Submitted:
30 April 2026
Posted:
02 May 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
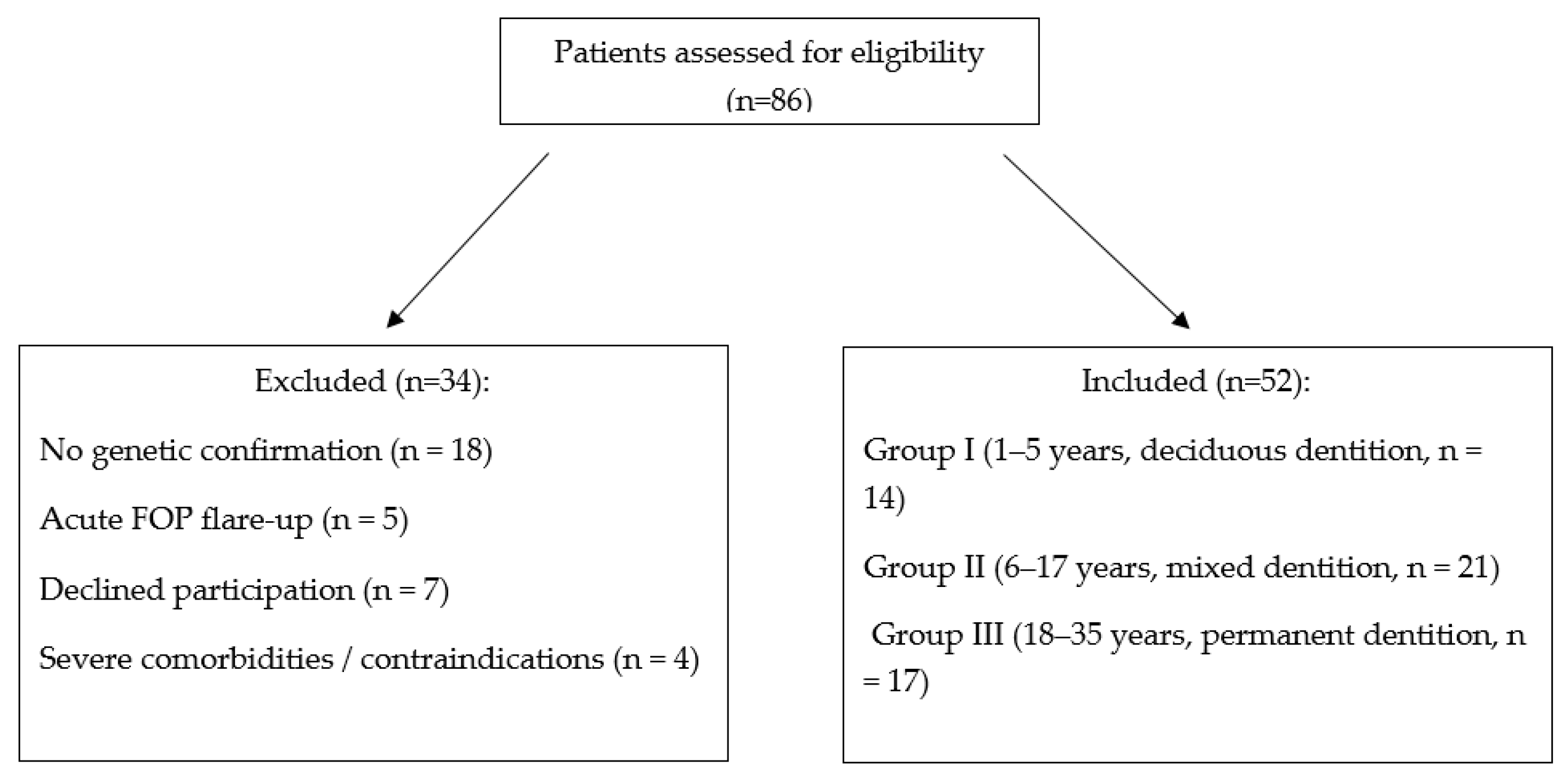
2.1. Study Design and Setting
2.2. Participants
2.3. Age Grouping and Dentition
- Group I (n = 14): 1–5 years, deciduous dentition.
- Group II (n = 21): 6–17 years, mixed dentition.
- Group III (n = 17): 18–35 years, permanent dentition.
2.4. Clinical and Dental Examination
- Intraoral examination: caries was recorded using the DMFT index according to the World Health Organization (WHO) 2013 criteria. Chronic stomatitis was diagnosed based on clinical appearance (erythema, atrophy, or lichenoid lesions persisting for >3 months). Enamel hypoplasia was recorded as present/absent. Malocclusion was classified according to Angle’s classification.
- Oral hygiene: OHI-S (Greene & Vermillion, 1964) was used. Poor hygiene was defined as OHI-S > 2.0.
- Temporomandibular joint (TMJ) assessment: maximal mouth opening (interincisal distance, mm) was measured with a calibrated caliper. TMJ disorders were defined as: limitation of mouth opening (<30 mm in adults, <20 mm in children <6 years), painful jaw movement, or presence of crepitation/clicks on palpation or auscultation. For Group II (6–17 years), the same adult threshold of <30 mm was applied, recognising that this may underestimate limitation in younger children; therefore, the reported prevalence (57.1%) is a conservative estimate.
- Sialometry and pH-metry: unstimulated whole saliva was collected by spitting method for 5 minutes. Salivary flow rate (ml/min) was calculated (normal reference: 0.3–0.4 ml/min). Salivary pH was measured immediately using an electronic pH meter (Hanna HI 98107). The presence of calcium phosphate crystals was assessed by light microscopy of air-dried saliva smears.
- CT densitometry: cone-beam computed tomography of the maxillofacial region was performed using a standard protocol (120 kV, 5 mA, voxel size 0.2 mm). Bone and soft-tissue calcification were evaluated using Ez3D2009 software with 3D reconstructions. Hounsfield units (HU) were measured in predefined regions of interest (parotid gland, masseter muscle, TMJ capsule). The degree of calcification was graded as: 0 = none, 1 = mild (<200 HU), 2 = moderate (200–400 HU), 3 = severe (>400 HU).
2.5. Quality of Life Assessment
2.6. Sample Size Calculation
2.7. Statistical Analysis
2.8. Bias Control
3. Results
3.1. Baseline Characteristics of the Study Population
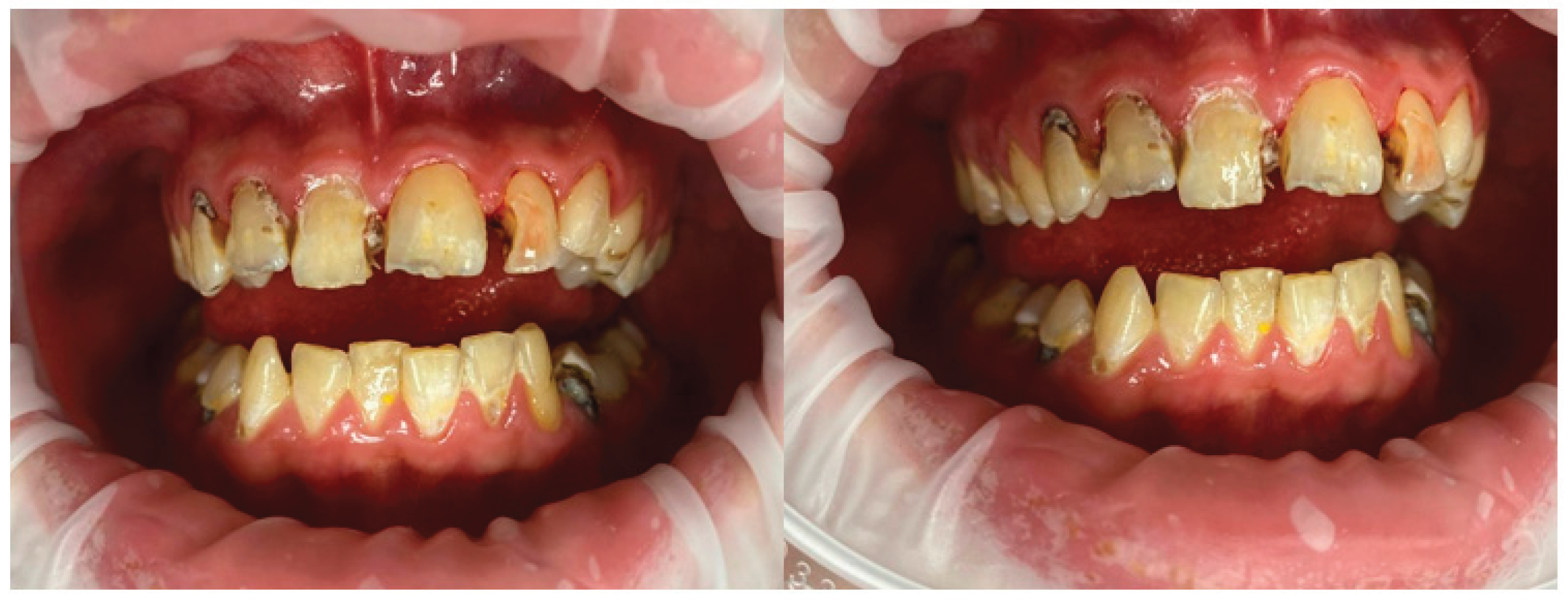
3.2. Prevalence of Dental Pathologies
3.3. Salivary Changes and Correlation with Tissue Calcification
3.4. Temporomandibular Joint Disorders
| Type of disorder | Group I (n=14) |
Group II (n=21) |
Group III (n=17) |
Control group (n=156) 1 |
p value (FDR adj) |
| Total TMJ disorders, % (95% CI) |
7.1 (0.2–33.9) | 71.4 (47.8–88.7) | 100 (80.5–100) | 8.3 (4.5–13.8) | <0.001 |
| Limitation of mouth opening (<30 mm), % (95% CI) | 0 (0.0–23.2) | 57.1 (34.0–78.2) | 88.2 (63.6–98.5) | 2.6 (0.7–6.5) | <0.001 |
| Painful jaw movement, % (95% CI) | 0 (0.0–23.2) | 33.3 (14.6–57.0) | 58.8 (32.9–81.6) | 5.8 (2.7–10.7) | <0.001 |
| Crepitation / clicks, % (95% CI) |
0 (0.0–23.2) | 23.8 (8.2–47.2) | 47.1 (23.0–72.2) | 3.2 (1.0–7.3) | <0.001 |
3.5. Quality of Life (OHIP 14)
4. Discussion
4.1. Age Dependent Progression of TMJ Dysfunction and Stomatitis
4.2. Salivary Changes and Correlation with Calcification
4.3. High Caries Burden and Enamel Hypoplasia
4.4. Malocclusion and Its Functional Consequences
4.5. Quality of Life Impact
4.6. Comparison with International Registry and Case Series
4.7. Clinical Phenotype of a FOP Patient in Dental Practice
- High caries prevalence (DMFT ≥ 4 in >85%) despite variable hygiene.
- Chronic stomatitis – universal (100%) in adults, likely fibrotic.
- Enamel hypoplasia – present in >50% of adults.
- Angle class II malocclusion – in approximately half of patients over 6 years.
- Progressive TMJ dysfunction – mouth opening declines by ~0.5 mm per year of age (estimated from our cross-sectional data).
- Salivary abnormalities – reduced flow rate and calcium phosphate crystals in ~18% of adults, correlating with CT calcification.
- Severely impaired oral health-related quality of life – OHIP-14 total score >45 in adults.
4.8. Practical Recommendations for Dentists
- Suspicion and early diagnosis: look for valgus deformity of the great toes, painful soft tissue nodules, progressive limitation of mouth opening, and the dental features listed above. Confirm with genetic testing for ACVR1 (R206H) [16].
- Non invasive caries management: prefer ICON resin infiltration or laser ablation over drilling whenever possible [22].
- Follow up: CT of the maxillofacial region every 12 months to detect early heterotopic ossification of the TMJ and salivary glands [23].
4.9. Strengths and Limitations
4.10. Future Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| FOP | Fibrodysplasia Ossificans Progressiva |
| DMFT | Decayed, Missing, Filled Teeth |
| OHI-S | Oral Hygiene Index – Simplified |
| TMJ | Temporomandibular Joint |
| CT | Computed Tomography |
| FDR | False Discovery Rate |
| ACVR1 | Activin A receptor type I |
| BMP | Bone Morphogenetic Protein |
| WHO | World Health Organization |
| STROBE | Strengthening the Reporting of Observational Studies in Epidemiology |
| OHStat | Oral Health Statistics (guidelines) |
| ICC | Intraclass Correlation Coefficient |
| CI | Confidence Interval |
| HU | Hounsfield Units |
| MID | Minimal Important Difference |
| SD | Standard Deviation |
| CPP-ACP | Casein Phosphopeptide – Amorphous Calcium Phosphate |
References
- Allen, R.S.; Jones, W.D.; Hale, M.; Warder, B.N.; Shore, E.M.; Mullins, M.C. Reduced GS domain serine/threonine requirements of fibrodysplasia ossificans progressiva mutant type I BMP receptor ACVR1 in the zebrafish. J. Bone Mineral. Res. 2023, 38, 1364–1385. [Google Scholar] [CrossRef]
- Yang, S.; Cui, R.; Li, J.; Dai, R. Challenges in the diagnosis of fibrodysplasia ossificans progressiva with the ACVR1 mutation (c. 774G> C, p. R258S): a case report and review of literature. Orphanet J. Rare Dis. 2024, 19, 360. [Google Scholar] [CrossRef]
- Szoszkiewicz, A.; Szczepanek, M.; Bukowska-Olech, E.; Sowińska-Seidler, A.; Socha, M.; Jamsheer, A. Fibrodysplasia ossificans progressiva: genetic and clinical characterization in a cohort of Polish patients and review of potential therapies. J. Appl. Genet. 2025, 1. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, F.S.; Shore, E.M.; Pignolo, R.J. Fibrodysplasia ossificans progressiva emerges from obscurity. Trends Mol. Med. 2025, 31, 106–116. [Google Scholar] [CrossRef]
- Anwar, S.; Yokota, T. Navigating the complex landscape of fibrodysplasia ossificans progressiva: from current paradigms to therapeutic frontiers. Genes. 2023, 14, 2162. [Google Scholar] [CrossRef]
- Kaplan, F.S.; Xu, M.; Seeman, P.; et al. Classic and atypical fibrodysplasia ossificans progressiva (FOP) phenotypes are caused by mutations in the bone morphogenetic protein (BMP) type I receptor ACVR1. Hum. Mutat. 2009, 30, 379–390. [Google Scholar] [CrossRef] [PubMed]
- Towler, O.W.; Shore, E.M. BMP signaling and skeletal development in fibrodysplasia ossificans progressiva (FOP). Dev. Dyn. 2022, 251, 164–177. [Google Scholar] [CrossRef]
- Susami, T.; Kubota, S.; Moriyama, K.; et al. Facial morphology and occlusion of a patient with fibrodysplasia ossificans progressiva (FOP): a case report. Spec. Care Dent. 2012, 32, 165–170. [Google Scholar] [CrossRef] [PubMed]
- Kriegbaum, R.K.; Hillerup, S. Fibrodysplasia ossificans progressiva (FOP): report of a case with extra-articular ankylosis of the mandible. J. Cranio-Maxillo-Facial Surg. 2013, 41, 856–860. [Google Scholar] [CrossRef]
- Nussbaum, B.L.; O’Hara, I.; Kaplan, F.S. Fibrodysplasia ossificans progressiva: report of a case with guidelines for pediatric dental and anesthetic management. ASDC J. Dent. Child. 1996, 63, 448–450. [Google Scholar]
- Kilmartin, E.; Grunstein, I.; Al Mukaddam, M.; et al. General anesthesia for dental procedures in patients with fibrodysplasia ossificans progressiva: a review of 42 cases in 30 patients. Anesth. Analg. 2014, 118, 298–301. [Google Scholar] [CrossRef]
- Chakkalakal, S.A.; Zhang, D.; Culbert, A.L.; et al. An Acvr1 R206H knock-in mouse has fibrodysplasia ossificans progressiva. J. Bone Mineral. Res. 2012, 27, 1746–1756. [Google Scholar] [CrossRef] [PubMed]
- Botman, E.; de Vries, J.J.; Schober, P.; et al. Evolution of heterotopic bone in fibrodysplasia ossificans progressiva: An [18F]NaF PET/CT study. Bone 2019, 124, 1–6. [Google Scholar] [CrossRef]
- Mejias Rivera, L.; Shore, E.M.; Mourkioti, F. Cellular and molecular mechanisms of heterotopic ossification in fibrodysplasia ossificans progressiva. Biomedicines 2024, 12, 779. [Google Scholar] [CrossRef]
- Schoenmaker, T.; Bouchankouk, A.D.; Özkan, S.; et al. Limitations of Jaw Movement in Fibrodysplasia Ossificans Progressiva: A Review. Front Med. 2022, 9, 852678. [Google Scholar] [CrossRef]
- Pignolo, R.J.; Shore, E.M.; Kaplan, F.S. Fibrodysplasia Ossificans Progressiva: Clinical and Genetic Aspects. Orphanet J. Rare Dis. 2011, 6, 80. [Google Scholar] [CrossRef]
- Oliveira, F.A.F.; Fernandes, C.P.; Araujo, K.S.B.; Alves, A.; Sousa, F.; Mota, M. Clinical aspects and conservative dental management of a patient with fibrodysplasia ossificans progressiva. J. Contemp. Dent. Pract. 2014, 15, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.; Stephen, L.; Scott, C.; Urban, M.; Sudi, S.; Beighton, P. Fibrodysplasia ossificans progressiva (FOP) in South Africa: dental implications in 5 cases. Oral. Surg. Oral. Med. Oral. Pathol. Oral. Radiol. Endod. 2011, 112, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Gluhak-Heinrich, J.; Guo, D.; Yang, W.; et al. New roles and mechanism of action of BMP4 in postnatal tooth cytodifferentiation. Bone 2010, 46, 1533–1545. [Google Scholar] [CrossRef]
- Bennadi, D.; Reddy, C.V.K. Oral health related quality of life. J. Int. Soc. Prev. Community Dent. 2013, 3, 1–6. [Google Scholar] [CrossRef]
- Pignolo, R.; Cheung, K.; Kile, S.; et al. Self-reported baseline phenotypes from the International Fibrodysplasia Ossificans Progressiva (FOP) Association Global Registry. Bone 2020, 134, 115274. [Google Scholar] [CrossRef]
- Hietanen, B.; Sullivan, M.; Frustino, J.; Cantie, S.; Kapral, E. Dental management of a 26-year-old female with fibrodysplasia ossificans progressiva: A case report. Spec. Care Dent. 2021, 42, 194–199. [Google Scholar] [CrossRef]
- Rapekta, S.; Antakov, G.; Khokhryakov, V.I.; Sultanova, S.A.; Liskov, N.B. Experience in Conducting Oral Cavity Surgery in a Patient with Fibrodysplasia Ossificans Progressiva. Aktual. Probl. Meditsiny 2024, 47, 502–513. [Google Scholar] [CrossRef]
- Tucker-Bartley, A.; Lemme, J.; Gomez-Morad, A.; et al. Pain Phenotypes in Rare Musculoskeletal and Neuromuscular Diseases. Neurosci. Biobehav Rev. 2021, 124, 267–290. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.S.; Xie, J.; Wang, D.; et al. Suppression of heterotopic ossification in fibrodysplasia ossificans progressiva using AAV gene delivery. Nat. Commun. 2022, 13, 6175. [Google Scholar] [CrossRef]
- Kaplan, F.S.; Al Mukaddam, M.; Pignolo, R.J. Acute unilateral hip pain in fibrodysplasia ossificans progressiva (FOP). Bone 2018, 109, 115–119. [Google Scholar] [CrossRef]
- Pignolo, R.J.; Bedford-Gay, C.; Liljesthröm, M.; et al. The natural history of fibrodysplasia ossificans progressiva: A prospective, global 36-month study. Genet Med. 2022, 24, 2422–2433. [Google Scholar] [CrossRef] [PubMed]
- Convente, M.R.; Wang, H.; de Carvalho, C.E.; et al. Depletion of Mast Cells and Macrophages Impairs Heterotopic Ossification in an Acvr1R206H Mouse Model of Fibrodysplasia Ossificans Progressiva. J. Bone Min. Res. 2018, 33, 269–282. [Google Scholar] [CrossRef]
- Barruet, E.; Morales, B.M.; Cain, C.J.; et al. The ACVR1 R206H mutation found in fibrodysplasia ossificans progressiva increases human induced pluripotent stem cell-derived endothelial cell formation. Stem Cell. Res. Ther. 2016, 7, 115. [Google Scholar] [CrossRef]
- Eekhoff, E.M.W.; Botman, E.; Coen Netelenbos, J.; et al. [18F]NaF PET/CT scan as an early marker of heterotopic ossification in fibrodysplasia ossificans progressiva. Bone 2018, 109, 143–146. [Google Scholar] [CrossRef]
| Characteristic |
Group I (n=14) |
Group II (n=21) |
Group III (n=17) | p value 1 |
| Age, years, mean (standard deviation (SD)) |
3.2 (1.4) | 11.5 (3.1) | 26.4 (5.2) | <0.001 |
| Female, n (%) | 8 (57.1) | 12 (57.1) | 10 (58.8) | 0.99 |
| Disease duration, years, mean (SD) | 2.1 (1.2) | 8.9 (3.4) | 18.3 (6.1) | <0.001 |
| ACVR1 R206H mutation, n (%) | 14 (100) | 20 (95.2) | 17 (100) | 0.54 |
| Prior invasive dental procedures, n (%) | 1 (7.1) | 8 (38.1) | 14 (82.4) | <0.001 +2 |
| Indicator |
Group I (n=14) |
Group II (n=21) |
Group III (n=17) | Control group (n=156) 1 |
p value (FDR adj) |
Cramér’s V |
| Caries (DMFT ≥ 4), % (95% CI) | 85.7 (56.2–98.0) | 85.7 (63.7–96.9) | 82.4 (56.6–96.2) | 38.5 (30.7–46.6) | >0.05 | 0.07 |
| Chronic stomatitis, % (95% CI) | 7.1 (0.2–33.9) | 38.1 (18.1–61.6) | 100 (80.5–100) | 6.4 (3.1–11.5) | <0.001 | 0.72 |
| Enamel hypoplasia, % (95% CI) | 21.4 (4.7–50.8) | 42.9 (21.8–66.0) | 58.8 (32.9–81.6) | 4.5 (1.8–9.1) | 0.03 | 0.36 |
| Malocclusion (Angle II), % (95% CI) |
0 (0.0–23.2) | 47.6 (25.7–70.2) | 47.1 (23.0–72.2) | 14.7 (9.6–21.3) | 0.008 | 0.52 |
| OHI S > 2.0, % (95% CI) |
21.4 (4.7–50.8) | 28.6 (11.3–52.2) | 29.4 (10.3–55.9) | 11.5 (7.0–17.7) | 0.049 | 0.31 |
| Domain |
Group I (n=14) |
Group II (n=21) |
Group III (n=17) |
Control (adults, n=46)2 | p-value3 |
| Physical functioning | 78.4 (74.5–82.3) | 52.1 (48.8–55.4) | 41.3 (38.3–44.3) | 84.6 (81.4–87.8) | <0.001 |
| Emotional state | 85.0 (82.7–87.3) | 63.5 (60.2–66.8) | 34.7 (32.2–37.2) | 80.3 (77.5–83.1) | <0.001 |
| Social activity | 89.2 (86.8–91.6) | 70.8 (68.0–73.6) | 48.9 (45.5–52.3) | 81.5 (78.6–84.4) | <0.001 |
| Total OHIP 14 score | 12.4 (10.0–14.8) | 34.6 (30.7–38.5) | 48.9 (45.4–52.4) | 13.8 (11.5–16.1) | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




