Submitted:
29 April 2026
Posted:
30 April 2026
You are already at the latest version
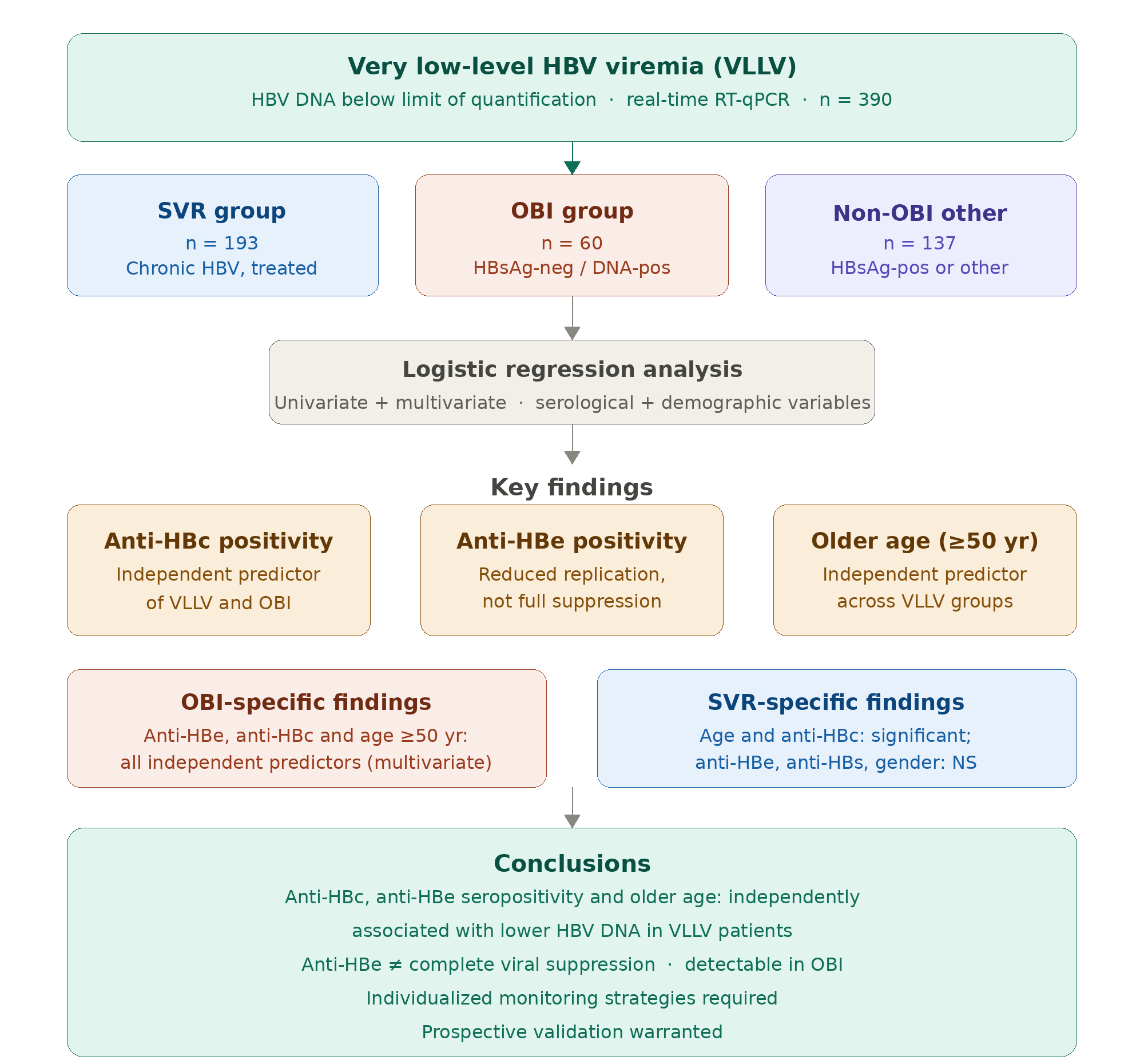
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design, Measurements and Ethical Approval
2.2. Description of OBI Diagnosis and SVR Group in the Study
2.3. Statistical Analysis
3. Results
3.1. Summary of Baseline Characteristics of the Patients
3.2. Comparison of Specific Characteristics within OBI and SVR Groups
3.3. Variables of OBI and Non-OBI Groups
3.4. Analysis of Specific Factors Among SVR Patients
4. Discussion
4.1. Limitations
5. Conclusion
References
- World Health Organization. Global Hepatitis Report 2024: Action for Access in Low- and Middle-Income Countries; WHO: Geneva, Switzerland, 2024. Available online: https://www.who.int/publications/i/item/9789240091672 (accessed on 31 August 2024).
- Xie, L.; Liu, G.; Liu, Y.; Li, P.; Guo, H. Prevalence of low-level viremia in the treatment of chronic hepatitis B in China: a systematic review and meta-analysis. BMJ Open 2025, 15, e088756. [CrossRef]
- Han, J.; Guo, Y.; Zhang, X.; Zhang, Y.; Sun, J.; He, J.; Mao, R.; Huang, Y.; Zhang, J. Prevalence and associated factors of low-level viremia in chronic hepatitis B patients after long-term therapy with nucleos(t)ide analogs. Turk. J. Gastroenterol. 2023, 34, 53–61. [CrossRef]
- Yin, G.Q.; Li, J.; Zhong, B.; Yang, Y.F.; Wang, M.R. New therapeutic options for persistent low-level viremia in patients with chronic hepatitis B virus infection: increase of entecavir dosage. World J. Gastroenterol. 2021, 27, 666–676. [CrossRef]
- Amponsah-Dacosta, E.; Selabe, S.G.; Mphahlele, M.J. Evolution of the serologic and virologic course of occult HBV infection in therapy experienced HIV co-infected patients. J. Med. Virol. 2018, 90, 291–303. [CrossRef]
- Thu, H.H.K.; Ostankova, Y.V.; Shchemelev, A.N.; Serikova, E.N.; Davydenko, V.S.; Ton, T.; Lien, T.T.X.; Ramsay, E.S.; Totolian, A.A. Unveiling the Hidden Reservoir: High Prevalence of Occult Hepatitis B and Associated Surface Gene Mutations in a Healthy Vietnamese Adult Cohort. Microorganisms 2026, 14, 238. [CrossRef]
- Allweiss, L.; Dandri, M. The Role of cccDNA in HBV Maintenance. Viruses 2017, 9, 156. [CrossRef]
- Sun, F.; Liu, Z.; Wang, B. Correlation between low-level viremia and hepatitis B-related hepatocellular carcinoma and recurrence: a retrospective study. BMC Cancer 2021, 21, 1103. [CrossRef]
- Zhang, Q.; Peng, H.; Liu, X.; Wang, H.; Du, J.; Luo, X.; Ren, H.; Hu, P. Chronic hepatitis B infection with low level viremia correlates with the progression of the liver disease. J. Clin. Transl. Hepatol. 2021, 9, 850–859. [CrossRef]
- Sun, Y.; Wu, X.; Zhou, J.; Meng, T.; Wang, B.; Chen, S.; Liu, H.; Wang, T.; Zhao, X.; Wu, S.; Kong, Y.; Ou, X.; Wee, A.; Theise, N.D.; Qiu, C.; Zhang, W.; Lu, F.; Jia, J.; You, H. Persistent low level of hepatitis B virus promotes fibrosis progression during therapy. Clin. Gastroenterol. Hepatol. 2020, 18, 2582–2591.e6. [CrossRef]
- Zhang, Q.; Cai, D.C.; Hu, P.; Ren, H. Low-level viremia in nucleoside analog-treated chronic hepatitis B patients. Chin. Med. J. 2021, 134, 2810–2817. [CrossRef]
- Im, Y.R.; Jagdish, R.; Leith, D.; Kim, J.U.; Yoshida, K.; Majid, A.; Ge, Y.; Ndow, G.; Shimakawa, Y.; Lemoine, M. Prevalence of occult hepatitis B virus infection in adults: a systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2022, 7, 932–942. [CrossRef]
- Wang, C.; Xue, R.; Wang, X.; Xiao, L.; Xian, J. High-sensitivity HBV DNA test for the diagnosis of occult HBV infection: commonly used but not reliable. Front. Cell. Infect. Microbiol. 2023, 13, 1186877. [CrossRef]
- Wang, B.; Wang, X.; Xiao, L.; Xian, J. Misuse of the lower limit of detection in HBV DNA testing and anti-HBe positive status will significantly impact the diagnosis of occult HBV infection. J. Viral Hepat. 2025, 32, e14046. [CrossRef]
- Saitta, C.; Pollicino, T.; Raimondo, G. Occult Hepatitis B Virus Infection: An Update. Viruses 2022, 14, 1504. [CrossRef]
- Jiang, X.; Tian, J.; Song, L.; Meng, J.; Yang, Z.; Qiao, W.; Zou, J. Multi-omic molecular characterization and diagnostic biomarkers for occult hepatitis B infection and HBsAg-positive hepatitis B infection. Front. Endocrinol. 2024, 15, 1409079. [CrossRef]
- Wang, B.; Zeng, T.; Yin, W.; Ren, C.; Chen, Y.; Qian, L.; Huang, P.; Zhu, C.; Yue, M. Unveiling low level viremia in chronic hepatitis B: challenges and new horizons. Front. Cell. Infect. Microbiol. 2025, 15, 1663858. [CrossRef]
- Kim, T.S.; Sinn, D.H.; Kang, W.; Gwak, G.Y.; Paik, Y.H.; Choi, M.S.; Lee, J.H.; Koh, K.C.; Paik, S.W. Hepatitis B virus DNA levels and overall survival in hepatitis B-related hepatocellular carcinoma patients with low-level viremia. J. Gastroenterol. Hepatol. 2019, 34, 2028–2035. [CrossRef]
- Li, Z.B.; Chen, D.D.; Jia, Y.F.; He, Q.J.; Cui, L.; Du, F.X.; Kang, Y.J.; Feng, X.; He, M.; Jin, X.Y.; Chen, J.; Wang, Y.; Ji, D.; Lau, G.; Wu, S.G. Risk factors related to low-level viraemia in chronic hepatitis B patients receiving entecavir treatment. Front. Cell. Infect. Microbiol. 2024, 14, 1413589. [CrossRef]
- Pronier, C.; Candotti, D.; Boizeau, L.; Bomo, J.; Laperche, S.; Thibault, V. The contribution of more sensitive hepatitis B surface antigen assays to detecting and monitoring hepatitis B infection. J. Clin. Virol. 2020, 129, 104507. [CrossRef]
- Chen, H.; Fu, J.J.; Li, L.; Wang, X.; Pan, X.C. Risk factors of low-level viremia in chronic hepatitis B patients receiving entecavir monotherapy: a retrospective cohort study. J. Gastroenterol. Hepatol. 2024, 39, 180–184. [CrossRef]
- Lu, J.; Zhang, C.; He, P.; Ou, M.; Xia, J.; Huang, M. Risk factors for very low-level viremia in patients with chronic hepatitis B virus infection: a single-center retrospective study. Liver Res. 2022, 6, 39–44. [CrossRef]
- Wu, J.; Yu, Y.; Dai, Y.; Zhang, Y.; Cheng, J. Research progress on the mechanism of persistent low-level HBsAg expression in the serum of patients with chronic HBV infection. J. Immunol. Res. 2022, 2022, 1372705. [CrossRef]
| Variable | All group (n=390) | CHB group (n=193) | OBI group (n=60) | Non-OBI group (n=137) |
| Age, years (mean±sd) | 50.9±14.2 | 49.1±13.7 | 55.6±12.6 | 51.5±14.7 |
| <50 years, n (%) | 143 (36.7) | 72 (37.3) | 16 (26.7) | 55 (40.1) |
| ≥50 years, n (%) | 247 (63.3) | 121 (62.7) | 44 (73.3) | 82 (59.9) |
| Gender | ||||
| Male, n (%) | 208 (53.3) | 102 (52.8) | 28 (46.7) | 78 (56.9) |
| Female, n (%) | 182 (46.7) | 91 (47.2) | 32 (53.3) | 59 (43.1) |
| HBcAb status | ||||
| Negative, n (%) | 89 (22.8) | 2 (1) | 9 (15) | 69 (50.4) |
| Positive, n (%) | 301 (77.2) | 191 (99) | 51 (85) | 68 (49.6) |
| HBeAb status | ||||
| Negative, n (%) | 79 (20.3) | 29 (15) | 8 (13.3) | 42 (30.7) |
| Positive, n (%) | 311 (79.7) | 164 (85) | 52 (86.7) | 95 (69.3) |
| HBsAb status | ||||
| Negative, n (%) | 310 (79.5) | 189 (97.9) | 42 (70) | 79 (57.7) |
| Positive, n (%) | 80 (20.5) | 4 (2.1) | 18 (30) | 58 (42.3) |
| Variables | Univariate analysis | Multivariate analysis | |||||||
| OR (95% CI) | P-value | OR (95% CI) | P-value | ||||||
| Age (years) ≥50 vs <50 Sex Male vs Female Anti-HBe Pos. vs Neg. Anti-HBc Pos. vs Neg. Anti-HBs Pos. vs Neg. |
1.845 (0.963-3.473) 1.511 (0.806-2.728) 2.874 (1.256- 6.172) 2.368 (1.216-4.527) 0.583 (0.304-1.089) |
0.077 0.215 0.012 0.008 0.113 |
4.055 (2.236-6.981) / 7.893 (4.117-12.66) 11.41 (4.149-26.18) / |
0.044 / 0.005 <0.001 / |
|||||
| Variables | Univariate analysis | Multivariate analysis | |||||||
| OR (95% CI) | P-value | OR (95% CI) | P-value | ||||||
| Age (years) ≥50 vs <50 Sex Male vs Female Anti-HBe Pos. vs Neg. Anti-HBc Pos. vs Neg. Anti-HBs Pos. vs Neg. |
0.596 (0.396-0.888) 1.265 (0.853-1.882) 1.505 (0.884- 2.545) 13.76 (3.69-59.56) 0.170 (0.062-0.482) |
0.014 0.264 0.144 <0.001 0.176 |
5.500 (2.087-12.547) / / 21.18 (6.011-60.266) / |
0.019 / / <0.001 / |
|||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




