Submitted:
28 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Statistical Analysis
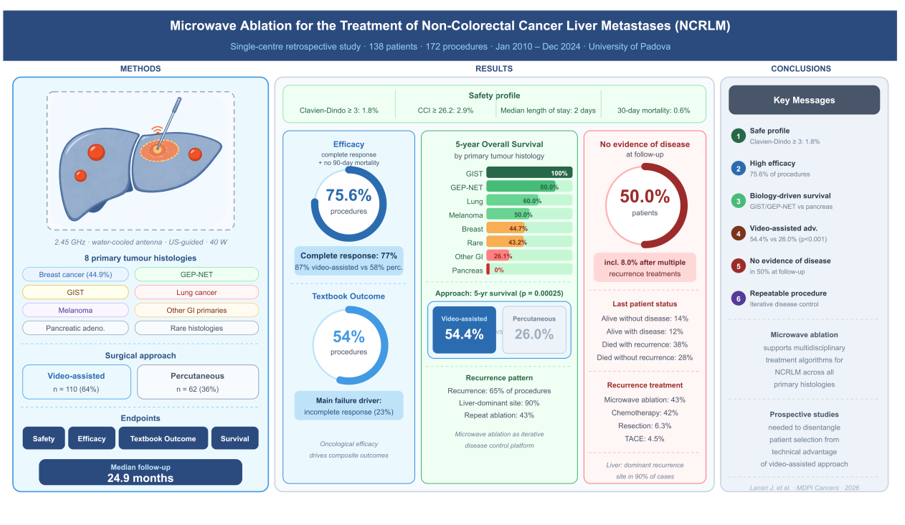
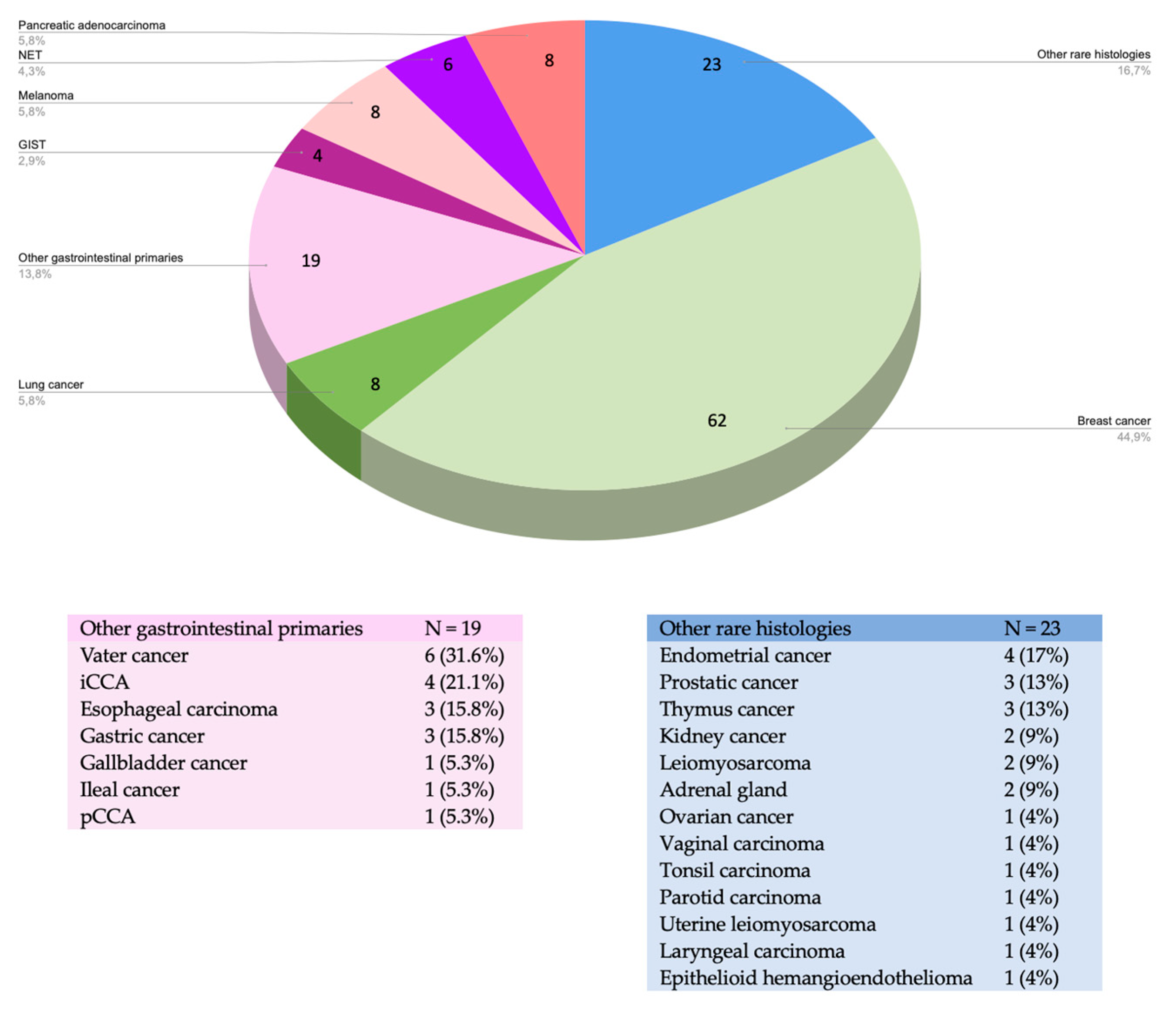
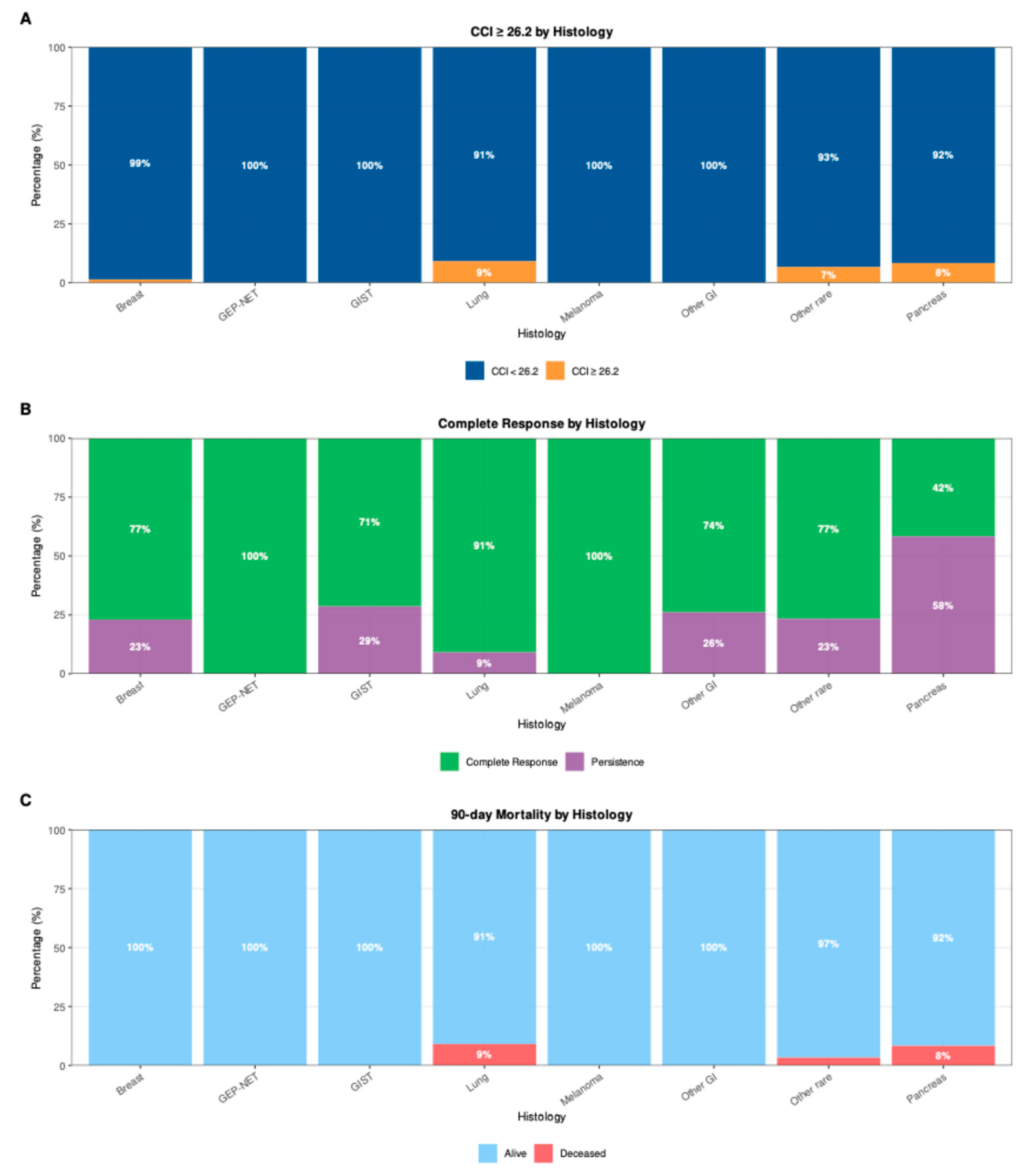
3. Results
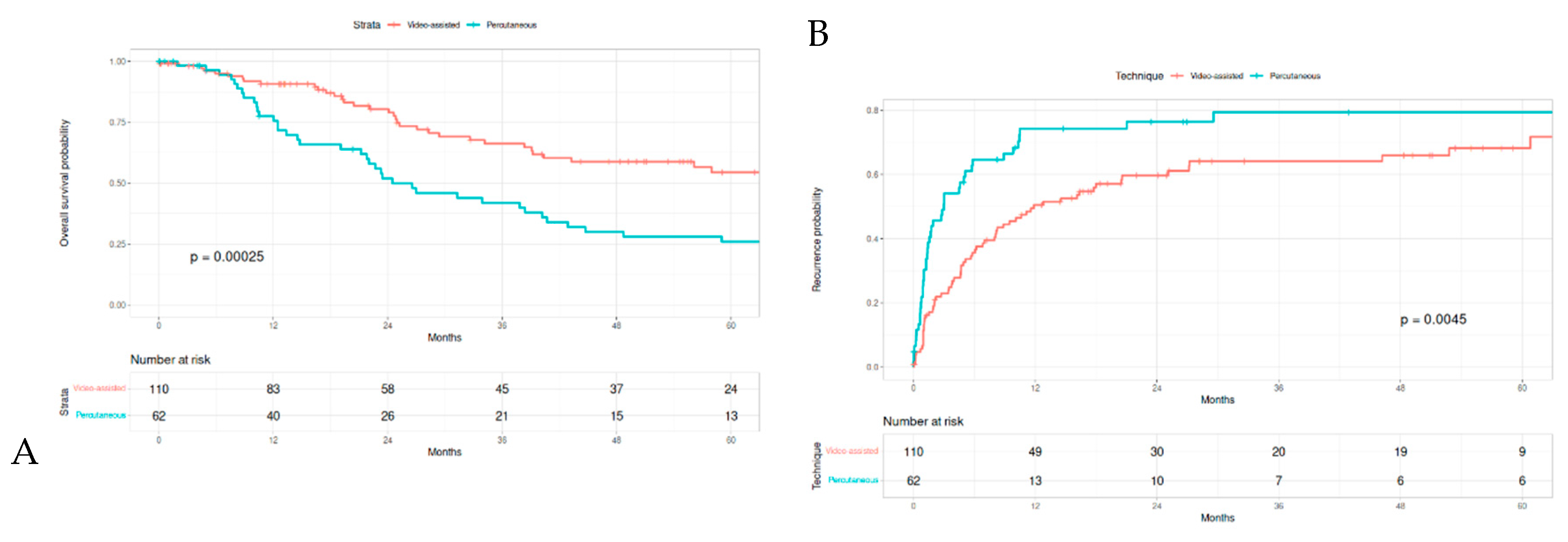
3.1. Survival Analysis
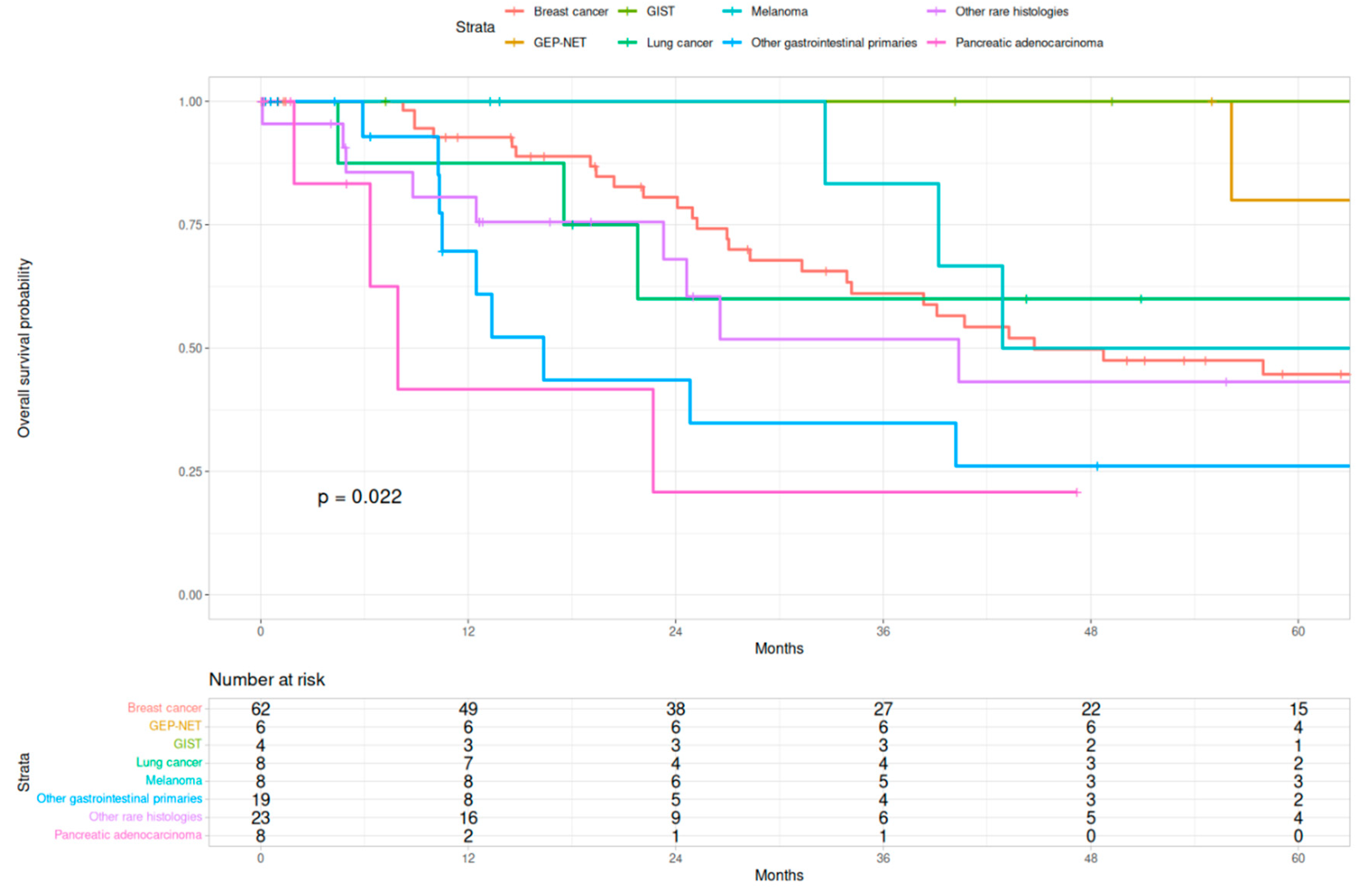
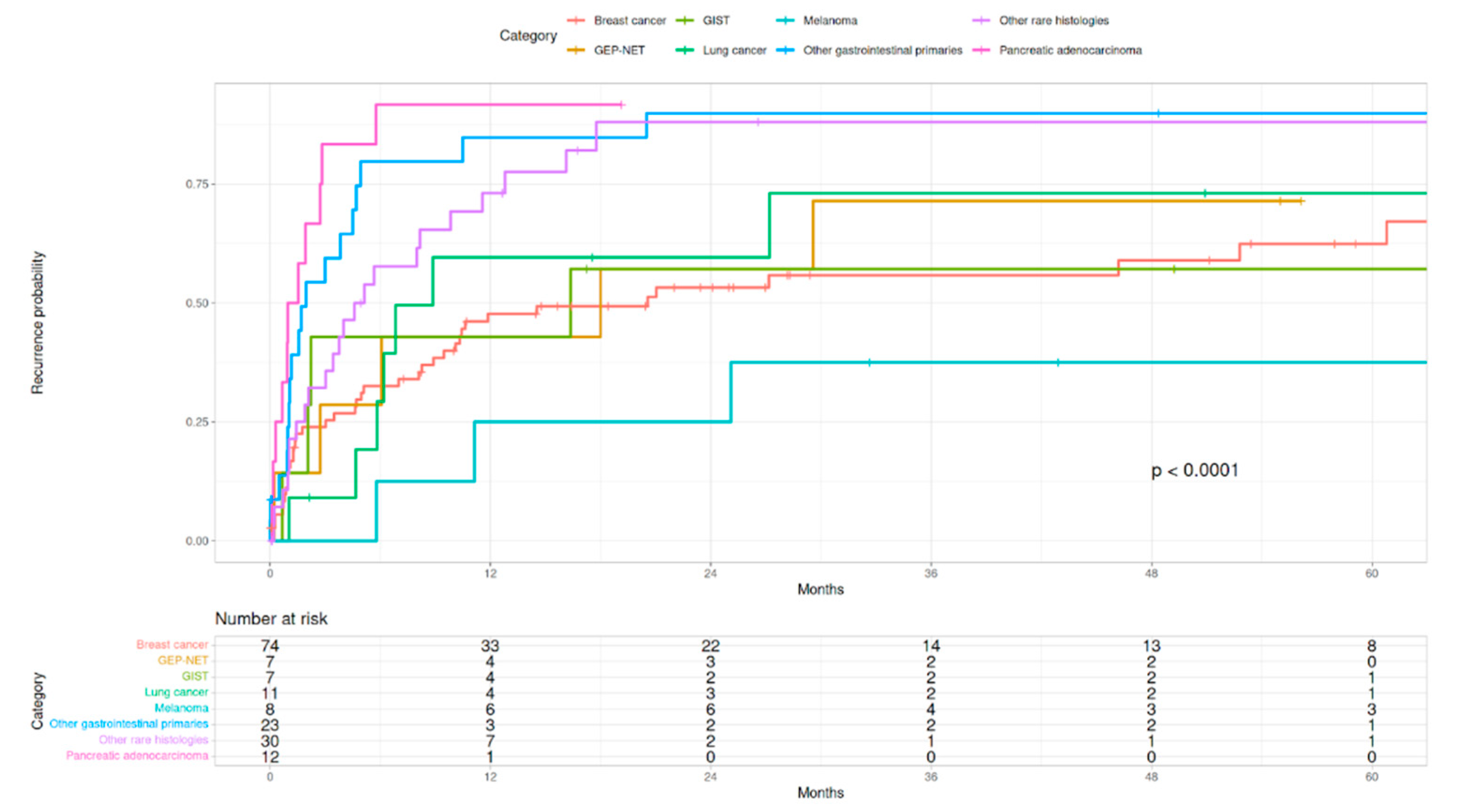
3.2. Factors Associated with Procedure Efficacy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BSC | Best Supportive Care |
| CCA | Cholangiocarcinoma |
| CCI | Comprehensive Complication Index |
| CR | Complete Response |
| CRLM | Colorectal Cancer Liver Metastases |
| CSPH | Clinically Significant Portal Hypertension |
| ECOG PS | Eastern Cooperative Oncology Group Performance Status |
| GEP-NET | Gastro-Entero-Pancreatic Neuroendocrine Tumours |
| GIST | Gastrointestinal Stromal Tumour |
| HCC | Hepatocellular Carcinoma |
| HCV | Hepatitis C Virus |
| ICU | Intensive Care Unit |
| LOS | Length of Hospital Stay |
| LR | Liver Resection |
| MWA | Microwave Ablation |
| NAFLD | Non-Alcoholic Fatty Liver Disease |
| NCRLM | Non-Colorectal Cancer Liver Metastasis |
| NED | Non-Evidence of Disease |
| OS | Overall Survival |
| PEI | Percutaneous Ethanol Injection |
| pRBC | Packed Red Blood Cells |
| RFA | Radiofrequency Ablation |
| TACE | Trans-Arterial Chemoembolization |
References
- Lanari, J.; Caregari, S.; Billato, I.; Gringeri, E.; D’Amico, F.; Gemo, G.; et al. Textbook Outcome of Laparoscopic Microwave Ablation for Hepatocellular Carcinoma. Cancers 2023, 15(2), 436. [Google Scholar] [CrossRef] [PubMed]
- Cillo, U.; Lanari, J.; Masutti, M.; D’Amico, F.E.; Vitale, A.; Gringeri, E. Percutaneous and Laparoscopic-Assisted Ablation of Hepatocellular Carcinoma; Hepatocellular Carcinoma [Internet]; Ettorre, G.M., Ed.; Springer International Publishing: Cham, Switzerland, 2023; pp. 63–70, [cited 2025 Oct 30]. [Google Scholar] [CrossRef]
- Cillo, U.; Bertacco, A.; Fasolo, E.; Carandina, R.; Vitale, A.; Zanus, G.; et al. Videolaparoscopic microwave ablation in patients with HCC at a European high-volume center: Results of 815 procedures. J. Surg. Oncol. 2019, 120(6), 956–65. [Google Scholar] [CrossRef] [PubMed]
- Del Basso, C.; Usai, S.; Levi Sandri, G.B. Non-colorectal non-neuroendocrine liver metastasis: a narrative review of surgical treatment. Chin. Clin. Oncol. 2022, 11(4), 28. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, G.M.; Braghiroli, M.I.F.M.; Pirola Kruger, J.A.; Coelho, F.F.; Herman, P. Is There a Role for Locoregional Therapies for Non-colorectal Gastrointestinal Malignancies? Hematol. Oncol. Clin. North Am. 2025, 39(1), 125–41. [Google Scholar] [CrossRef] [PubMed]
- Cillo, U.; Noaro, G.; Vitale, A.; Neri, D.; D’Amico, F.; Gringeri, E.; et al. Laparoscopic microwave ablation in patients with hepatocellular carcinoma: a prospective cohort study. HPB 2014, 16(11), 979–986. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Morioka, D.; Conci, S.; Margonis, G.A.; Sawada, Y.; Ruzzenente, A.; et al. The Tumor Burden Score: A New “Metro-ticket” Prognostic Tool For Colorectal Liver Metastases Based on Tumor Size and Number of Tumors. Ann. Surg. 2018, 267(1), 132. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J. Chronic Dis. 1987, 40(5), 373–383. [Google Scholar] [CrossRef] [PubMed]
- 9 Rosenthal, R.; Hoffmann, H.; Clavien, P.A.; Bucher, H.C.; Dell-Kuster, S. Definition and Classification of Intraoperative Complications (CLASSIC): Delphi Study and Pilot Evaluation. World J. Surg. 2015, 39(7), 1663–1671. [Google Scholar] [CrossRef] [PubMed]
- Balzan, S.; Belghiti, J.; Farges, O.; Ogata, S.; Sauvanet, A.; Delefosse, D.; et al. The “50-50 Criteria” on Postoperative Day 5. Ann. Surg. 2005, 242(6), 824–829. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240(2), 205–213. [Google Scholar] [CrossRef] [PubMed]
- Slankamenac, K.; Graf, R.; Barkun, J.; Puhan, M.A.; Clavien, P.A. The comprehensive complication index: a novel continuous scale to measure surgical morbidity. Ann. Surg. 2013, 258(1), 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cloyd, J.M.; Mizuno, T.; Kawaguchi, Y.; Lillemoe, H.A.; Karagkounis, G.; Omichi, K.; et al. Comprehensive Complication Index Validates Improved Outcomes Over Time Despite Increased Complexity in 3707 Consecutive Hepatectomies. Ann. Surg. 2020, 271(4), 724–731. [Google Scholar] [CrossRef] [PubMed]
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines on the management of hepatocellular carcinoma. J. Hepatol. 2025, 82(2), 315–374. [Google Scholar] [CrossRef] [PubMed]
- Serra, C.; Cossiga, V.; Serenari, M.; Felicani, C.; Mazzotta, E.; Pinato, D.J.; et al. Safety and efficacy of percutaneous radiofrequency ablation for hepatocellular carcinoma: a textbook outcome analysis. HPB 2022, 24(5), 664–671. [Google Scholar] [CrossRef] [PubMed]
- van der Lei, S.; Puijk, R.S.; Dijkstra, M.; Schulz, H.H.; Vos, D.J.W.; De Vries, J.J.J.; et al. Thermal ablation versus surgical resection of small-size colorectal liver metastases (COLLISION): an international, randomised, controlled, phase 3 non-inferiority trial. Lancet Oncol. 2025, 26(2), 187–199. [Google Scholar] [CrossRef] [PubMed]
- Frilling, A.; Modlin, I.M.; Kidd, M.; Russell, C.; Breitenstein, S.; Salem, R.; Kwekkeboom, D.; Lau, W.Y.; Klersy, C.; Vilgrain, V.; Davidson, B.; Siegler, M.; Caplin, M.; Solcia, E.; Schilsky, R. Working Group on Neuroendocrine Liver Metastases. Recommendations for management of patients with neuroendocrine liver metastases. Lancet Oncol. 2014, 15(1), e8–e21. [Google Scholar] [CrossRef] [PubMed]
- Torielli, P.; McGale, J.; Liao, M.J.; Rhaiem, R.; Bouche, O.; Botsen, D.; Gerin, O.; Lamane, A.; Lawrence, Y.; Madelis, G.; Rozenblum, L.; Sajan, A.; Tordjman, M.; Dercle, L.; Beddok, A. Hepatic metastases management: A comparative review of surgical resection, thermal ablation, and stereotactic body radiation therapy. Eur. J. Cancer 2025, 228, 115691. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, G.; Ruiz, E.M.; Dijkstra, M.; Gentile, N.T.; Donahue, D.; Gandhi, R.T.; Mahtani, R.L.; Mautner, S.; van den Bemd, B.A.T. Clinical Efficacy of Percutaneous Image-Guided Ablation in Breast Cancer Metastases to the Liver. Cancers 2025, 17(23), 3823. [Google Scholar] [CrossRef] [PubMed]
- Hallemeier, C.L.; Sharma, N.; Anker, C.; Selfridge, J.E.; Lee, P.; Jabbour, S.; Williams, V.; Liu, D.; Kennedy, T.; Jethwa, K.R.; Kim, E.; Kumar, R.; Small, W., Jr.; Tchelebi, L.; Russo, S. American Radium Society Appropriate Use Criteria for the use of liver-directed therapies for nonsurgical management of liver metastases: Systematic review and guidelines. Cancer 2023, 129(20), 3193–3212. [Google Scholar] [CrossRef] [PubMed]
| Variables | N = 1381 |
|---|---|
| Age (years) | 61.5 (51.0, 70.0) |
| Sex (female) | 100 (72%) |
| ECOG PS ≥ 2 *missing | 4 (3.1%) *10 |
| CCI ≥ 7 *missing | 98 (73%) *4 |
| Diabetes (yes) *missing | 12 (8.8%) *1 |
| Cirrhosis (yes) | 4 (2.9%) |
| HCV positive | 1 (0.7%) |
| NAFLD | 2 (1.4%) |
| CSPH | 2 (1.4%) |
| Last patient status | |
| Died with recurrence | 52 (38%) |
| Died without recurrence | 38 (28%) |
| Alive with disease | 17 (12%) |
| Alive without disease | 20 (14%) |
| Alive without disease after last recurrence treatment | 11 (8.0%) |
| Mean follow-up (months) | 24.9 (10.3, 55.8) |
| 1 Median (Q1, Q3); n / N (%) | |
| Variable | Video-assisted N = 1101 |
Percutaneous N = 621 |
p2 |
|---|---|---|---|
| Pre-operative variables | |||
| Platelets (109/L) *missing | 208(165.0,267.0) *3 | 213(166.0,277.0) *7 | 0.81 |
| Bilirubin (umol/L) *missing | 10.4 (7.4, 16.4) *6 | 10 (6.9, 13.5) *11 | 0.23 |
| Nodules (n) | 2 (1.0, 3.0) | 1 (1.0, 2.0) | 0.001 |
| Major diameter | 2.3 (1.6, 3.2) | 1.9 (1.4, 3.5) | 0.36 |
| TBS | 3.4 (2.4, 5.2) | 2.7 (1.8, 4.3) | 0.009 |
| Bilobar disease | 22 (20%) | 10 (16%) | 0.53 |
| Major vassels invasion | 1 (0.9%) | 0 (0%) | >0.99 |
| Extrahepatic metastasis | 14 (13%) | 12 (19%) | 0.24 |
| Previous liver-dir. surgery | |||
| None | 76 (69%) | 37 (60%) | 0.27 |
| 1 | 21 (19%) | 17 (27%) | |
| 2 - 4 | 11 (9.9%) | 6 (9.6%) | |
| ≥5 | 2(1.8%) | 2 (3.2%) | |
| Previous liver-dir. treatments | 0.25 | ||
| None | 72 (65%) | 35 (56%) | |
| 1 | 23 (21%) | 19 (31%) | |
| 2 - 4 | 12 (10.9%) | 5 (8%) | |
| ≥5 | 3 (2.7%) | 3 (4.8%) | |
| Intra-operative variables | |||
| Radical Intent | 89 (81%) | 43 (69%) | 0.085 |
| Concurrent procedure | 48 (44%) | 3 (4.8%) | <0.001 |
| Operative time (min) *missing | 100 (80.0, 125.0) *2 | 25 (15.0, 32.5) *2 | <0.001 |
| Nodules treated *missing | 2 (1.0, 3.0) *2 | 1 (1.0, 1.0) *2 | <0.001 |
| Duration, Σ (min) *missing | 12 (8.0, 18.0) *2 | 7 (5.0, 10.0) *1 | <0.001 |
| Power (Watt) | 40 (40.0, 60.0) | 40 (40.0, 60.0) | 0.062 |
| Post-operative variables | |||
| LOS | 2.5 (2.0, 3.0) | 1 (1.0, 1.0) | <0.001 |
| Postoperative complications | 21 (19%) | 2 (3.2%) | 0.003 |
| Reoperation | 2 (1.8%) | 0 (0%) | 0.54 |
| Clavien-Dindo ≥ 3 | 3 (2.7%) | 0 (0%) | 0.70 |
| CCI > 26.2 | 5 (4.5%) | 0 (0%) | 0.16 |
| CR of the target lesion | 96 (87%) | 36 (58%) | <0.001 |
| TO (achieved) | 61 (55%) | 32 (52%) | 0.63 |
| 90-day mortality | 2 (1.8%) | 1 (1.6%) | >0.99 |
|
1 n / N (%); Median (Q1, Q3). 2 Pearson’s Chi-squared test; Fisher’s exact test; Wilcoxon rank sum test; NA. | |||
| Variable | N (%) |
|---|---|
| LOS > 3 | 27 (15.6%) |
| Fever | 13 (7.6%) |
| Nausea and vomiting | 7 (4.1%) |
| Pleural effusion | 2 (1.2%) |
| Pneumothorax | 0 (0.0%) |
| Ascites | 1 (0.6%) |
| Liver function impairment (50-50) | 1 (0.6%) |
| Hemoperitoneum | 1 (0.6%) |
| Readmission within 30 days | 9 (5.2%) |
| 30 days mortality | 1 (0.6%) |
| No CR | 40 (23%) |
| LOS, length of hospital stay; CR, complete response. | |
| Variable | OR | 95% CI | p |
|---|---|---|---|
| Breast cancer | 1.15 | 0.57, 2.36 | 0.7 |
| GIST | 0.80 | 0.17, 5.74 | 0.8 |
| Lung cancer | 1.49 | 0.36, 10.0 | 0.6 |
| Other gastrointestinal primaries | 0.90 | 0.35, 2.66 | 0.8 |
| Other rare histologies | 0.87 | 0.36, 2.23 | 0.8 |
| Pancreatic adenocarcinoma | 0.20 | 0.06, 0.66 | 0.009 |
| Age | 0.99 | 0.97, 1.02 | 0.7 |
| Sex (male) | 0.89 | 0.42, 1.94 | 0.8 |
| ECOG PS ≥ 2 | 0.16 | 0.02, 0.84 | 0.037 |
| Number of nodules: | |||
| • 1 | - | - | - |
| • 2 -3 | 1.02 | 0.46, 2.29 | >0.9 |
| • > 3 | 0.40 | 0.16, 1.03 | 0.054 |
| Major diameter | 0.96 | 0.82, 1.15 | 0.6 |
| TBS | 0.94 | 0.84, 1.04 | 0.2 |
| Bilobar disease | 0.54 | 0.24, 1.28 | 0.15 |
| Extrahepatic metastasis | 0.86 | 0.34, 2.34 | 0.7 |
| Previous Liver Resection | 0.30 | 0.13, 0.73 | 0.007 |
| Previous Liver Ablation | 0.77 | 0.37, 1.65 | 0.5 |
| Previous Liver Treatments | 0.66 | 0.33, 1.35 | 0.3 |
| Number of previous liver surgery | 0.76 | 0.58, 0.98 | 0.039 |
| Number of previous liver treatments | 0.80 | 0.62, 1.02 | 0.066 |
| Percutaneous approach | 0.24 | 0.11, 0.48 | <0.001 |
| Concurrent procedure | 1.47 | 0.68, 3.42 | 0.3 |
| Radical Surgical Intent | 2.33 | 1.07, 5.01 | 0.030 |
| Operative time (min) | 1.01 | 1.00, 1.02 | 0.013 |
| Nodules treated | 0.98 | 0.81, 1.21 | 0.8 |
| Duration, Σ (min) | 1.02 | 0.97, 1.07 | 0.5 |
| Power (Watt) | 1.01 | 0.99, 1.04 | 0.4 |
| LOS | 1.09 | 0.90, 1.39 | 0.4 |
| Postoperative complications | 1.63 | 0.57, 5.87 | 0.4 |
| Clavien-Dindo ≥ 3 | 0.56 | 0.09, 1.30 | 0.3 |
| CCI ≥ 26.2 | 1.30 | 0.19, 25.8 | 0.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




