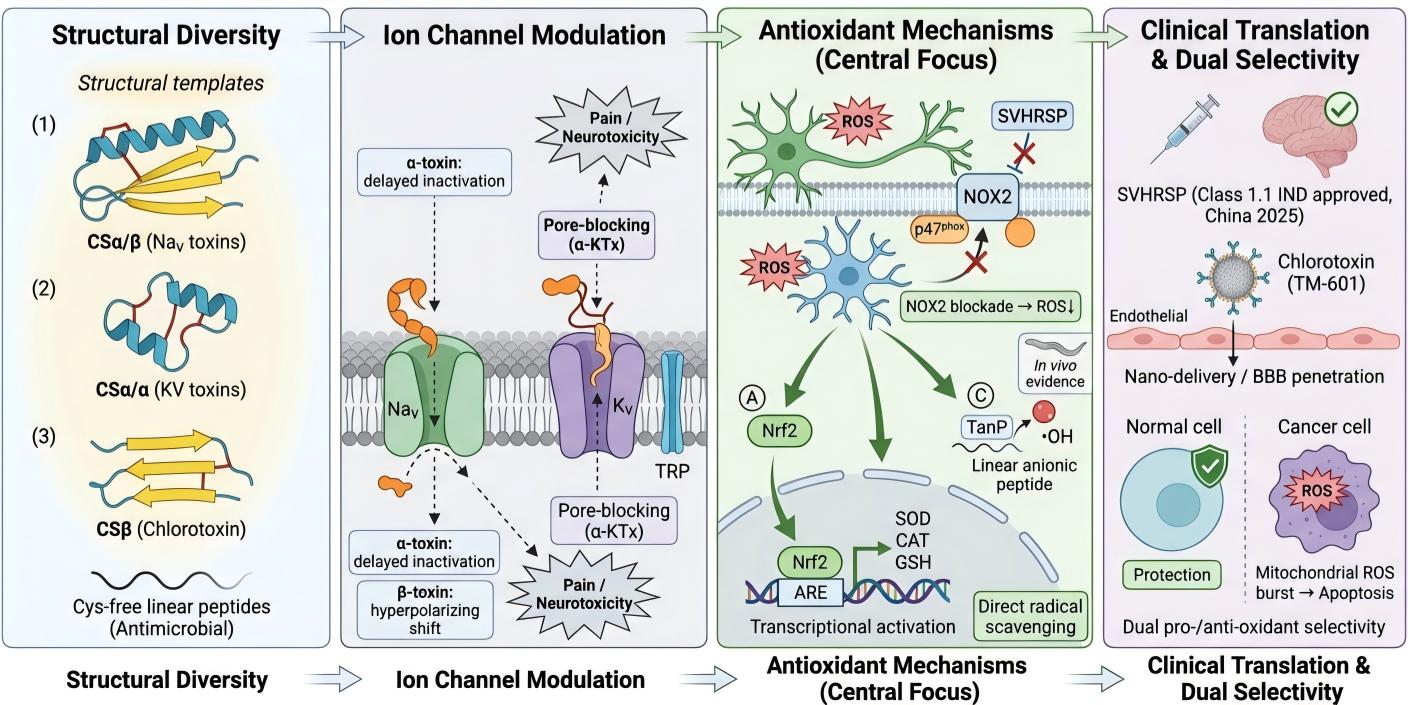
Scorpion venom peptides, with their stable disulphide backbone, compact structural framework, and highly selective regulation of ion channels, have long been regarded as important molecular probes in neuropharmacology. However, recent studies have revealed their potential for regulating oxidative stress, anti-inflammation, and neuroprotection, making them a new research frontier. This article focuses on “scorpion venom peptides as drugs,” constructing an integrated knowledge framework from structural classification to clinical translation. First, scorpion venom peptides are systematically classified based on cysteine arrangement patterns and three-dimensional folding topology, and their structure-activity relationships are summarised. Based on this, the molecular mechanisms by which scorpion venom peptides regulate ion channels are systematically analysed. In addition, we review the emerging pharmacological activities of scorpion venom peptides. Of particular note, the representative molecule SVHRSP has shown multi-target synergistic antioxidant and neuroprotective activity in models of Parkinson’s disease. This article also systematically evaluates the application of engineering strategies, including cyclisation modification, nanodelivery, recombinant expression, and AI-assisted optimisation, to overcome the translational bottlenecks in the development of scorpion venom peptides. By integrating molecular structure, redox regulation mechanisms, and translational medicine perspectives, this review aims to provide a theoretical basis and practical pathways for scorpion venom peptides as precision therapeutic molecules for oxidative stress-related diseases.