Submitted:
30 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Structural Classification and Molecular Architecture
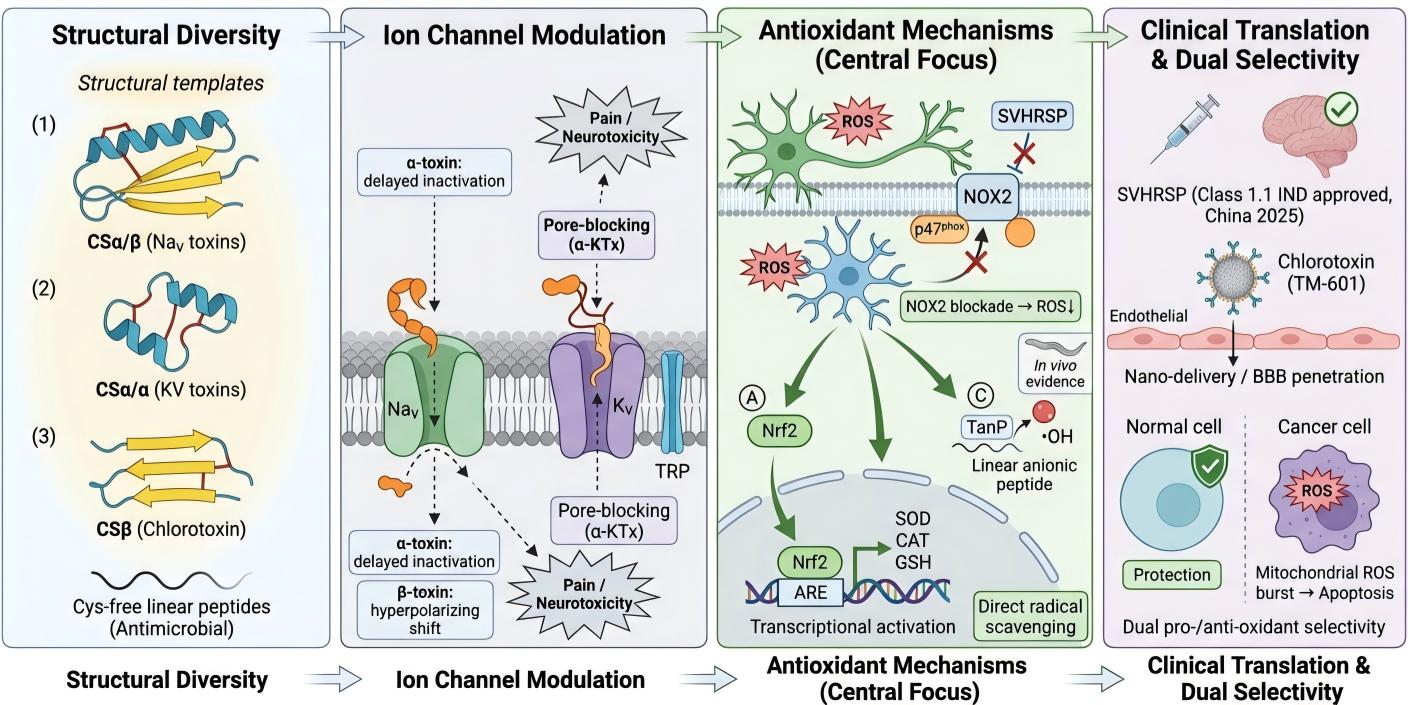
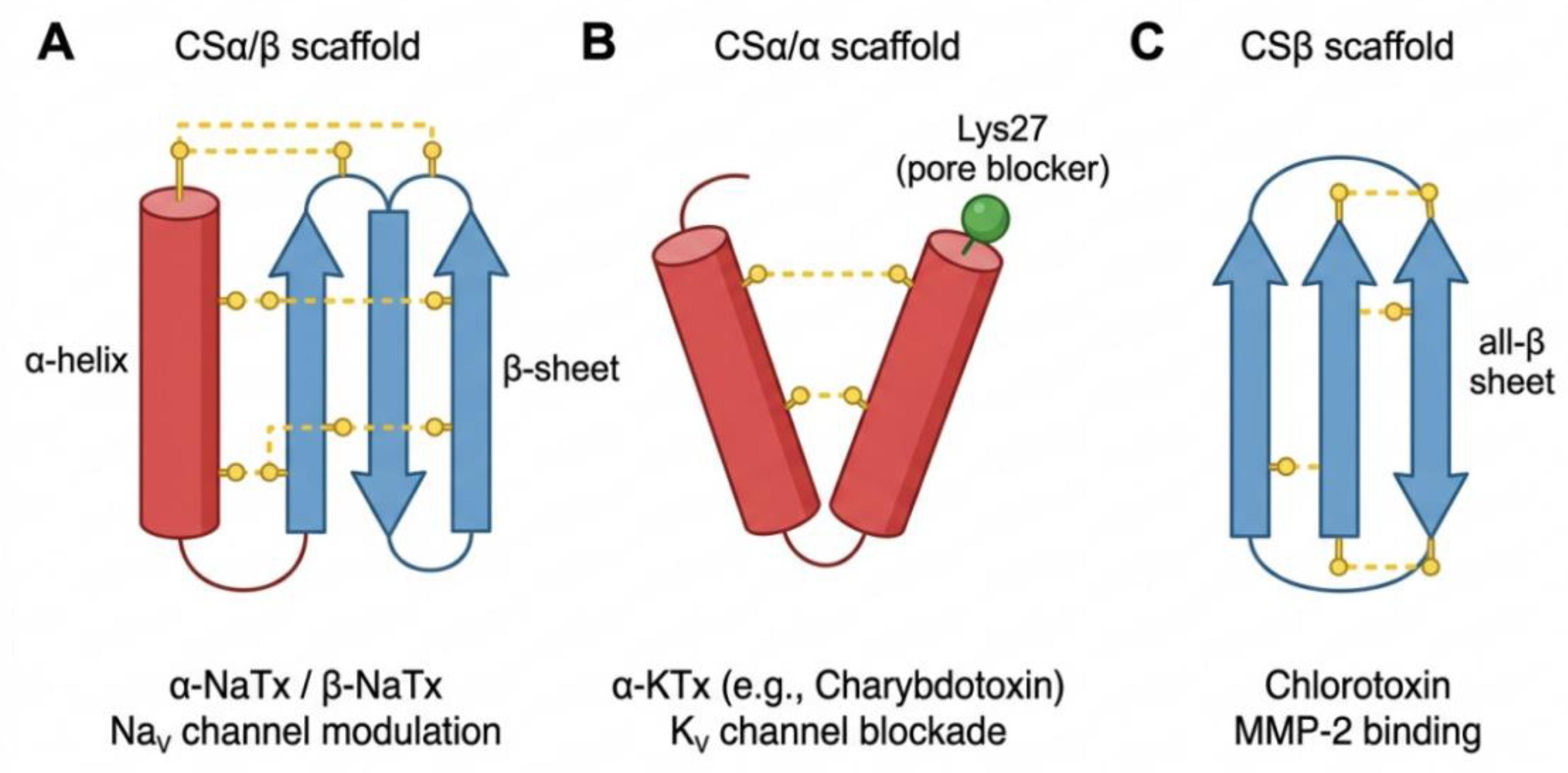
2.1. Cysteine-Based Scaffold Classification System
2.1.1. CSα/β Family: Classic α-Helix-β-Fold Double-Chain Structure
2.1.2. CSα/α Family: Potassium Channel Toxins
2.1.3. CSβ Family of Non-Disulphide Bonded Linear Peptides
2.2. Three-Dimensional Structural Features and Dynamic Properties
2.2.1. Conformational Constraints of Disulphide Bonds
2.2.2. Functional Surfaces and Molecular Recognition
2.2.3. Post-Translational Modifications Expand Chemical Diversity
2.3. Advances in Structural Biology Methodology
2.3.1. X-Ray Crystallography and Nuclear Magnetic Resonance Spectroscopy
2.3.2. The Cryo-Electron Microscopy Structural Revolution
2.3.3. Computational Structural Biology and Artificial Intelligence
2.4. An Integrated Perspective on Structure-Function Relationships
3. Ion Channel Modulation: Mechanisms and Selectivity
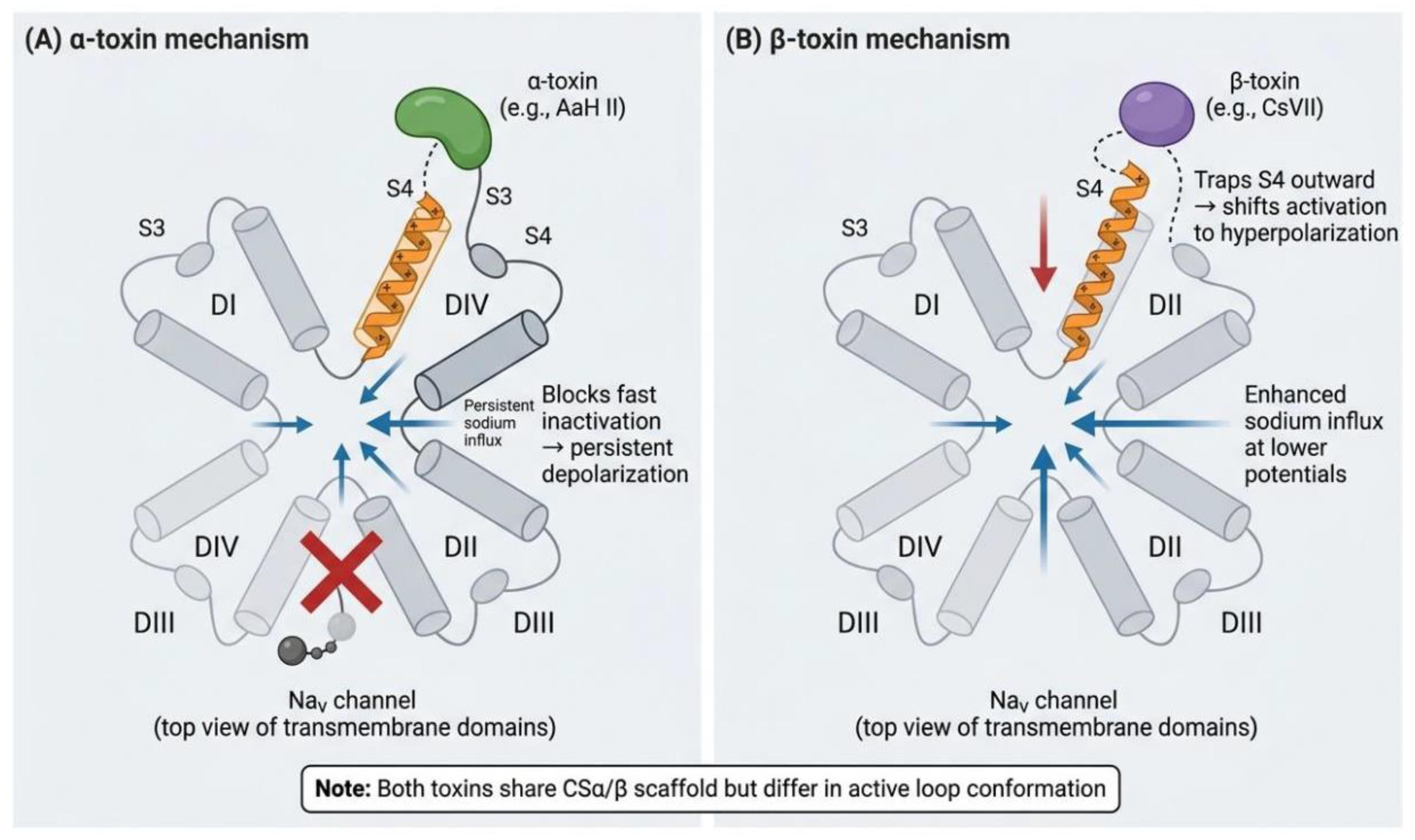
3.1. Voltage-Gated Sodium Channel Regulation Mechanism
3.2. Regulation of Voltage-Gated Potassium Channels
3.3. Other Ion Channel Targets
3.4. Integrated Determinants of Selectivity
4. Beyond Ion Channels: Emerging Targets and Pharmacological Profiles
4.1. Analgesic Mechanisms and Targets
4.2. Anticancer Mechanisms and Targets
4.3. Antimicrobial Activity
4.4. Neuroprotective Effects
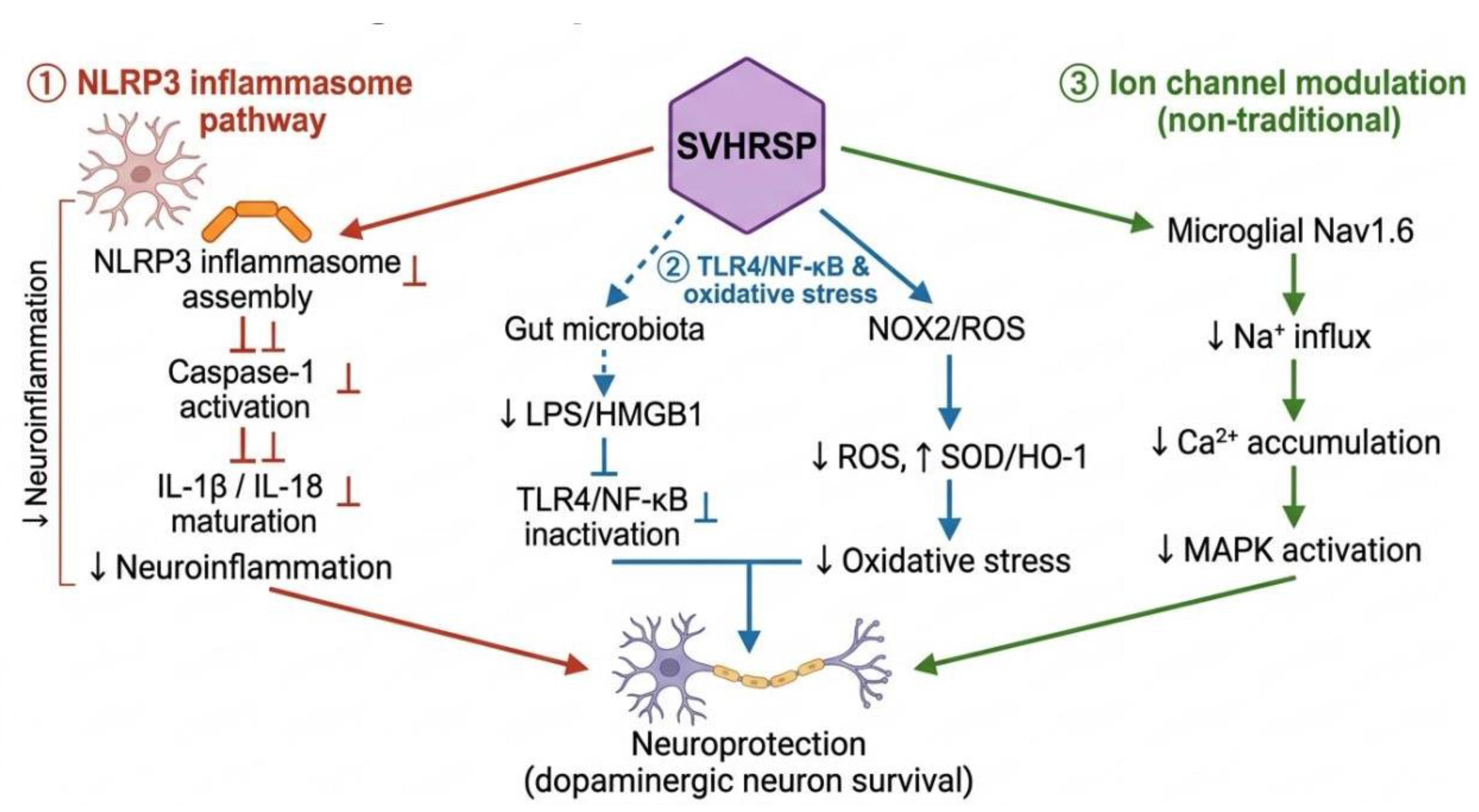
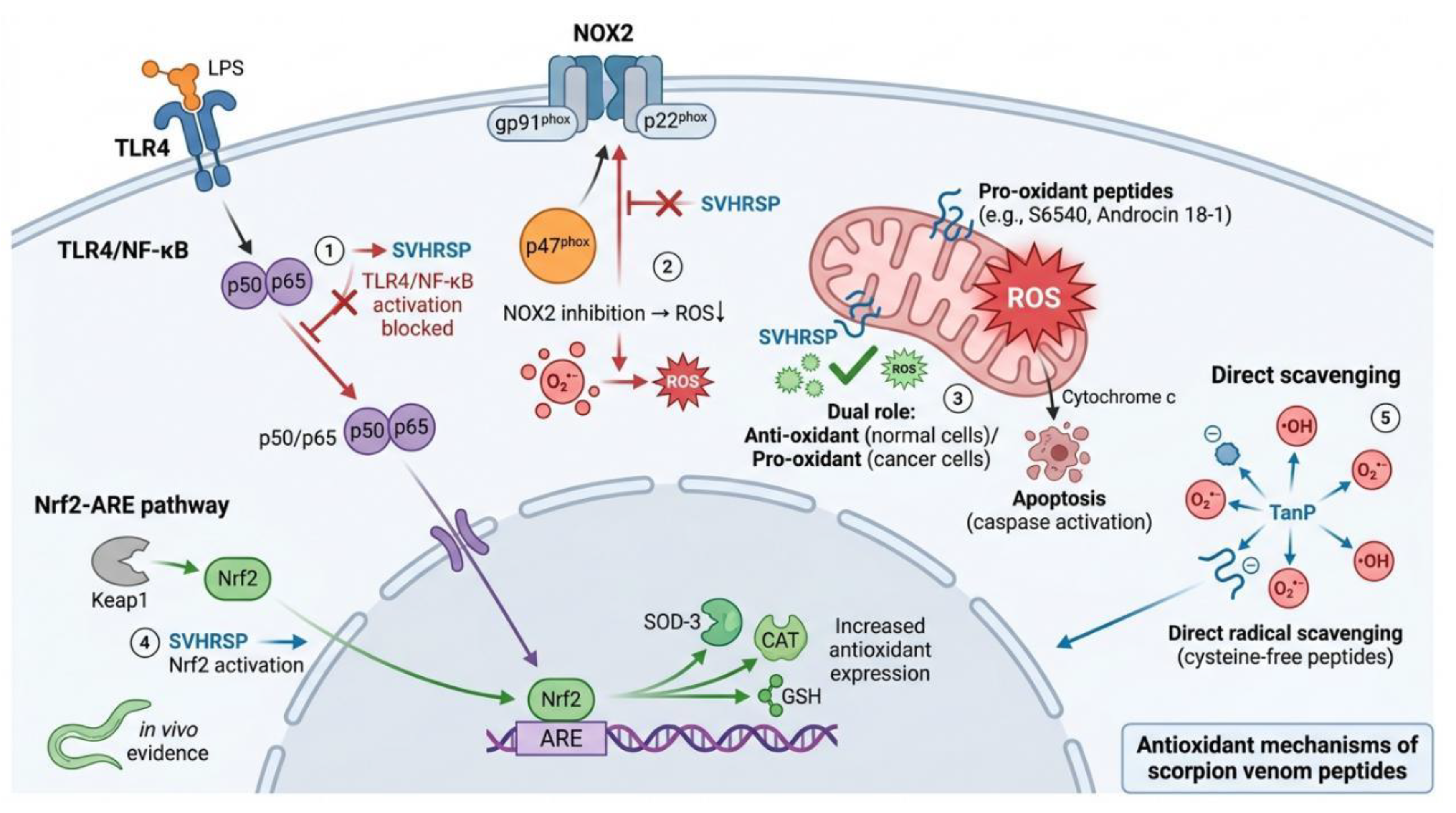
4.5. Antioxidant Mechanism of Scorpion Venom Peptides
| Mechanism Type | Peptide | Target /Pathway |
Key Experimental Evidence | Citations |
|---|---|---|---|---|
| Nrf2-ARE pathway activation | SVHRSP | Nrf-2; p38 MAPK; Lnc Gm6410 |
Upregulates Nrf-2 expression in PM2.5-exposed AD model, alleviating ER stress and neuronal pyroptosis; Regulates Lnc Gm6410 to mitigate necroptosis; Reduces ROS, increases SOD-3 activity, and upregulates ctl-1, egl-1, cat-2 expression in C. elegans | [77] |
| NOX2 targeted inhibition | SVHRSP | NOX2; p47phox membrane translocation |
Blocks membrane translocation of p47phox, preventing NOX2 assembly and activation; NOX2 siRNA knockdown significantly attenuates anti-inflammatory and neuroprotective effects of SVHRSP | [78] |
| Endogenous antioxidant enzyme regulation | Smp24 | SOD; CAT; GSH; MDA; NO | Increases SOD, CAT and GSH levels while reducing MDA and NO content in solid-Ehrlich carcinoma mouse model | [69] |
| Direct free radical scavenging | TanP | Free radical direct scavenging | Scavenges free radicals independent of intracellular signaling pathways | [67] |
| Pro-oxidant / antioxidant dual selectivity | S6540, Androcin 18-1 | Mitochondrial ROS; PI3K/Akt | Induces excessive mitochondrial ROS generation and triggers apoptosis in tumor cells; tends to inhibit oxidative stress in normal cells | [61,81] |
5. Peptide Engineering and Therapeutic Development
5.1. Molecular Optimization and Delivery Strategies
5.2. Clinical Translation and Patent Landscape
6. Challenges and Future Perspectives
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Data Availability Statement
Conflicts of Interest
References
- Guan, C.G.; Torres, M.D.T.; Li, S.F.; de la Fuente-nunez, C. Computational exploration of global venoms for antimicrobial discovery with Venomics artificial intelligence. Nat Commun 2025, 16. [CrossRef]
- Xia, Z.Q.; He, D.G.; Wu, Y.L.; Kwok, H.F.; Cao, Z.J. Scorpion venom peptides: Molecular diversity, structural characteristics, and therapeutic use from channelopathies to viral infections and cancers. Pharmacol Res 2023, 197. [CrossRef]
- Mendes, L.C.; Viana, G.M.M.; Nencioni, A.L.A.; Pimenta, D.C.; Beraldo-Neto, E. Scorpion Peptides and Ion Channels: An Insightful Review of Mechanisms and Drug Development. Toxins 2023, 15. [CrossRef]
- Xia, Z.Q.; Xie, L.X.; Li, B.; Lv, X.Y.; Zhang, H.Z.; Cao, Z.J. Antimicrobial Potential of Scorpion-Venom-Derived Peptides. Molecules 2024, 29. [CrossRef]
- Pashmforoosh, N.; Baradaran, M. Peptides with Diverse Functions from Scorpion Venom: A Great Opportunity for the Treatment of a Wide Variety of Diseases. Iran Biomed J 2023, 27, 84-99. [CrossRef]
- Kamau, P.M.; Zhong, J.; Yao, B.; Lai, R.; Luo, L. Bioactive peptides from scorpion venoms: therapeutic scaffolds and pharmacological tools. Chin J Nat Medicines 2023, 21, 19-35. [CrossRef]
- Barnham, K.J.; Masters, C.L.; Bush, A.I. Neurodegenerative diseases and oxidative stress. Nature Reviews Drug Discovery 2004, 3, 205-214. [CrossRef]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat Rev Mol Cell Bio 2020, 21, 363-383. [CrossRef]
- Abdallnasser Amen, R.; Atef Essmat, R.; Farid, A.; Abdel-Rahman, M.A.; El-Sherif, A.A.; Zhang, Y. Scorpion venom as a natural peptide source for innovative therapeutic solutions: A comprehensive review of its potential in emerging medical frontiers. Toxicon 2025, 268. [CrossRef]
- Liu, M.; Svirskis, D.; Proft, T.; Loh, J.; Yin, N.; Li, H.; Li, D.; Zhou, Y.; Chen, S.; Song, L.; et al. Progress in peptide and protein therapeutics: Challenges and strategies. Acta Pharm Sin B 2025, 15, 6342-6381. [CrossRef]
- He, D.G.; Lei, Y.N.; Qin, H.X.; Cao, Z.J.; Kwok, H.F. Deciphering Scorpion Toxin-Induced Pain: Molecular Mechanisms and Ion Channel Dynamics. International Journal of Biological Sciences 2025, 21, 2921-2934. [CrossRef]
- Liu, M.; Svirskis, D.; Proft, T.; Loh, J.; Yin, N.; Li, H.; Li, D.; Zhou, Y.; Chen, S.; Song, L.; et al. Progress in peptide and protein therapeutics: Challenges and strategies. Acta Pharmaceutica Sinica B 2025, 15, 6342-6381. [CrossRef]
- Rafferty, J.; Nagaraj, H.; P. McCloskey, A.; Huwaitat, R.; Porter, S.; Albadr, A.; Laverty, G. Peptide Therapeutics and the Pharmaceutical Industry: Barriers Encountered Translating from the Laboratory to Patients. Current Medicinal Chemistry 2016, 23, 4231-4259. [CrossRef]
- Shariati, S.; Mafakher, L.; Shirani, M.; Baradaran, M. Unveiling new Kv1.3 channel blockers from scorpion venom: Characterization of Meuk7–3 and in silico design of its analogs for enhanced affinity and therapeutic potential. International Journal of Biological Macromolecules 2025, 319. [CrossRef]
- Huang, Y.; Kamau, P.M.; Wang, J.M.; Gao, M.Y.; Li, B.W. Scorpion Venom Neurotoxins: Molecular Diversity, Mechanisms, and Drug Scaffolds. Toxins 2026, 18. [CrossRef]
- Aoki-Shioi, N.; Nomura, S.; Tanaka, Y.; Hirose, S. Ion Channel-Targeting Toxins: Structural Mechanisms of Activation, Inhibition, and Therapeutic Potential. Toxins 2025, 17. [CrossRef]
- Vacher, H.; Diochot, S.; Bougis, P.E.; Martin-Eauclaire, M.F.; Mourre, C. Kv4 channels sensitive to BmTX3 in rat nervous system: autoradiographic analysis of their distribution during brain ontogenesis. Eur J Neurosci 2006, 24, 1325-1340. [CrossRef]
- Shakeel, K.; Naseem, M.U.; Olamendi-Portugal, T.; Zamudio, F.Z.; Possani, L.D.; Panyi, G. Cvill6 and Cvill7: Potent and Selective Peptide Blockers of Kv1.2 Ion Channel Isolated from Mexican Scorpion Centruroides villegasi. Toxins (Basel) 2025, 17. [CrossRef]
- Ramsoomair, D.; Ramsoomair, C.K.; Daftari, M.; Himic, V.; Shlobin, N.A.; Wang, S.E.; Ivan, M.E.; Komotar, R.J.; Shah, A.H. Translating Venom to Medicine: A Comprehensive Review on the Role of Chlorotoxin in Glioblastoma Diagnosis and Therapy. Molecular Cancer Therapeutics 2025, 24, 1867-1877. [CrossRef]
- Megaly, A.M.A.; Nakamichi, R.; Wakayu, M.; Nakagawa, Y.; Abdel-Wahab, M.; Miyashita, M. Identification of a novel insecticidal chlorotoxin-like peptide from the venom of the Compsobuthus egyptiensis scorpion. Toxicon 2025, 267. [CrossRef]
- Giugliano, R.; Della Marca, R.; Chianese, A.; Monti, A.; Donadio, F.; Esposito, E.; Doti, N.; Zannella, C.; Galdiero, M.; De Filippis, A. The inhibitory potential of three scorpion venom peptides against multidrug-resistant Klebsiella pnemoniae. Frontiers in Microbiology 2025, 16. [CrossRef]
- Zhu, S.; Gao, B.; Tytgat, J. Phylogenetic distribution, functional epitopes and evolution of the CSαβ superfamily. Cellular and Molecular Life Sciences 2005, 62, 2257-2269. [CrossRef]
- Saucedo, A.L.; Flores-Solis, D.; Rodriguez de la Vega, R.C.; Ramirez-Cordero, B.; Hernandez-Lopez, R.; Cano-Sanchez, P.; Noriega Navarro, R.; Garcia-Valdes, J.; Coronas-Valderrama, F.; de Roodt, A.; et al. New tricks of an old pattern: structural versatility of scorpion toxins with common cysteine spacing. J Biol Chem 2012, 287, 12321-12330. [CrossRef]
- Qin, C.; Yang, X.; Zuo, Z.; Yang, L.; Yang, F.; Cao, Z.; Chen, Z.; Wu, Y. BmK86-P1, a New Degradation Peptide with Desirable Thermostability and Kv1.2 Channel-Specific Activity from Traditional Chinese Scorpion Medicinal Material. Toxins 2021, 13. [CrossRef]
- Catterall, W.A.; Cestèle, S.; Yarov-Yarovoy, V.; Yu, F.H.; Konoki, K.; Scheuer, T. Voltage-gated ion channels and gating modifier toxins. Toxicon 2007, 49, 124-141. [CrossRef]
- Martin-Eauclaire, M.F.; Bougis, P.E.; de Lima, M.E. Ts1 from the Brazilian scorpion: A half-century of studies on a multifunctional beta like-toxin. Toxicon 2018, 152, 106-120. [CrossRef]
- Delgado-Prudencio, G.; Possani, L.D.; Becerril, B.; Ortiz, E. The Dual α-Amidation System in Scorpion Venom Glands. Toxins 2019, 11. [CrossRef]
- Adegoke, V.A.; Dongol, Y.; Gonzalez, T.; Song, A.; Clark, R.J.; Lewis, R.J.; Conibear, A.C.; Rosengren, K.J.; Aguilar, M. Proline hydroxylation and C-terminal amidation in µ-conotoxins increase structural stability and potency at sodium channels. Australian Journal of Chemistry 2025, 78. [CrossRef]
- Möller, D.S.; van der Walt, M.; Oosthuizen, C.; Serian, M.; Serem, J.C.; Lorenz, C.D.; Mason, A.J.; Bester, M.J.; Gaspar, A.R.M. Improving the Activity and Selectivity of a Scorpion-Derived Peptide, A3a, against through Rational Design. Acs Omega 2025, 10, 4699-4710. [CrossRef]
- Du, C.; Li, H.; Liu, C.; Fan, H. Understanding of the postgerminative development response to salinity and drought stresses in cucumber seeds by integrated proteomics and transcriptomics analysis. Journal of Proteomics 2021, 232. [CrossRef]
- Zeng, L.; Sun, Y.; Zhang, H.; Yi, X.; Du, R.; Chen, Z.; Wang, Q. Scorpion venom peptides enhance immunity and survival in Litopenaeus vannamei through antibacterial action against Vibrio parahaemolyticus. Frontiers in Immunology 2025, 16. [CrossRef]
- Xia, Z.; He, D.; Wu, Y.; Kwok, H.F.; Cao, Z. Scorpion venom peptides: Molecular diversity, structural characteristics, and therapeutic use from channelopathies to viral infections and cancers. Pharmacol Res 2023, 197. [CrossRef]
- Zhang, N.; Li, M.; Chen, X.; Wang, Y.; Wu, G.; Hu, G.; Wu, H. Solution structure of BmKK2, a new potassium channel blocker from the venom of chinese scorpion Buthus martensi Karsch. Proteins: Structure, Function, and Bioinformatics 2004, 55, 835-845. [CrossRef]
- Clairfeuille, T.; Cloake, A.; Infield, D.T.; Llongueras, J.P.; Arthur, C.P.; Li, Z.R.; Jian, Y.; Martin-Eauclaire, M.-F.; Bougis, P.E.; Ciferri, C.; et al. Structural basis of α-scorpion toxin action on Nav channels. Science 2019, 363. [CrossRef]
- Chen, R.; Chung, S.-H. Computational Studies of Venom Peptides Targeting Potassium Channels. Toxins 2015, 7, 5194-5211. [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Zídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583-+. [CrossRef]
- Abramson, J.; Adler, J.; Dunger, J.; Evans, R.; Green, T.; Pritzel, A.; Ronneberger, O.; Willmore, L.; Ballard, A.J.; Bambrick, J.; et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature 2024, 630, 493-500. [CrossRef]
- Miyashita, M.; Mitani, N.; Iwamoto, F.; Hirota, M.; Nakagawa, Y. Discovery of a Novel Insecticidal Peptide with a Cystine-Stabilized α-Helix/α-Helix Motif from the Venom of Scorpion Liocheles australasiae. Molecules 2024, 30. [CrossRef]
- Montero-Dominguez, P.A.; Restano-Cassulini, R.; Magaña-Ávila, L.C.; Almanza, A.; Mercado, F.; Corzo, G. Design of antinociceptive peptide by grafting domains between scorpion β-neurotoxins. Bioorganic Chemistry 2025, 162. [CrossRef]
- Wai, D.C.C.; Naseem, M.U.; Mocsár, G.; Reddiar, S.B.; Pan, Y.J.; Csoti, A.; Hajdu, P.; Nowell, C.; Nicolazzo, J.A.; Panyi, G.; et al. Fluorescent Peptide Toxin for Selective Visualization of the Voltage-Gated Potassium Channel K1.3. Bioconjugate Chem 2022. [CrossRef]
- Attali, B.; Ma, Z.; Kong, J.; Gordon, D.; Gurevitz, M.; Kallen, R.G. Direct Evidence that Scorpion α-Toxins (Site-3) Modulate Sodium Channel Inactivation by Hindrance of Voltage-Sensor Movements. PLoS ONE 2013, 8. [CrossRef]
- Catterall, W.A.; Swanson, T.M. Structural Basis for Pharmacology of Voltage-Gated Sodium and Calcium Channels. Mol Pharmacol 2015, 88, 141-150. [CrossRef]
- Gurevitz, M., Zhorov, B. S. and Dong, K. Allosteric interactions among voltage-sensor modules of sodium channels probed by scorpion toxin modifiers. Journal of Neurobiology and Physiology 2022, 4. [CrossRef]
- Bennett, D.L.H.; Woods, C.G. Painful and painless channelopathies. The Lancet Neurology 2014, 13, 587-599. [CrossRef]
- Xin, K.; Sun, R.; Xiao, W.; Lu, W.; Sun, C.; Lou, J.; Xu, Y.; Chen, T.; Wu, D.; Gao, Y. Short Peptides from Asian Scorpions: Bioactive Molecules with Promising Therapeutic Potential. Toxins 2025, 17. [CrossRef]
- Liu, Y.Y.; Liu, Y.Y.; Liu, Y.X.; Cui, Y.; Meng, T.; Song, Y.B.; Zhao, F. From traditional medicine to targeted therapy: Structure-activity relationship-guided optimization of scorpion toxin DKK2 for pain-associated sodium channel blockade. J Ethnopharmacol 2025, 352. [CrossRef]
- Bennett, D.L.H.; Woods, C.G. Painful and painless channelopathies. Lancet Neurol 2014, 13, 587-599. [CrossRef]
- Gigolaev, A.M.; Pinheiro, E.; Peigneur, S.; Tytgat, J.; Vassilevski, A.A. KV1.2-Selective Peptide with High Affinity. J Evol Biochem Phys+ 2022, 58, 2048-2057. [CrossRef]
- Gigolaev, A.M.; Kuzmenkov, A.I.; Peigneur, S.; Tabakmakher, V.M.; Pinheiro-Junior, E.L.; Chugunov, A.O.; Efremov, R.G.; Tytgat, J.; Vassilevski, A.A. Tuning Scorpion Toxin Selectivity: Switching From K1.1 to K1.3. Front Pharmacol 2020, 11. [CrossRef]
- Shakeel, K.; Olamendi-Portugal, T.; Naseem, M.U.; Becerril, B.; Zamudio, F.Z.; Delgado-Prudencio, G.; Possani, L.D.; Panyi, G. Of Seven New K+ Channel Inhibitor Peptides of Centruroides bonito, α-KTx 2.24 Has a Picomolar Affinity for Kv1.2. Toxins 2023, 15. [CrossRef]
- Short, B. A scorpion toxin takes the sting out of T cell activation. J Gen Physiol 2022, 154. [CrossRef]
- Meredith, A.L. BK Channelopathies and -Linked Disease Models. Annual Review of Physiology 2024, 86, 277-300. [CrossRef]
- Chen, Y.H.; Markov, N.; Gigon, L.; Hosseini, A.; Yousefi, S.; Stojkov, D.; Simon, H.U. The BK Channel Limits the Pro-Inflammatory Activity of Macrophages. Cells-Basel 2024, 13. [CrossRef]
- Ancatén-González, C.; Segura, I.; Alvarado-Sánchez, R.; Chávez, A.E.; Latorre, R. Ca2+- and Voltage-Activated K+ (BK) Channels in the Nervous System: One Gene, a Myriad of Physiological Functions. International Journal of Molecular Sciences 2023, 24. [CrossRef]
- Hakim, M.; Jiang, W.; Luo, L.; Li, B.; Yang, S.; Song, Y.; Lai, R. Scorpion Toxin, BmP01, Induces Pain by Targeting TRPV1 Channel. Toxins 2015, 7, 3671-3687. [CrossRef]
- Heusser, S.A.; Borg, C.B.; Colding, J.M.; Pless, S.A. Conformational decoupling in acid-sensing ion channels uncovers mechanism and stoichiometry of PcTx1-mediated inhibition. Elife 2022, 11. [CrossRef]
- Jiang, D.; Tonggu, L.; Gamal El-Din, T.M.; Banh, R.; Pomès, R.; Zheng, N.; Catterall, W.A. Structural basis for voltage-sensor trapping of the cardiac sodium channel by a deathstalker scorpion toxin. Nat Commun 2021, 12. [CrossRef]
- Sumino, A.; Sumikama, T.; Uchihashi, T.; Oiki, S. High-speed AFM reveals accelerated binding of agitoxin-2 to a K+ channel by induced fit. Science Advances 2019, 5. [CrossRef]
- Lee, S.-Y.; MacKinnon, R. A membrane-access mechanism of ion channel inhibition by voltage sensor toxins from spider venom. Nature 2004, 430, 232-235. [CrossRef]
- Zhu, Q.; Du, Y.; Nomura, Y.; Gao, R.; Cang, Z.; Wei, G.-W.; Gordon, D.; Gurevitz, M.; Groome, J.; Dong, K. Charge substitutions at the voltage-sensing module of domain III enhance actions of site-3 and site-4 toxins on an insect sodium channel. Insect Biochem Molec 2021, 137. [CrossRef]
- Zhang, X.M.; Tu, D.Z.; Li, S.; Li, N.; Li, D.L.; Gao, Y.; Tian, L.; Liu, J.N.; Zhang, X.; Hong, J.S.; et al. A novel synthetic peptide SVHRSP attenuates dopaminergic neurodegeneration by inhibiting NADPH oxidase-mediated neuroinflammation in experimental models of Parkinson’s disease. Free Radical Biology and Medicine 2022, 188, 363-374. [CrossRef]
- Xiao, Q.; Zhang, Z.P.; Hou, Y.B.; Qu, D.X.; Tang, L.L.; Chen, L.J.; Li, G.Y.; Ji, Y.H.; Tao, J.; Zhu, Y.D. Anti-epileptic/pro-epileptic effects of sodium channel modulators from Buthus martensii Karsch. Sheng Li Xue Bao 2022, 74, 621-632.
- Wang, X.; Luo, H.; Peng, X.; Chen, J. Spider and scorpion knottins targeting voltage-gated sodium ion channels in pain signaling. Biochemical Pharmacology 2024, 227. [CrossRef]
- Aftabizadeh, M.; Bawa, L.; Wang, S.; Brewster, B.; Wang, D.; Wang, X.; Brown, C.; Barish, M. Exth-10. Exploration of a Novel Toxin-Incorporating Car T Cell: How Does Chlorotoxin Recognize Glioblastoma Cells? Neuro-Oncology 2021, 23, vi165-vi165. [CrossRef]
- Nguyen, T.; Guo, R.Y.; Chai, J.W.; Wu, J.N.; Liu, J.F.; Chen, X.; Abdel-Rahman, M.A.; Xia, H.; Xu, X.Q. Smp24, a Scorpion-Venom Peptide, Exhibits Potent Antitumor Effects against Hepatoma HepG2 Cells via Multi-Mechanisms In Vivo and In Vitro. Toxins 2022, 14. [CrossRef]
- Xu, L.P.; Zhang, Y.; Dai, Q.D.; Lin, N.; Guan, T.R.; Song, X.W.; Hong, S.H. Scorpion venom polypeptide governs alveolar macrophage M1/M2 polarization to alleviate pulmonary fibrosis. Tissue Cell 2022, 79. [CrossRef]
- Amen, R.A.; Essmat, R.A.; Farid, A.; Abdel-Rahman, M.A.; El-Sherif, A.A.; Zhang, Y.H. Scorpion venom as a natural peptide source for innovative therapeutic solutions: A comprehensive review of its potential in emerging medical frontiers. Toxicon 2025, 268. [CrossRef]
- Rincón-Cortés, C.A.; Bayona-Rojas, M.A.; Reyes-Montaño, E.A.; Vega-Castro, N.A. Antimicrobial Activity Developed by Scorpion Venoms and Its Peptide Component. Toxins 2022, 14. [CrossRef]
- Nasr, S.; Borges, A.; Sahyoun, C.; Nasr, R.; Roufayel, R.; Legros, C.; Sabatier, J.M.; Fajloun, Z. Scorpion Venom as a Source of Antimicrobial Peptides: Overview of Biomolecule Separation, Analysis and Characterization Methods. Antibiotics-Basel 2023, 12. [CrossRef]
- Zhang, Y.; Li, S.; Hou, L.; Wu, M.; Liu, J.; Wang, R.; Wang, Q.; Zhao, J. NLRP3 mediates the neuroprotective effects of SVHRSP derived from scorpion venom in rotenone-induced experimental Parkinson’s disease model. J Ethnopharmacol 2023, 312. [CrossRef]
- Balde, A.; Benjakul, S.; Nazeer, R.A. A review on NLRP3 inflammasome modulation by animal venom proteins/peptides: mechanisms and therapeutic insights. Inflammopharmacology 2025, 33, 1013-1031. [CrossRef]
- Chen, M.D.; Zhang, Y.; Hou, L.Y.; Zhao, Z.R.; Tang, P.Y.; Sun, Q.Q.; Zhao, J.; Wang, Q.S. SVHRSP protects against rotenone-induced neurodegeneration in mice by inhibiting TLR4/NF-κB-mediated neuroinflammation via gut microbiota. Npj Parkinsons Dis 2025, 11. [CrossRef]
- Li, S.; Guo, Z.Y.; Liu, J.N.; Ma, Y.; Zhang, X.M.; Hou, L.Y.; Wang, Q.H.; Jiang, W.W.; Wang, Q.S. CD11b-NOX2 mutual regulation-mediated microglial exosome release contributes to rotenone-induced inflammation and neurotoxicity in BV2 microglia and primary cultures. Free Radical Biology and Medicine 2024, 224, 436-446. [CrossRef]
- Wang, Q.H.; Liu, J.N.; Zhang, Y.; Li, Z.; Zhao, Z.R.; Jiang, W.W.; Zhao, J.; Hou, L.Y.; Wang, Q.S. Microglial CR3 promotes neuron ferroptosis via NOX2-mediated iron deposition in rotenone-induced experimental models of Parkinson’s disease. Redox Biol 2024, 77. [CrossRef]
- Li, X.; Wu, X.; Li, N.; Li, D.; Sui, A.; Khan, K.; Ge, B.; Li, S.; Li, S.; Zhao, J. Scorpion venom heat-resistant synthesized peptide ameliorates 6-OHDA-induced neurotoxicity and neuroinflammation: likely role of Na(v) 1.6 inhibition in microglia. Br J Pharmacol 2021, 178, 3553-3569. [CrossRef]
- Qin, C.; Li, D.; Zhang, J.; Yin, Z.; Li, F. Scorpion Venom Heat-Resistant Synthetic Peptide Alleviates Neuronal Necroptosis in Alzheimer’s Disease Model by Regulating Lnc Gm6410 Under PM2.5 Exposure. International Journal of Molecular Sciences 2025, 26. [CrossRef]
- Wang, Y.-Z.; Guo, S.-Y.; Kong, R.-L.; Sui, A.-R.; Wang, Z.-H.; Guan, R.-X.; Supratik, K.; Zhao, J.; Li, S. Scorpion Venom Heat–Resistant Synthesized Peptide Increases Stress Resistance and Extends the Lifespan of Caenorhabditis elegans via the Insulin/IGF-1-Like Signal Pathway. Front Pharmacol 2022, 13. [CrossRef]
- Zhang, X.; Tu, D.; Li, S.; Li, N.; Li, D.; Gao, Y.; Tian, L.; Liu, J.; Zhang, X.; Hong, J.-S.; et al. A novel synthetic peptide SVHRSP attenuates dopaminergic neurodegeneration by inhibiting NADPH oxidase-mediated neuroinflammation in experimental models of Parkinson’s disease. Free Radical Biology and Medicine 2022, 188, 363-374. [CrossRef]
- Fawzy, B.S.; Nafie, M.S.; Ali, I.A.I.; El-Baz, L.M.F.; Xu, X.; Abdel-Rahman, M.A. Scorpion Venom Peptide Smp24 Revealed Apoptotic and Antiangiogenic Activities in Solid-Ehrlich Carcinoma Bearing Mice. International Journal of Peptide Research and Therapeutics 2023, 29. [CrossRef]
- Dahiya, R.; Goyal, K.; Sharma, K.; Rawat, A.; Sharma, V.; Mathur, P. Scorpion venom as a molecular treasure: emerging bioactive compounds and translational therapeutic insights. Archives of Toxicology 2025, 100, 437-450. [CrossRef]
- Duan, K.; Qin, C.; Zhang, Y.; Li, D.; Zhang, J.; Li, F. Scorpion venom heat-resistant synthetic peptide regulates Nrf-2 to alleviate neuronal pyroptosis in an AD model induced by endoplasmic reticulum stress under PM2.5 exposure. Toxicon 2025, 265. [CrossRef]
- Zheng, Q.; Na, R.; Yang, L.; Yu, H.; Zhao, X.; Huang, X. The binding process of BmKTX and BmKTX-D33H toward to Kv1.3 channel: a molecular dynamics simulation study. Journal of Biomolecular Structure and Dynamics 2020, 39, 2788-2797. [CrossRef]
- Edwards, W.; Fung-Leung, W.P.; Huang, C.C.; Chi, E.; Wu, N.; Liu, Y.; Maher, M.P.; Bonesteel, R.; Connor, J.; Fellows, R.; et al. Targeting the Ion Channel Kv1.3 with Scorpion Venom Peptides Engineered for Potency, Selectivity, and Half-life. Journal of Biological Chemistry 2014, 289, 22704-22714. [CrossRef]
- Rashid, M.H.; Huq, R.; Tanner, M.R.; Chhabra, S.; Khoo, K.K.; Estrada, R.; Dhawan, V.; Chauhan, S.; Pennington, M.W.; Beeton, C.; et al. A potent and Kv1.3-selective analogue of the scorpion toxin HsTX1 as a potential therapeutic for autoimmune diseases. Sci Rep-Uk 2014, 4. [CrossRef]
- Boltman, T.; Meyer, M.; Ekpo, O. Diagnostic and Therapeutic Approaches for Glioblastoma and Neuroblastoma Cancers Using Chlorotoxin Nanoparticles. Cancers 2023, 15. [CrossRef]
- Durham, P.G.; Butnariu, A.; Alghorazi, R.; Pinton, G.; Krishna, V.; Dayton, P.A. Current clinical investigations of focused ultrasound blood-brain barrier disruption: A review. Neurotherapeutics 2024, 21. [CrossRef]
- Zhang, Z.; Li, J.; Wang, Y.; Tang, C.; Zhou, Y.; Li, J.; Lu, X.; Wang, Y.; Ma, T.; Xu, H.; et al. Angiopep-2 conjugated biomimetic nano-delivery system loaded with resveratrol for the treatment of methamphetamine addiction. International Journal of Pharmaceutics 2024, 663. [CrossRef]
- Wang, T.; Wang, S.W.; Zhang, Y.; Wu, X.F.; Peng, Y.; Cao, Z.; Ge, B.Y.; Wang, X.; Wu, Q.; Lin, J.T.; et al. Scorpion Venom Heat-Resistant Peptide (SVHRP) Enhances Neurogenesis and Neurite Outgrowth of Immature Neurons in Adult Mice by Up-Regulating Brain-Derived Neurotrophic Factor (BDNF). Plos One 2014, 9. [CrossRef]
- Batrakova, E.V.; Kim, M.S. Using exosomes, naturally-equipped nanocarriers, for drug delivery. J Control Release 2015, 219, 396-405. [CrossRef]
- Mamelak, A.N.; Rosenfeld, S.; Bucholz, R.; Raubitschek, A.; Nabors, L.B.; Fiveash, J.B.; Shen, S.; Khazaeli, M.B.; Colcher, D.; Liu, A.; et al. Phase I single-dose study of intracavitary-administered iodine-131-TM-601 in adults with recurrent high-grade glioma. J Clin Oncol 2006, 24, 3644-3650. [CrossRef]
- Neff, R.A.; Wickenden, A.D. Selective Targeting of Nav1.7 with Engineered Spider Venom-Based Peptides. Channels 2021, 15, 179-193. [CrossRef]
- Abramson, J.; Adler, J.; Dunger, J.; Evans, R.; Green, T.; Pritzel, A.; Ronneberger, O.; Willmore, L.; Ballard, A.J.; Bambrick, J.; et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3 (vol 630, pg 493, 2024). Nature 2024, 636, E4-E4. [CrossRef]
- Dauparas, J.; Anishchenko, I.; Bennett, N.; Bai, H.; Ragotte, R.J.; Milles, L.F.; Wicky, B.I.M.; Courbet, A.; de Haas, R.J.; Bethel, N.; et al. Robust deep learning-based protein sequence design using ProteinMPNN. Science 2022, 378, 49-55. [CrossRef]
- Ren, C.-Z.; Wu, Z.-T.; Wang, W.; Tan, X.; Yang, Y.-H.; Wang, Y.-K.; Li, M.-L.; Wang, W.-Z. SIRT1 exerts anti-hypertensive effect via FOXO1 activation in the rostral ventrolateral medulla. Free Radical Biology and Medicine 2022, 188, 1-13. [CrossRef]
- Radhakrishna, K.; Rajshekhar, P.K.; Arshitha, R.; Patil, K.; Dhannura, S.; Gowd, K.H. Dissecting oxidative folding of conotoxins using 3D structures of cysteine mutants predicted by AlphaFold 3: A case study of α-conotoxin RgIA, χ-conotoxin CMrVIA and ω-conotoxin MVIIA-Gly. Toxicon 2025, 262. [CrossRef]
- Callaway, E. ‘It will change everything’: DeepMind’s AI makes gigantic leap in solving protein structures. Nature 2020, 588, 203-204. [CrossRef]
- Jumper, J.; Evans, R.; Pritzel, A.; Green, T.; Figurnov, M.; Ronneberger, O.; Tunyasuvunakool, K.; Bates, R.; Žídek, A.; Potapenko, A.; et al. Highly accurate protein structure prediction with AlphaFold. Nature 2021, 596, 583-589. [CrossRef]
- Wang, F.; Wang, Y.; Feng, L.; Zhang, C.; Lai, L. Target-Specific De Novo Peptide Binder Design with DiffPepBuilder. Journal of Chemical Information and Modeling 2024, 64, 9135-9149. [CrossRef]
- Borrego, J.; Naseem, M.U.; Sehgal, A.N.A.; Panda, L.R.; Shakeel, K.; Gaspar, A.; Nagy, C.; Varga, Z.; Panyi, G. Recombinant Expression in Pichia pastoris System of Three Potent Kv1.3 Channel Blockers: Vm24, Anuroctoxin, and Ts6. Journal of Fungi 2022, 8. [CrossRef]
- Thornton, E.L.; Boyle, J.T.; Laohakunakorn, N.; Regan, L. Cell-Free Protein Synthesis as a Method to Rapidly Screen Machine Learning-Generated Protease Variants. Acs Synth Biol 2025, 14, 1710-1718. [CrossRef]
- Wang, Z.; Sang, M.; Zhang, Y.; Chen, S.; Li, S.; Chen, Y.; Xu, E.; Zhou, Q.; Xu, W.; Zhao, C.; et al. BmKK2, a thermostable Kv1.3 blocker from Buthus martensii Karsch (BmK) scorpion, inhibits the activation of macrophages via Kv1.3-NF-κB- NLRP3 axis. J Ethnopharmacol 2023, 314. [CrossRef]
| Structural family | disulphide pattern | 3D folding feature | Representative member | Target/mechanism | Citations |
|---|---|---|---|---|---|
| CSα/β | 8 Cys, 4 disulphide bonds |
α-helix + three-stranded antiparallel β-sheet | AaH II (α-NaTx), CsVII (β-NaTx) | NaV channels: α-toxin slows inactivation; β-toxin shifts activation voltage | [15] |
| CSα/α | C-C-CC-C-C | Two α-helices (“two-finger” or “three-finger”) | Charybdotoxin (α-KTx 1.1), HsTx1 (κ-KTx) | KV channels: physical pore blocking or voltage sensor trapping | [14,40] |
| CSβ | C-C-C-C | Three-stranded antiparallel β-sheet (no α-helix) | Chlorotoxin | MMP-2/Annexin A2, glioma targeting | [19] |
| Cysteine-free linear peptide | No disulphide bond | Amphipathic α-helix | Pandinin-1 | Membrane disruption (“carpet” or “barrel-stave” model) | [21] |
| Molecule | Structural family | Primary target | Pharmacological activity | Clinical/translation status | Citations |
|---|---|---|---|---|---|
| OD1 | CSα/β (α-like) | NaV1.7 | Analgesia | Preclinical (engineering for subtype selectivity) | [39] |
| DKK2-N18W | CSα/β | NaV1.7 | Analgesia | Preclinical (effective in formalin pain model) | [45,46] |
| Charybdotoxin (ChTx) | CSα/α (α-KTx 1.1) | KV1.3 | Immunosuppression | Tool molecule; engineered for autoimmune diseases | [48] |
| Cvill7 | CSα/α (α-KTx 2) | KV1.2 | Anti-epileptic potential | Lead candidate (450-fold selective over KV1.3) | [18] |
| BmKK2 | CSα/α | KV1.3 | Anti-inflammatory (NF-κB-NLRP3 inhibition) | Preclinical (from traditional Chinese medicine) | [70] |
| Chlorotoxin (TM-601) | CSβ | MMP-2 | Glioma-targeted diagnosis and therapy | Phase I/II trials (intracavitary); shifted to imaging/delivery | [19] |
| SVHRSP | Synthetic derivative (heat-resistant peptide) | Multi-target (NLRP3, TLR4, NOX2, Nav1.6) | Neuroprotection (Parkinson’s, Alzheimer’s, epilepsy) | NMPA Class 1.1 IND approved (2025) | [70,72,75] |
| Pantinin-1/2 | Cysteine-free linear peptide | Bacterial outer membrane LPS | Anti-MDR Klebsiella pneumoniae | Preclinical (MIC 6-25 μM) | [21] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




