Submitted:
30 April 2026
Posted:
01 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
- Denture base material significantly influenced the debonding occurrence of resin-cemented attachment housing-denture base complexes under cyclic mechanical loading.
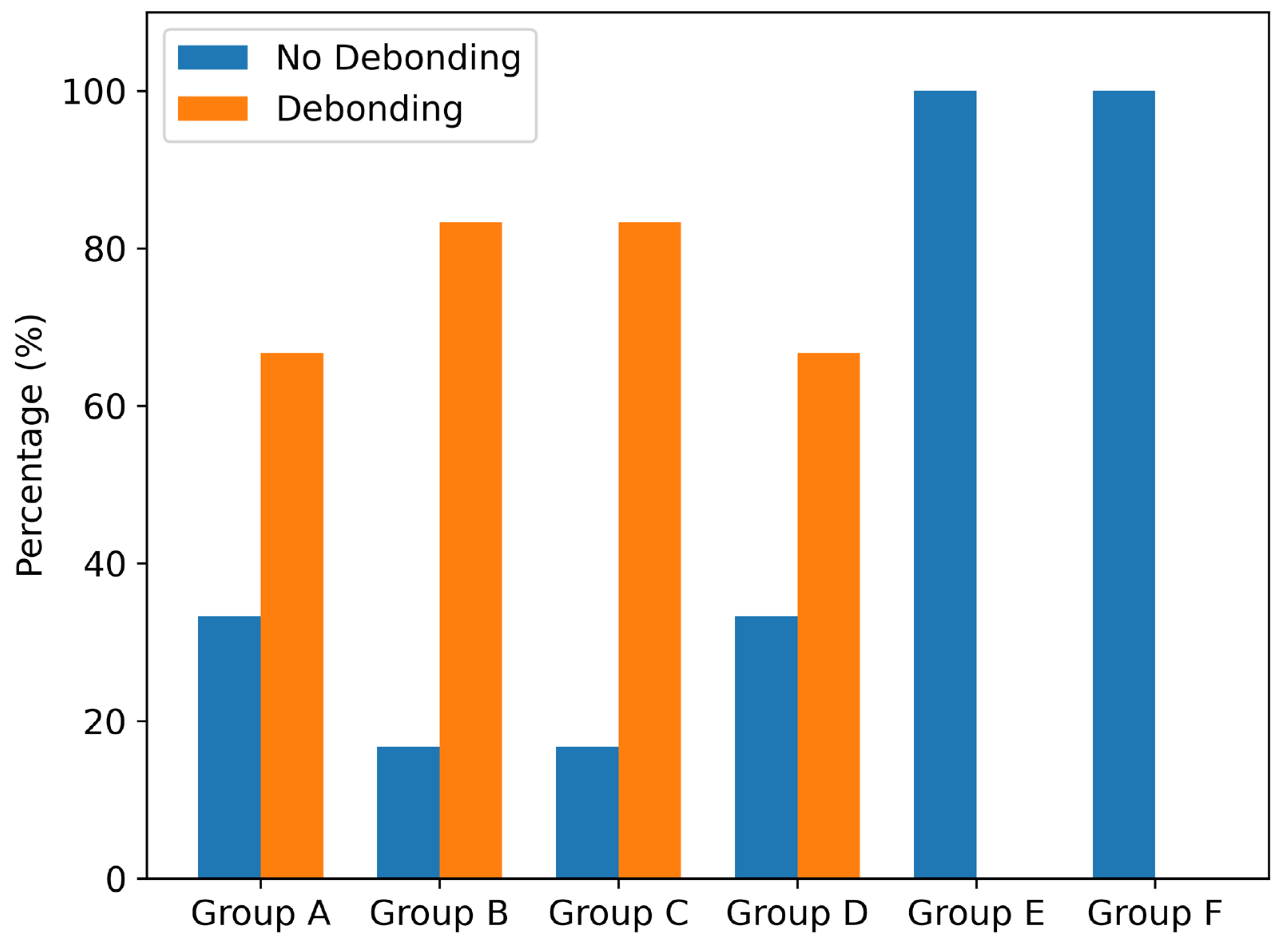
- Co-Cr denture base specimens did not exhibit debonding within the applied cyclic loading protocol, whereas debonding was frequently observed in PMMA and PEEK denture base specimens.
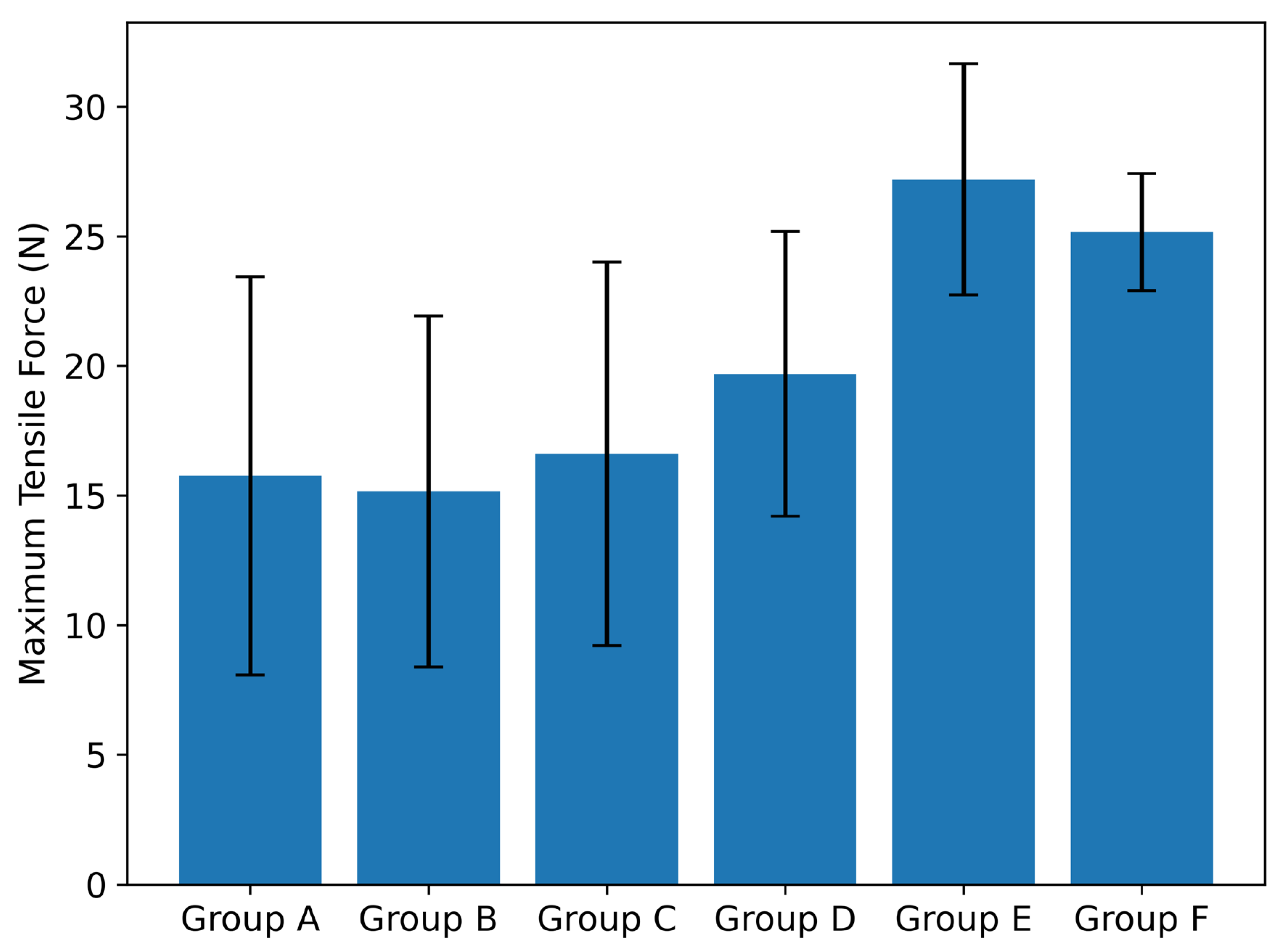
- Denture base material significantly affected the maximum tensile force measured during cyclic mechanical loading, with Co-Cr specimens demonstrating higher values than PMMA and PEEK specimens.
- Attachment housing material did not significantly affect debonding occurrence or maximum tensile force as a main effect. However, a significant interaction with denture base material was observed for debonding occurrence.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Girundi, A.L.G.; Ribeiro, M.C. de O.; Vargas-Moreno, V.F.; Borges, G.A.; Magno, M.B.; Maia, L.C.; Del Bel Cury, A.A; Marcello-Machado, R.M. Patient-reported outcome measures and clinical performance of implant-retained mandibular overdentures with stud and ball attachments: A systematic review and meta-analysis. J. Prosthet. Dent. 2024, 131, 197–211. [CrossRef]
- Gonçalves, F.; Campestrini, V.L.L.; Rigo-Rodrigues, M.A.; Zanardi, P.R. Effect of the attachment system on the biomechanical and clinical performance of overdentures: A systematic review. J. Prosthet. Dent. 2020, 123, 589–594. [CrossRef]
- Assaf, A.; Daas, M.; Boittin, A.; Eid, N.; Postaire, M. Prosthetic maintenance of different mandibular implant overdentures: A systematic review. J. Prosthet. Dent. 2017, 118, 144–152. [CrossRef]
- Prassad, S.; Faverani, L.P.; Junior, J.F.S.; Sukotjo, C.; Yuan, J.C.-C. Attachment systems for mandibular implant-supported overdentures: A systematic review and meta-analysis of randomized controlled trials. J. Prosthet. Dent. 2024, 132, 354–368. [CrossRef]
- Gibreel, M.F.; Khalifa, A.; Said, M.M.; Mahanna, F.; El-Amier, N.; Närhi, T.O.; Perea-Lowery, L.; Vallittu, P.K. Biomechanical aspects of reinforced implant overdentures: A systematic review. J. Mech. Behav. Biomed. Mater. 2019, 91, 202–211. [CrossRef]
- Takahashi, T.; Gonda, T.; Mizuno, Y.; Fujinami, Y.; Maeda, Y. Reinforcement in removable prosthodontics: A literature review. J. Oral Rehabil. 2017, 44, 133–143. [CrossRef]
- Gama, L.T.; Duque, T.M.; Özcan, M.; Philippi, A.G.; Mezzomo, L.A.M.; Gonçalves, T.M.S.V. Adhesion to high-performance polymers applied in dentistry: A systematic review. Dent. Mater. 2020, 36, e93–e108. [CrossRef]
- Hussain, M.; Godbole, S.G.; Nimonkar, S.; Reddy, K.M.; Saha, S.; Laskar, P.; Bansod, A. Evaluation of adhesion strength of pure PEEK and glass reinforced PEEK with titanium using different bonding methods- a systematic review. BMC Oral Health 2026, 26, 151. [CrossRef]
- Papathanasiou, I.; Kamposiora, P.; Papavasiliou, G.; Ferrari, M. The use of PEEK in digital prosthodontics: A narrative review. BMC Oral Health 2020, 20, 217. [CrossRef]
- Gama, L.T.; Bezerra, A.P.; Schimmel, M.; Garcia, R.C.M.R.; Canto, G. de L.; Gonçalves, T.M.S.V. Clinical performance of polymer frameworks in dental prostheses: A systematic review. J. Prosthet. Dent. 2024, 131, 579–590. [CrossRef]
- Alsabeeha, N.H.M.; Payne, A.G.T.; Swain, M.V. Attachment systems for mandibular two-implant overdentures: A review of in vitro investigations on retention and wear features. Int. J. Prosthodont. 2009, 22, 429–440.
- Ozkir, S.E.; Yilmaz, B.; Kurkcuoglu, I.; Culhaoglu, A.; Unal, S.M. Surface roughness and adaptation of different materials to secure implant attachment housings. J. Prosthet. Dent. 2017, 117, 87–92. [CrossRef]
- Basic information. Straumann Novaloc retentive system for hybrid dentures. Available online: https://www.straumann.com/content/dam/media-center/straumann/en/documents/brochure/technical-information/702067-en_low.pdf. (accessed 1 February 2026).
- Domingo, K.B.; Burgess, J.O.; Litaker, M.S.; McCracken, M.S. Strength comparison of four techniques to secure implant attachment housings to complete dentures. J. Prosthet. Dent. 2013, 110, 8–13. [CrossRef]
- Cayouette, M.J.; Barnes, L.; Vuthiganon, J.; McPherson, K. Shear bond strength comparison of implant-retained overdenture attachment pickup materials. Gen. Dent. 2016, 64, 47–50.
- Ozkir, S.; Yilmaz, B. Effect of different housing retaining materials on the flexural strength of an acrylic resin overdenture base. J. Prosthet. Dent. 2017, 118, 500–503. [CrossRef]
- Nakhaei, M.; Dashti, H.; Baghbani, A.; Ahmadi, Z. Bond strength of locator housing attached to denture base resin secured with different retaining materials. Dent. Res. J. (Isfahan) 2020, 17, 34-39. [CrossRef]
- Panittaveekul, M.; Phasuk, K.; Levon, J.; Chu, T.-M.G. Bonding between implant attachment pickup materials and CAD-CAM denture base material. J. Prosthet. Dent. 2021, 126, 102.e1–e7. [CrossRef]
- Yildiz, P.; Ünlü, D.G. Investigation of the effect of different surface treatments for preventing detachment of polyetheretherketone and titanium attachment matrix housings in overdenture: An in vitro study. J. Indian Prosthodont. Soc. 2023, 23, 142–149. [CrossRef]
- Nakhaei, M.; Dashti, H.; Sasannejad, S.; Mohammadipour, H.S. Bonding the titanium attachment matrix housing to acrylic overdentures using different primers, adhesives, and resin materials: An in vitro study. Int. J. Dent. 2025, 2025, 9702318. [CrossRef]
- Küçükekenci, A.S.; Dönmez, M.B.; Dede, D.Ö.; Çakmak, G.; Yilmaz, B. Bond strength of recently introduced computer-aided design and computer-aided manufacturing resin-based crown materials to polyetheretherketone and titanium. J. Prosthet. Dent. 2024, 132, 1066.e1–e8. [CrossRef]
- Stewart, B.L.; Edwards, R.O. Retention and wear of precision-type attachments. J. Prosthet. Dent. 1983, 49, 28–34. [CrossRef]

| Material | Type | Manufacturer | Ref number |
|---|---|---|---|
| breCAM.base | Polymethylmethacrylate (PMMA) milling disc | Bredent GmbH & Co. KG | 54PC2030 |
| breCAM.BioHPP | Polyetheretherketone (PEEK) milling disc | Bredent GmbH & Co. KG | 54002029 |
| AUDENTAL SLM POWDER | Cobalt-Chromium (Co-Cr) alloy powder |
Audental Bio-material CO., Ltd | AU-CCP-000 |
| Novaloc Matrix System | Titanium (Ti) housing | Institut Straumann AG | 2010.701-NOV |
| Novaloc Matrix System | Polyetheretherketone (PEEK) housing | Institut Straumann AG | 2010.702-NOV |
| PANAVIA V5 Paste | Dual-polymerized resin cement | Kuraray Noritake Dental Inc. | #3611-EU |
| Clearfil Ceramic Primer Plus | Single-component adhesive primer | Kuraray Noritake Dental Inc. | #3637-EU |
| visio.link | Composite resin primer | Bredent GmbH & Co. KG | VLPMMA10 |
| Denture base material | Attachment housing material | |
|---|---|---|
| Group A | PMMA 1 | Ti 4 |
| Group B | PEEK | |
| Group C | PEEK 2 | Ti |
| Group D | PEEK | |
| Group E | Co-Cr 3 | Ti |
| Group F | PEEK |
| Group A | Group B | Group C | Group D | Group E | Group F | Total (n*=30) |
|
|---|---|---|---|---|---|---|---|
| No debonding | 33.3% | 16.7% | 16.7% | 33.3% | 100% | 100% | 40% |
| Debonding | 66.7% | 83.3% | 83.3% | 66.7% | 0% | 0% | 60% |
| (I)Material | (J)Material | Mean difference (I-J) | Standard Error | P *-value | 95% confidence interval Lower bound Upper bound |
|
|---|---|---|---|---|---|---|
| PMMA 1 | PEEK | -2.689 | 2.527 | .89 | -9.139 | 3.761 |
| Co-Cr | -10.725 | 3.095 | .005 | -18.625 | -2.825 | |
| PEEK 2 | PMMA | 2.689 | 2.527 | .89 | -3.761 | 9.139 |
| Co-Cr | -8.036 | 3.095 | .045 | -15.936 | -.136 | |
| Co-Cr 3 | PEEK | 8.036 | 3.095 | .045 | .136 | 15.936 |
| PMMA | 10.725 | 3.095 | .005 | 2.825 | 18.625 | |
| Groups | Mean | ±SD | Minimum | Maximum |
|---|---|---|---|---|
| A | 15.76 | 7.68 | 6.38 | 25.81 |
| B | 15.16 | 6.77 | 8.59 | 26.54 |
| C | 16.61 | 7.4 | 8.54 | 26.52 |
| D | 19.69 | 5.49 | 13.77 | 26.79 |
| E | 27.2 | 4.47 | 23.6 | 32.2 |
| F | 25.17 | 2.26 | 23 | 27.5 |
| Total | 18.68 | 7.19 | 6.38 | 32.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




