Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
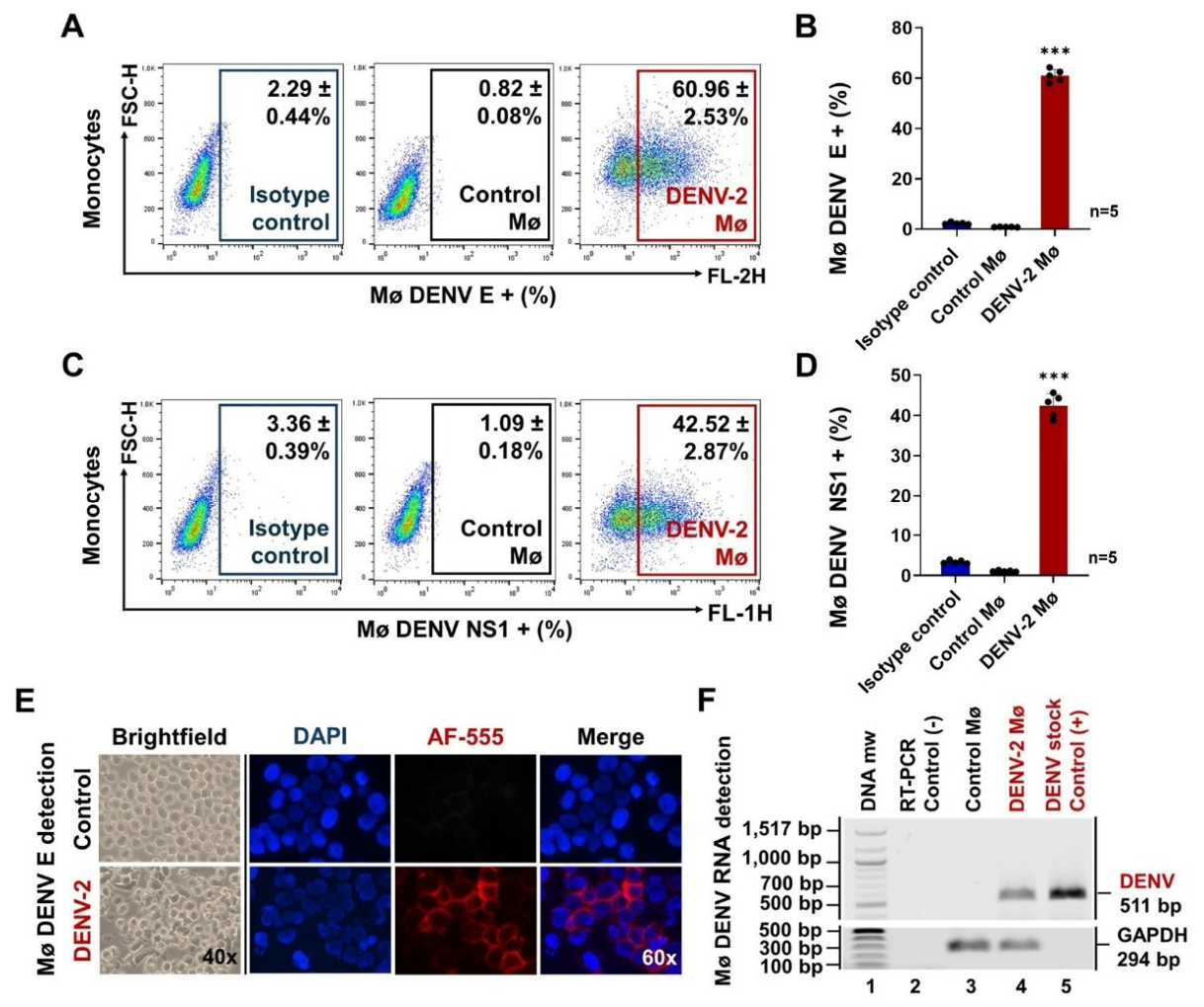
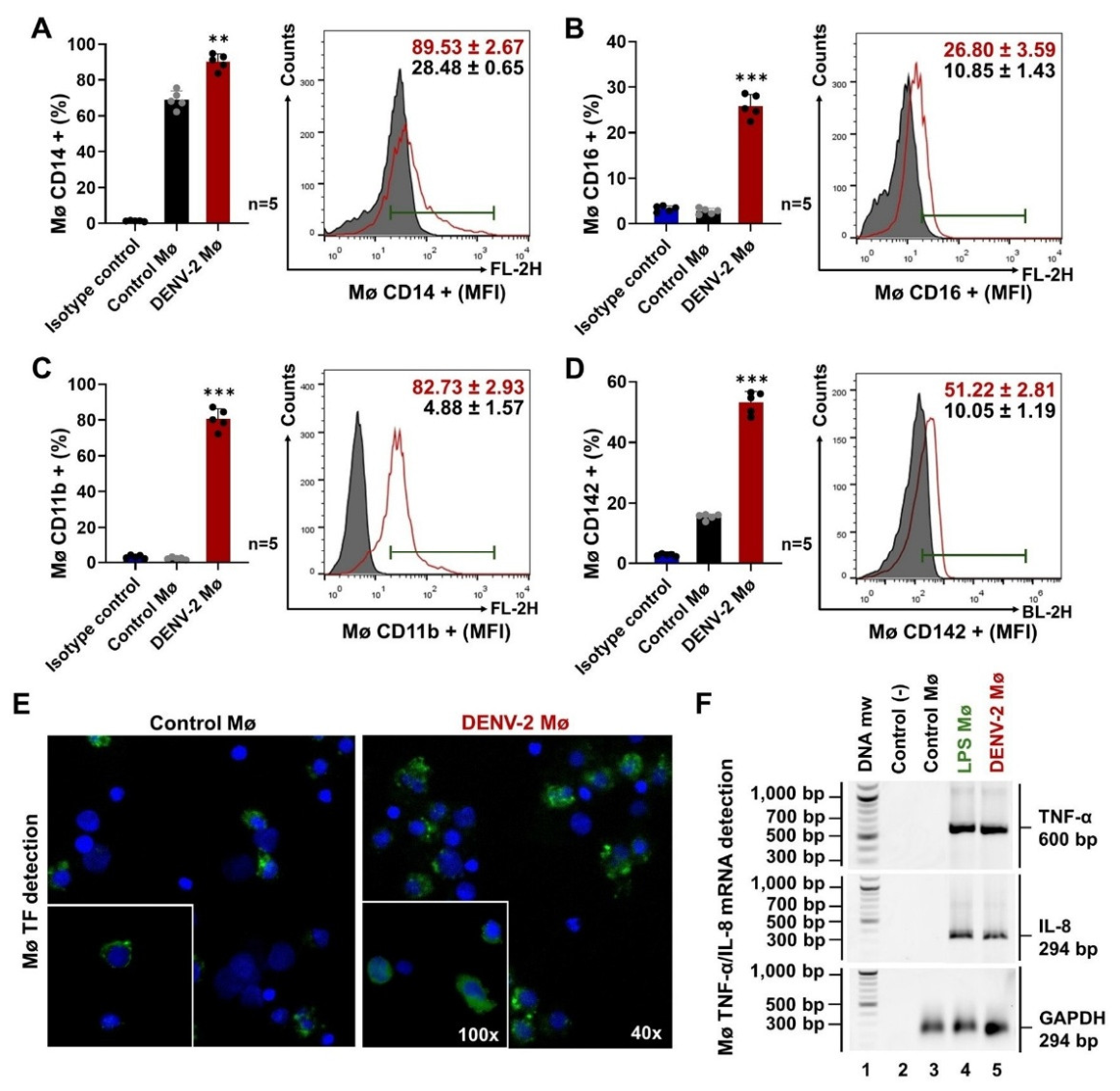
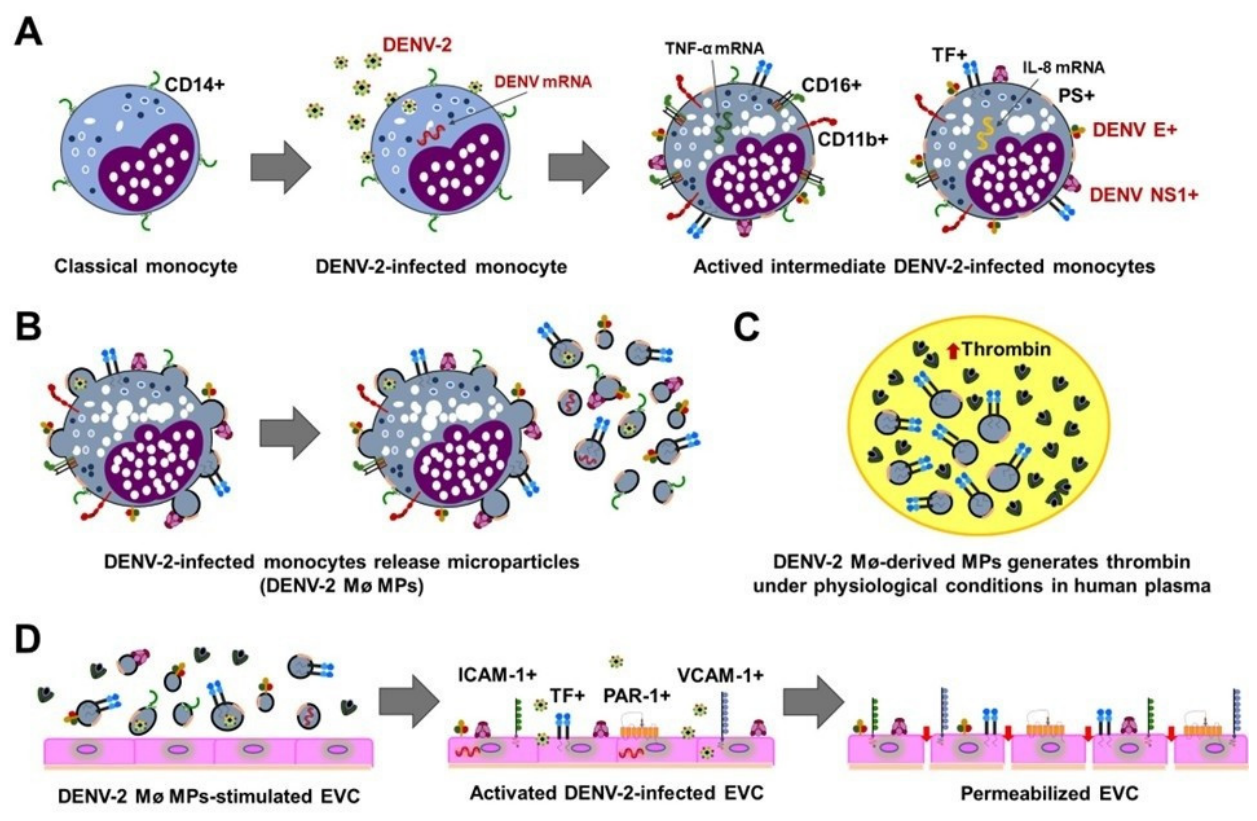
2.1. Dengue Virus (DENV) Infection Induces Procoagulant and Proinflammatory Phenotype in Monocytes
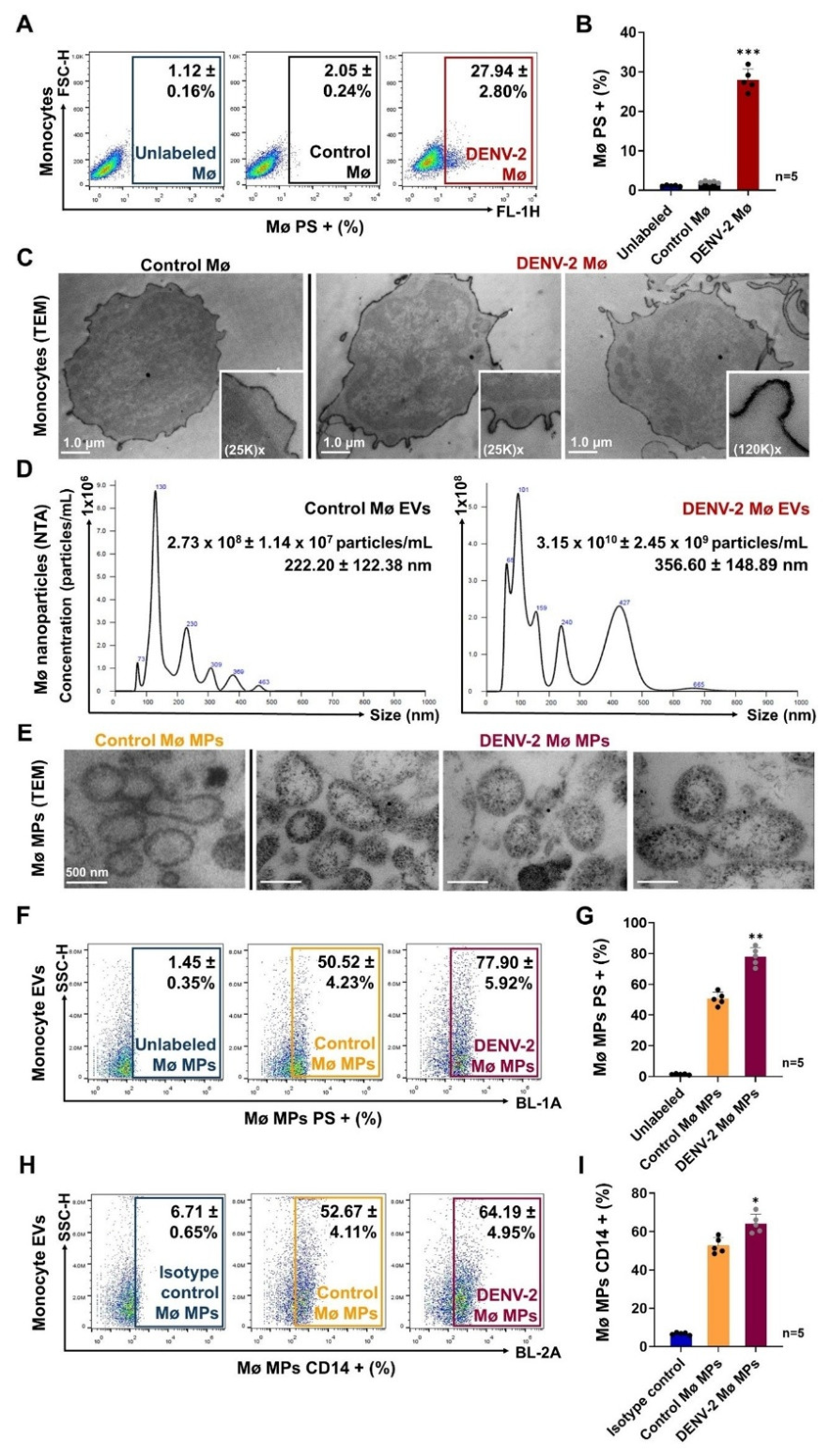
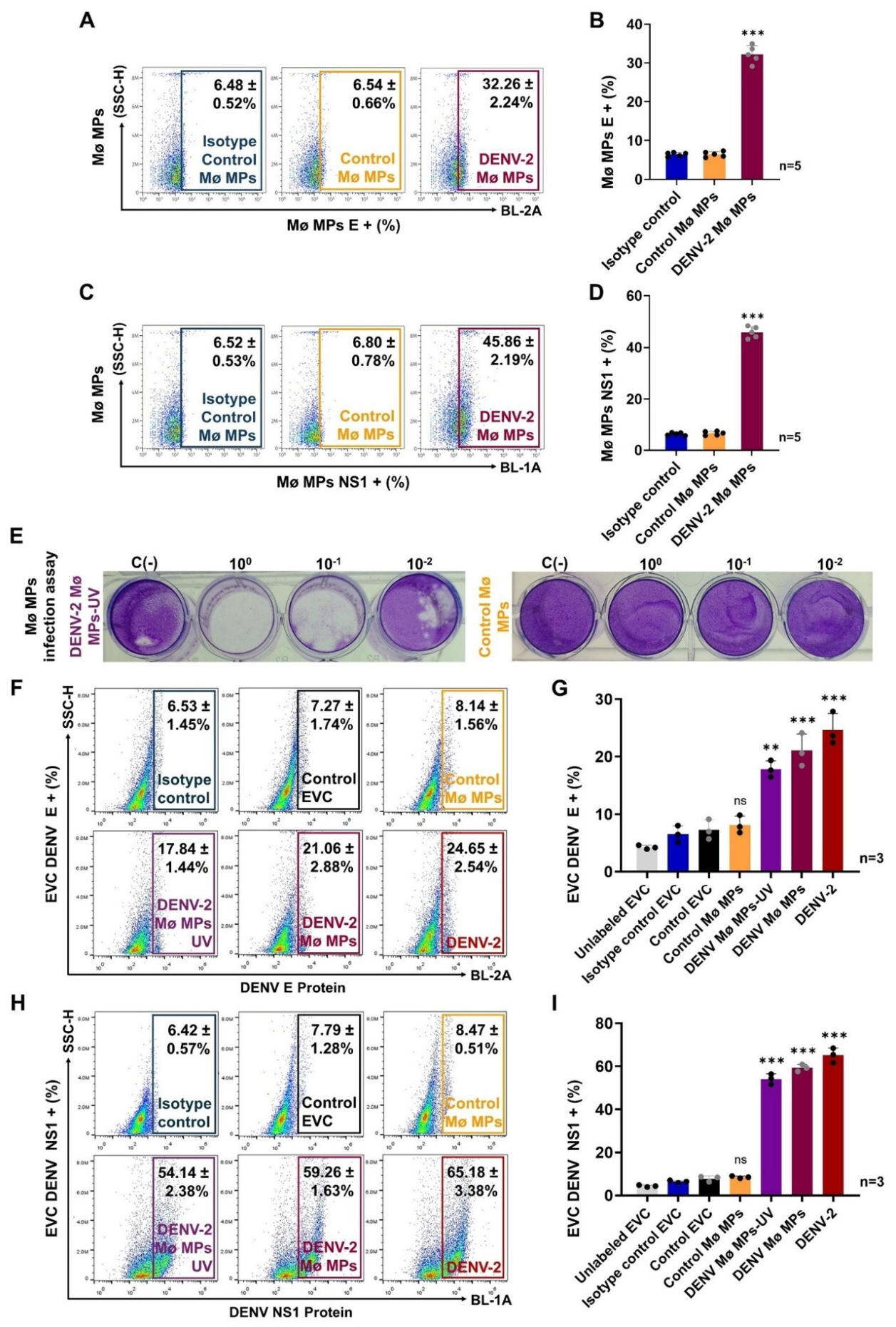
2.2. DENV-Infected Monocytes Release Extracellular Vesicles (Microparticles), Carrying Viral Components that Promote Viral Dissemination to Endothelial Vascular Cells
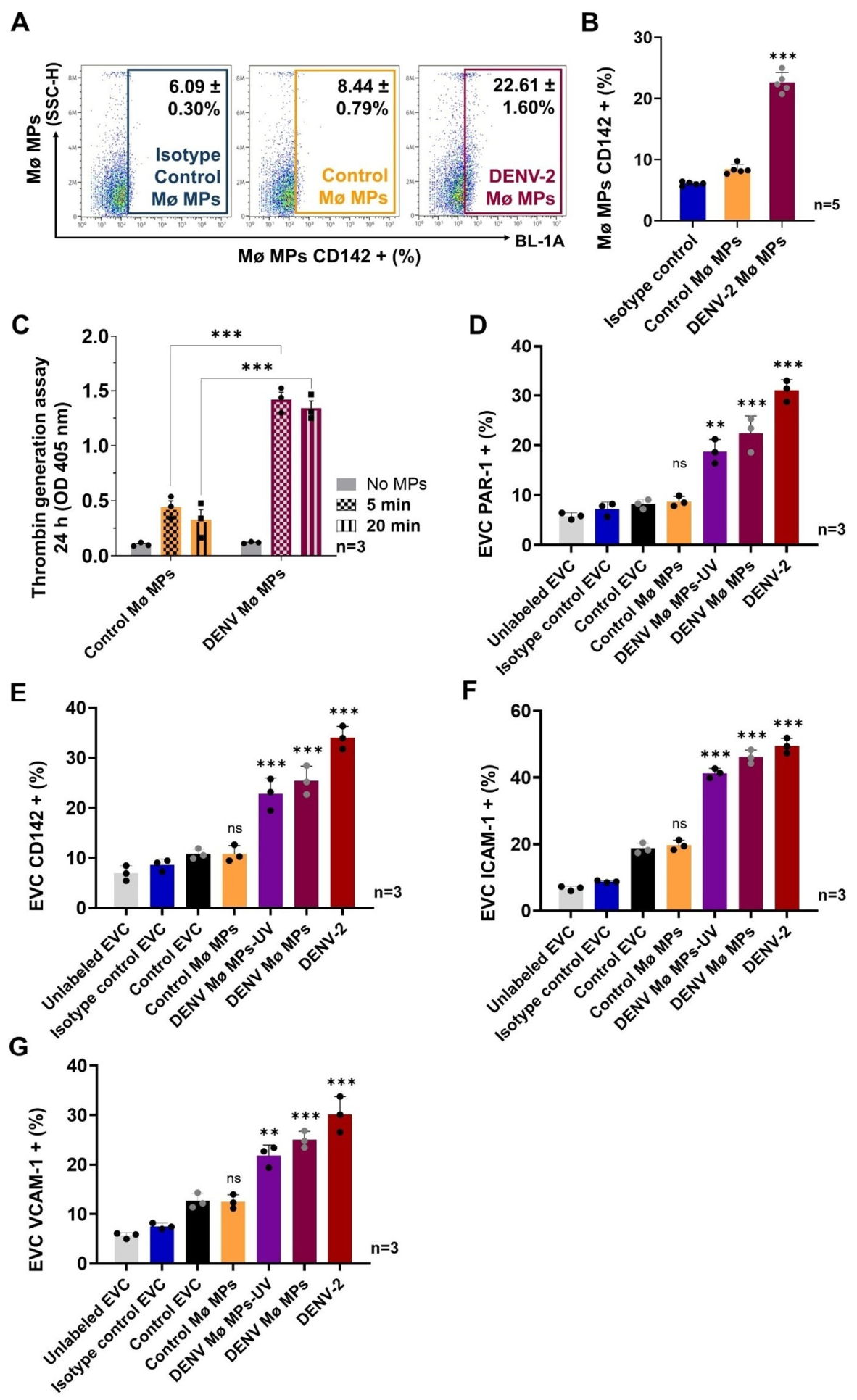
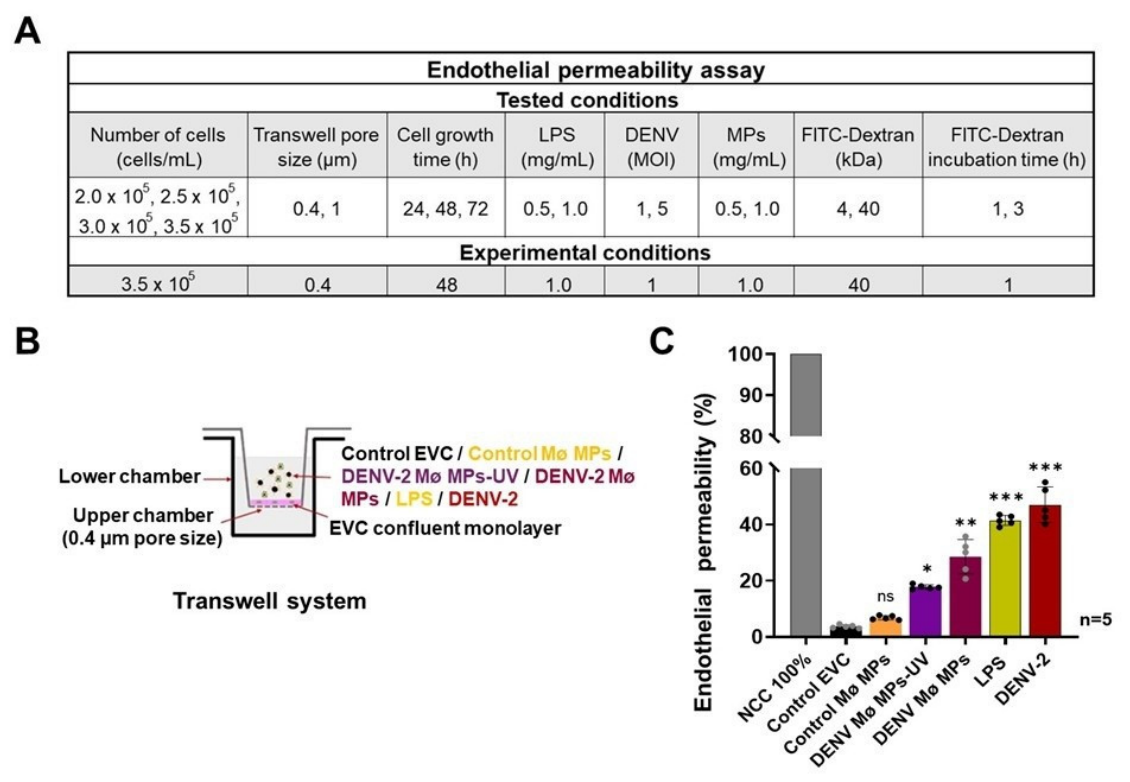
2.3. DENV-Infected Mø-Derived MPs Exhibit Procoagulant Activity and Promote Vascular Dysfunction and Endothelial Vascular Barrier Disruption
3. Discussion
4. Materials and Methods
4.1. Cell Cultures
4.2. Isolation, Propagation and Purification of Dengue Virus
4.3. Titration of DENV-2 Stock
4.4. Preparation of Extracellular Vesicles–Depleted Fetal Bovine Serum (EV-Depleted FBS)
4.5. Monocyte DENV-Infection Assay
4.6. Detection of DENV Proteins and Cellular Markers in Monocytes by Flow Cytometry
4.7. Detection of DENV E Protein and Tissue Factor in Monocytes by Immunofluorescence
4.8. Ruthenium Red Staining for Evaluation of Monocyte Surface Membranes by Transmission Electron Microscopy (TEM)
4.9. Isolation of Monocyte-Derived MPs by Differential Ultracentrifugation
4.10. Characterization of Monocyte-Derived MPs by Nanoparticle Tracking Analysis (NTA)
4.11. Morphological Characterization of Monocyte-Derived MPs by TEM
4.12. Detection of CD14, Tissue Factor (TF, CD142), Phosphatidylserine (PS), and Viral Proteins (E and NS1) in Monocyte-Derived MPs by Flow Cytometry
4.13. Virion Inactivation in MPs Derived from DENV-Infected Monocytes
4.14. Assessment of Infectious DENV Virions Associated with MP Isolates by Plaque Assay
4.15. Thrombin Generation Assay Mediated by Monocyte Microparticles
4.16. Protein Quantification in Isolates of Monocyte-Derived MPs by Micro BCA Protein Assay
4.17. Stimulation of Naïve Endothelial Vascular Cells (EVC) with Monocyte-Derived MPs
4.18. Detection of Viral Proteins and Cellular Markers in EVC by Flow Cytometry
4.19. Assessment of EVC Permeability by Transwell Assay
4.20. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CD | Cluster of differentiation |
| DENV | Dengue virus |
| DENV E | Dengue virus envelope protein |
| DENV NS1 | Dengue virus nonstructural protein 1 |
| DENV-2 Mø MPs | Microparticles derived from dengue virus-infected monocytes |
| DF | Dengue fever |
| EVC | Endothelial vascular cells |
| EVs | Extracellular vesicles |
| ICAM-1 | Intercellular adhesion molecule 1 |
| IL | Interleukin |
| MPs | Microparticles |
| Mø | Monocytes |
| mRNA | Messenger ribonucleic acid |
| NTA | Nanoparticle tracking analysis |
| TEM | Transmission electron microscopy |
| TF | Tissue Factor |
| TNF-α | Tumor necrosis factor alpha |
| SD | Severe dengue |
| PAR-1 | Protease-activated receptor 1 |
| PS | Phosphatidylserine |
| VCAM-1 | Vascular cell adhesion molecule 1 |
References
- World Health Organization (WHO). Dengue. Available online: https://www.who.int/news-room/fact-sheets/detail/dengue-and-severe-dengue (accessed on 28 January 2026).
- Yang, Z.S.; Baua, A.D.; Hemdan, M.S.; Assavalapsakul, W.; Wang, W.H.; Lin, C.Y.; Chao, D.Y.; Chen, Y.H.; Wang, S.F. Dengue virus infection: A systematic review of pathogenesis, diagnosis and management. J. Infect. Public. Health 2025, 18, 102982. [Google Scholar] [CrossRef]
- Kothari, D.; Patel, N.; Bishoyi, A.K. Dengue: epidemiology, diagnosis methods, treatment options, and prevention strategies. Arch. Virol. 2025, 170, 48. [Google Scholar] [CrossRef]
- World Health Organization (WHO); Special Programme for Research and Training in Tropical Diseases (TDR). Dengue: Guidelines for Diagnosis, Treatment, Prevention and Control, New Edition; WHO Press: Geneva, Switzerland, 2009; pp. 10–13. [Google Scholar]
- Aguilar-Briseño, J.A.; Upasani, V.; Ellen, B.M.T.; Moser, J.; Pauzuolis, M.; Ruiz-Silva, M.; Heng, S.; Laurent, D.; Choeung, R.; Dussart, P.; et al. TLR2 on blood monocytes senses dengue virus infection and its expression correlates with disease pathogenesis. Nat. Commun. 2020, 11, 3177. [Google Scholar] [CrossRef]
- Castillo, J.A.; Naranjo, J.S.; Rojas, M.; Castaño, D.; Velilla, P.A. Role of Monocytes in the Pathogenesis of Dengue. Arch. Immunol. Ther. Exp. (Warsz) . 2019, 67, 27–40. [Google Scholar] [CrossRef]
- Lertjuthaporn, S.; Keawvichit, R.; Polsrila, K.; Sukapirom, K.; Chuansumrit, A.; Chokephaibulkit, K.; Ansari, A.A.; Khowawisetsut, L.; Pattanapanyasat, K. Kinetic Changes of Peripheral Blood Monocyte Subsets and Expression of Co-Stimulatory Molecules during Acute Dengue Virus Infection. Pathogens. 2021, 10, 1458. [Google Scholar] [CrossRef] [PubMed]
- Dunagan, M.M.; Fox, J.M. Splenic macrophages escalate dengue disease. Nat. Microbiol. 2023, 8, 1378–1379. [Google Scholar] [CrossRef] [PubMed]
- Flipse, J.; Torres, S.; Diosa-Toro, M.; van der Ende-Metselaar, H.; Herrera-Rodriguez, J.; Urcuqui-Inchima, S.; Huckriede, A.; Rodenhuis-Zybert, I.A.; Smit, J.M. Dengue tropism for macrophages and dendritic cells: the host cell effect. J. Gen. Virol. 2016, 97, 1531–1536. [Google Scholar] [CrossRef]
- Losada, P.X.; DeLaura, I.; Narváez, C.F. Dengue Virus and Platelets: From the Biology to the Clinic. Viral. Immunol. 2022, 35, 349–358. [Google Scholar] [CrossRef] [PubMed]
- Quirino-Teixeira, A.C.; Andrade, F.B.; Pinheiro, M.B.M.; Rozini, S.V.; Hottz, E.D. Platelets in dengue infection: more than a numbers game. Platelets. 2022, 33, 176–183. [Google Scholar] [CrossRef]
- Vervaeke, P.; Vermeire, K.; Liekens, S. Endothelial dysfunction in dengue virus pathology. Rev. Med. Virol. 2015, 25, 50–67. [Google Scholar] [CrossRef]
- Malavige, G.N.; Ogg, G.S. Pathogenesis of vascular leak in dengue virus infection. Immunology. 2017, 151, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Naranjo-Gómez, J.S.; Castillo, J.A.; Rojas, M.; Restrepo, B.N.; Diaz, F.J.; Velilla, P.A.; Castaño, D. Different phenotypes of non-classical monocytes associated with systemic inflammation, endothelial alteration, and hepatic compromise in patients with dengue. Immunology. 2019, 156, 147–163. [Google Scholar] [CrossRef]
- Wong, K.L.; Chen, W.; Balakrishnan, T.; Toh, Y.X.; Fink, K.; Wong, S.C. Susceptibility and response of human blood monocyte subsets to primary dengue virus infection. PLoS ONE 2012, 7, e36435. [Google Scholar] [CrossRef]
- Silva, T.; Gomes, L.; Jeewandara, C.; Ogg, G.S.; Malavige, G.N. Dengue NS1 induces phospholipase A2 enzyme activity, prostaglandins, and inflammatory cytokines in monocytes. Antiviral. Res. 2022, 202, 105312. [Google Scholar] [CrossRef]
- Beltrami, S.; Ferraresi, M.; Cianci, G.; Narducci, M.; Rizzo, R.; Baroni, M.; Bortolotti, D. Flavivirus Nonstructural Protein 1-Driven Coagulation via Tissue Factor-Bearing Microvesicles: A Pilot Study. ACS. Omega. 2025, 10, 56645–56655. [Google Scholar] [CrossRef]
- de Azeredo, E.L.; Kubelka, C.F.; Alburquerque, L.M.; Barbosa, L.S.; Damasco, P.V.; Avila, C.A.; Motta-Castro, A.R.; da Cunha, R.V.; Monteiro, R.Q. Tissue factor expression on monocytes from patients with severe dengue fever. Blood. Cells. Mol. Dis. 2010, 45, 334–5. [Google Scholar] [CrossRef]
- Martins, S.T.; Kuczera, D.; Lötvall, J.; Bordignon, J.; Alves, L.R. Characterization of Dendritic Cell-Derived Extracellular Vesicles During Dengue Virus Infection. Front. Microbiol. 2018, 9, 1792. [Google Scholar] [CrossRef] [PubMed]
- Welsh, J.A.; Goberdhan, D.C.I.; O'Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal information for studies of extracellular vesicles (MISEV2023): From basic to advanced approaches. J. Extracell. Vesicles. 2024, 13, e12404. [Google Scholar] [CrossRef]
- Russell, A.E.; Sneider, A.; Witwer, K.W.; Bergese, P.; Bhattacharyya, S.N.; Cocks, A.; Cocucci, E.; Erdbrügger, U.; Falcon-Perez, J.M.; Freeman, D.W.; et al. Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: an ISEV position paper arising from the ISEV membranes and EVs workshop. J. Extracell. Vesicles. 2019, 8, 1684862. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles. 2018, 7, 1535750. [Google Scholar] [CrossRef] [PubMed]
- Cocucci, E.; Meldolesi, J. Ectosomes and exosomes: shedding the confusion between extracellular vesicles. Trends. Cell. Biol. 2015, 25, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Voukalis, C.; Shantsila, E.; Lip, G.Y.H. Microparticles and cardiovascular diseases. Ann. Med. 2019, 51, 193–223. [Google Scholar] [CrossRef]
- Lugo-Gavidia, L.M.; Burger, D.; Matthews, V.B.; Nolde, J.M.; Galindo Kiuchi, M.; Carnagarin, R.; Kannenkeril, D.; Chan, J.; Joyson, A.; Herat, L.Y.; et al. Role of Microparticles in Cardiovascular Disease: Implications for Endothelial Dysfunction, Thrombosis, and Inflammation. Hypertension. 2021, 77, 1825–1844. [Google Scholar] [CrossRef]
- Singh, A.; Bisht, P.; Bhattacharya, S.; Guchhait, P. Role of Platelet Cytokines in Dengue Virus Infection. Front. Cell. Infect. Microbiol. 2020, 10, 561366. [Google Scholar] [CrossRef] [PubMed]
- Hottz, E.D.; Lopes, J.F.; Freitas, C.; Valls-de-Souza, R.; Oliveira, M.F.; Bozza, M.T.; Da Poian, A.T.; Weyrich, A.S.; Zimmerman, G.A.; Bozza, F.A.; et al. Platelets mediate increased endothelium permeability in dengue through NLRP3-inflammasome activation. Blood. 2013, 122, 3405–3414. [Google Scholar] [CrossRef]
- Patil, R.; Bajpai, S.; Ghosh, K.; Shetty, S. Microparticles as prognostic biomarkers in dengue virus infection. Acta. Trop. 2018, 181, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Punyadee, N.; Mairiang, D.; Thiemmeca, S.; Komoltri, C.; Pan-Ngum, W.; Chomanee, N.; Charngkaew, K.; Tangthawornchaikul, N.; Limpitikul, W.; Vasanawathana, S.; et al. Microparticles provide a novel biomarker to predict severe clinical outcomes of dengue virus infection. J. Virol. 2015, 89, 1587–1607. [Google Scholar] [CrossRef]
- Martínez-Rojas, P.P.; Monroy-Martínez, V.; Agredano-Moreno, L.T.; Jiménez-García, L.F.; Ruiz-Ordaz, B.H. Zika Virus-Infected Monocyte Exosomes Mediate Cell-to-Cell Viral Transmission. Cells. 2024, 13, 144. [Google Scholar] [CrossRef]
- Meerschaert, J.; Furie, M.B. The adhesion molecules used by monocytes for migration across endothelium include CD11a/CD18, CD11b/CD18, and VLA-4 on monocytes and ICAM-1, VCAM-1, and other ligands on endothelium. J. Immunol. 1995, 154, 4099–112. [Google Scholar] [CrossRef]
- Osterud, B. Cellular interactions in tissue factor expression by blood monocytes. Blood. Coagul. Fibrinolysis. 1995, 6, S20–5. [Google Scholar] [CrossRef]
- Herring, J.M.; McMichael, M.A.; Smith, S.A. Microparticles in health and disease. J. Vet. Intern. Med. 2013, 27, 1020–1033. [Google Scholar] [CrossRef] [PubMed]
- van Niel, G.; D'Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Nour, A.M.; Modis, Y. Endosomal vesicles as vehicles for viral genomes. Trends. Cell. Biol. 2014, 24, 449–54. [Google Scholar] [CrossRef] [PubMed]
- Bargeron Clark, K.; Hsiao, H.M.; Noisakran, S.; Tsai, J.J.; Perng, G.C. Role of microparticles in dengue virus infection and its impact on medical intervention strategies. Yale. J. Biol. Med. 2012, 85, 3–18. [Google Scholar]
- Latanova, A.; Karpov, V.; Starodubova, E. Extracellular Vesicles in Flaviviridae Pathogenesis: Their Roles in Viral Transmission, Immune Evasion, and Inflammation. Int. J. Mol. Sci. 2024, 25, 2144. [Google Scholar] [CrossRef]
- Ishikawa, T.; Narita, K.; Matsuyama, K.; Masuda, M. Dissemination of the Flavivirus Subgenomic Replicon Genome and Viral Proteins by Extracellular Vesicles. Viruses. 2024, 16, 524. [Google Scholar] [CrossRef]
- Puerta-Guardo, H.; Glasner, D.R.; Espinosa, D.A.; Biering, S.B.; Patana, M.; Ratnasiri, K.; Wang, C.; Beatty, P.R.; Harris, E. Flavivirus NS1 Triggers Tissue-Specific Vascular Endothelial Dysfunction Reflecting Disease Tropism. Cell. Rep. 2019, 26, 1598–1613.e8. [Google Scholar] [CrossRef]
- Shu, B.; Ooi, J.S.G.; Tan, A.W.K.; Ng, T.S.; Dejnirattisai, W.; Mongkolsapaya, J.; Fibriansah, G.; Shi, J.; Kostyuchenko, V.A.; Screaton, G.R.; et al. CryoEM structures of the multimeric secreted NS1, a major factor for dengue hemorrhagic fever. Nat. Commun. 2022, 13, 6756. [Google Scholar] [CrossRef]
- Safadi, D.E.; Lebeau, G.; Lagrave, A.; Mélade, J.; Grondin, L.; Rosanaly, S.; Begue, F.; Hoareau, M.; Veeren, B.; Roche, M.; et al. Extracellular Vesicles Are Conveyors of the NS1 Toxin during Dengue Virus and Zika Virus Infection. Viruses. 2023, 15, 364. [Google Scholar] [CrossRef]
- Huerta-Zepeda, A.; Cabello-Gutiérrez, C.; Cime-Castillo, J.; Monroy-Martínez, V.; Manjarrez-Zavala, M.E.; Gutiérrez-Rodríguez, M.; Izaguirre, R.; Ruiz-Ordaz, B.H. Crosstalk between coagulation and inflammation during Dengue virus infection. Thromb. Haemost. 2008, 99, 936–43. [Google Scholar] [CrossRef]
- World Health Organization, Ministry of Health of Democratic Republic of Timor-Leste. National Guideline for Clinical Management of Dengue 2022. Timor-Leste: World Health Organization. Available online: https://cdn.who.int/media/docs/default-source/2021-dha-docs/5_national-clinical-guideline-of-dengue-timor-leste_clean_final-12-dec-2022.pdf?sfvrsn=e80bf9cd_1 (accessed on 1 April 2026).
- Nascimento, E.J.; Hottz, E.D.; Garcia-Bates, T.M.; Bozza, F.; Marques, E.T., Jr.; Barratt-Boyes, S.M. Emerging concepts in dengue pathogenesis: interplay between plasmablasts, platelets, and complement in triggering vasculopathy. Crit. Rev. Immunol. 2014, 34, 227–40. [Google Scholar] [CrossRef]
- Pozo-Aguilar, J.O.; Monroy-Martínez, V.; Díaz, D.; Barrios-Palacios, J.; Ramos, C.; Ulloa-García, A.; García-Pillado, J.; Ruiz-Ordaz, B.H. Evaluation of host and viral factors associated with severe dengue based on the 2009 WHO classification. Parasit. Vectors. 2014, 7, 590. [Google Scholar] [CrossRef]
- Rovai, E.S.; Alves, T.; Holzhausen, M. Protease-activated receptor 1 as a potential therapeutic target for COVID-19. Exp. Biol. Med. (Maywood) 2021, 246, 688–694. [Google Scholar] [CrossRef] [PubMed]
- Flaumenhaft, R. Protease-Activated Receptor-1 Signaling: The Big Picture. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1809–1811. [Google Scholar] [CrossRef]
- Mutiara Koh, S.C.L.; Bachtiar, A.; Hariman, H. The Vascular Endothelium in Patients with Dengue Haemorrhagic Fever. Open Access Maced. J. Med. Sci. 2019, 7, 2221–2225. [Google Scholar] [CrossRef] [PubMed]
- Ghita, L.; Yao, Z.; Xie, Y.; Duran, V.; Cagirici, H.B.; Samir, J.; Osman, I.; Rebellón-Sánchez, D.E.; Agudelo-Rojas, O.L.; Sanz, A.M.; et al. Global and cell type-specific immunological hallmarks of severe dengue progression identified via a systems immunology approach. Nat. Immunol. 2023, 24, 2150–2163. [Google Scholar] [CrossRef]
- Martínez-Rojas, P.P.; Monroy-Martínez, V.; Ruiz-Ordaz, B.H. Role of extracellular vesicles in the pathogenesis of mosquito-borne flaviviruses that impact public health. J. Biomed. Sci. 2025, 32, 4. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Lata, S.; Ali, A.; Banerjea, A.C. Dengue haemorrhagic fever: a job done via exosomes? Emerg. Microbes. Infect. 2019, 8, 1626–1635. [Google Scholar] [CrossRef]
- Martinez-Palomo, A.; Brailovski, C. Surface layer in tumor cells transformed by adeno-12 and SV40 viruses. Virology. 1968, 34, 379–82. [Google Scholar] [CrossRef]
- Lässer, C.; Eldh, M.; Lötvall, J. Isolation and characterization of RNA-containing exosomes. J. Vis. Exp. 2012, 59, e3037. [Google Scholar] [CrossRef]
- Nieuwland, R.; Berckmans, R.J.; McGregor, S.; Böing, A.N.; Romijn, F.P.; Westendorp, R.G.; Hack, C.E.; Sturk, A. Cellular origin and procoagulant properties of microparticles in meningococcal sepsis. Blood. 2000, 95, 930–935. [Google Scholar] [CrossRef] [PubMed]
- Berckmans, R.J.; Nieuwland, R.; Böing, A.N.; Romijn, F.P.; Hack, C.E.; Sturk, A. Cell-derived microparticles circulate in healthy humans and support low grade thrombin generation. Thromb. Haemost. 2001, 85, 639–646. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



