequently highlighted | Reflects systemic disease spread |
| Bone metastasis | Rare but reported more often in aggressive EOCRC | Rare | Associated with late-stage disease |
| Brain metastasis | Very rare but increasingly recognised | Extremely rare | Indicates advanced systemic dissemination |
| Histological features | Higher prevalence of mucinous and signet-ring cell carcinoma | Conventional adenocarcinoma more common | Histology may influence metastatic behaviour |
| Molecular features | Higher rates of MSI and distinct genomic signatures | More heterogeneous molecular landscape | May influence response to targeted therapy and immunotherapy |
Molecular Drivers of Aggressive Metastatic Behaviour
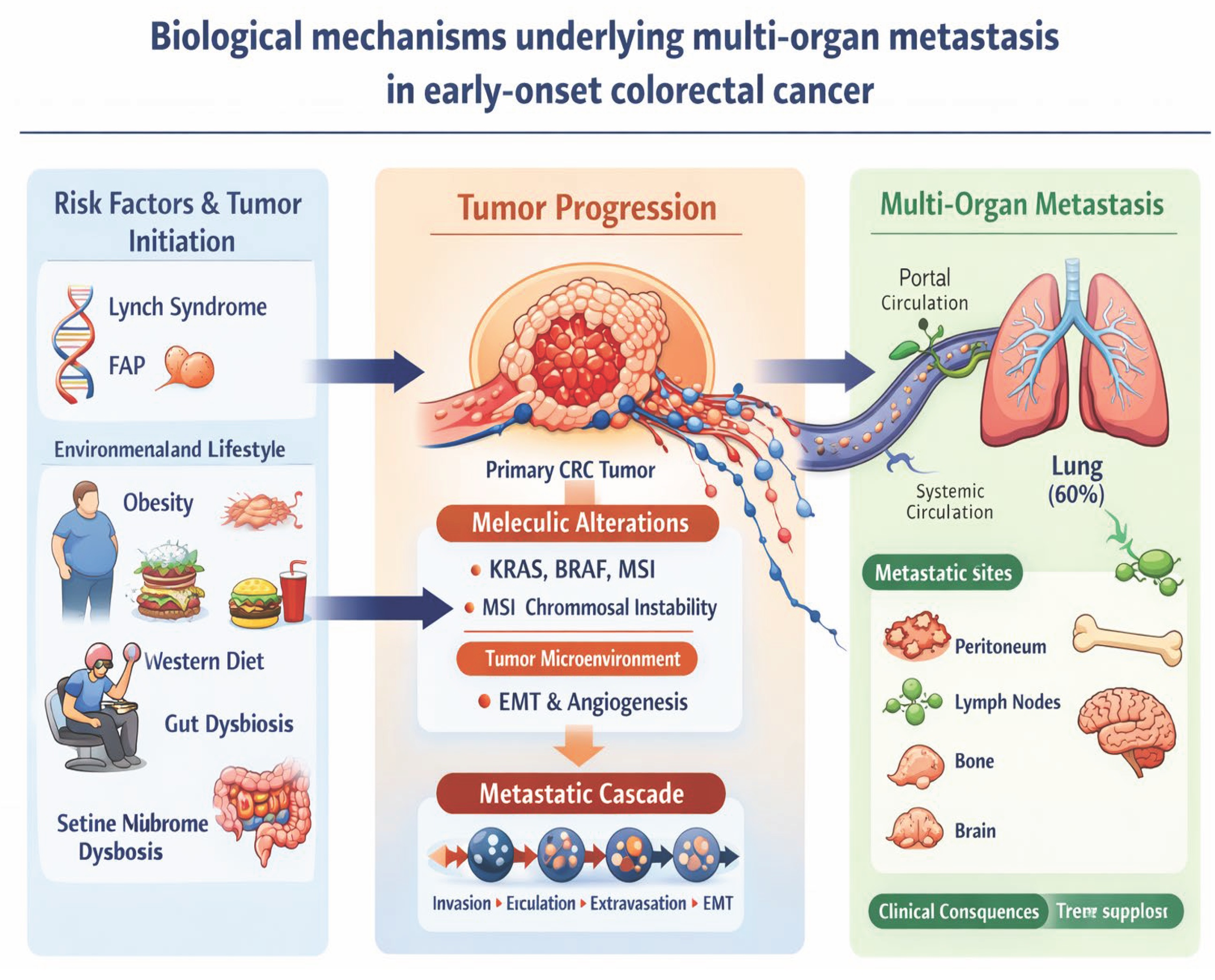
Therapeutic Strategies for Multi-Organ Metastatic EOCRC
Prognostic Implications
| Factor | Impact on Prognosis |
|---|---|
| Number of metastatic organs | Worse survival |
| Peritoneal metastasis | Poor prognosis |
| BRAF mutation | Poor response to therapy |
| MSI status | Better response to immunotherapy |
Future Perspectives
Conclusion
CRediT Authorship Contribution Statement
Declaration of Generative AI in Scientific Writing
References
- Sinicrope, F.A. Increasing incidence of early-onset colorectal cancer. N Engl. J. Med. 2022. [Google Scholar] [CrossRef]
- Patel, S.G.; Karlitz, J.J.; Yen, T.; Lieu, C.H.; Boland, C.R. The rising tide of early-onset colorectal cancer. Lancet Gastroenterol. Hepatol. 2022. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Fedewa, S.A.; Anderson, W.F.; et al. Colorectal cancer incidence patterns in the United States. J. Natl. Cancer Inst. 2017. [Google Scholar] [CrossRef] [PubMed]
- Zaborowski, A.M.; Abdile, A.; Adamina, M.; et al. Characteristics of early-onset vs late-onset colorectal cancer. JAMA Surg. 2021. [Google Scholar] [CrossRef]
- Medici, B.; Riccò, B.; Caffari, E.; et al. Early onset metastatic colorectal cancer: Current insights and clinical management. In Cancers (Basel); 2023. [Google Scholar] [CrossRef]
- Lawler, T.; Parlato, L.; Andersen, S.W. Histological and molecular characteristics of early-onset colorectal cancer. Front Oncol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Hua, H.; Jiang, Q.; Sun, P.; Xu, X. Risk factors for early-onset colorectal cancer: Systematic review and meta-analysis. Front Oncol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Stoffel, E.M.; Murphy, C.C. Epidemiology and mechanisms of early-onset colorectal cancer. Gastroenterology 2020. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Cervantes, A.; Adam, R.; et al. ESMO consensus guidelines for metastatic colorectal cancer. Ann. Oncol. 2023. [Google Scholar] [CrossRef]
- Kasi, P.M.; Shahjehan, F.; Cochuyt, J.J.; et al. Rising incidence of young-onset colorectal cancer. JAMA Netw. Open 2020. [Google Scholar] [CrossRef]
- Brown, K.G.; Solomon, M.J. Progress and challenges in metastatic colorectal cancer. Lancet 2023. [Google Scholar] [CrossRef]
- Jeri-Yabar, A.; Vittini-Hernandez, L.; Prado-Nuñez, S.; Dharmapuri, S. Survival analysis of metastatic early-onset colorectal cancer. In Cancers (Basel); 2024. [Google Scholar] [CrossRef]
- Dwyer, A.J.; Rathod, A.; King, C.; et al. Advancing early-onset colorectal cancer research. Front Oncol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Boland, C.R.; Goel, A. Microsatellite instability in colorectal cancer. Gastroenterology 2018. [Google Scholar] [CrossRef] [PubMed]
- Yaeger, R.; Corcoran, R.B. Targeting alterations in the RAF-MEK pathway in colorectal cancer. Cancer Discov. 2019. [Google Scholar] [CrossRef] [PubMed]
- Galon, J.; Bruni, D. Tumour immunology and the immune contexture of colorectal cancer. Nat. Rev. Gastroenterol. Hepatol. 2019. [Google Scholar] [CrossRef]
- Wong, S.H.; Yu, J. Gut microbiota in colorectal cancer. Nat. Rev. Gastroenterol. Hepatol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Biller, L.H.; Schrag, D. Diagnosis and treatment of metastatic colorectal cancer. JAMA 2021. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.; Kitano, Y. Multidisciplinary management of colorectal liver metastases. Ann. Surg. 2019. [Google Scholar] [CrossRef]
- Bhatt, A.; Goéré, D. Cytoreductive surgery plus HIPEC for colorectal peritoneal metastases. Indian J. Surg. Oncol. 2016. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




