Submitted:
28 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
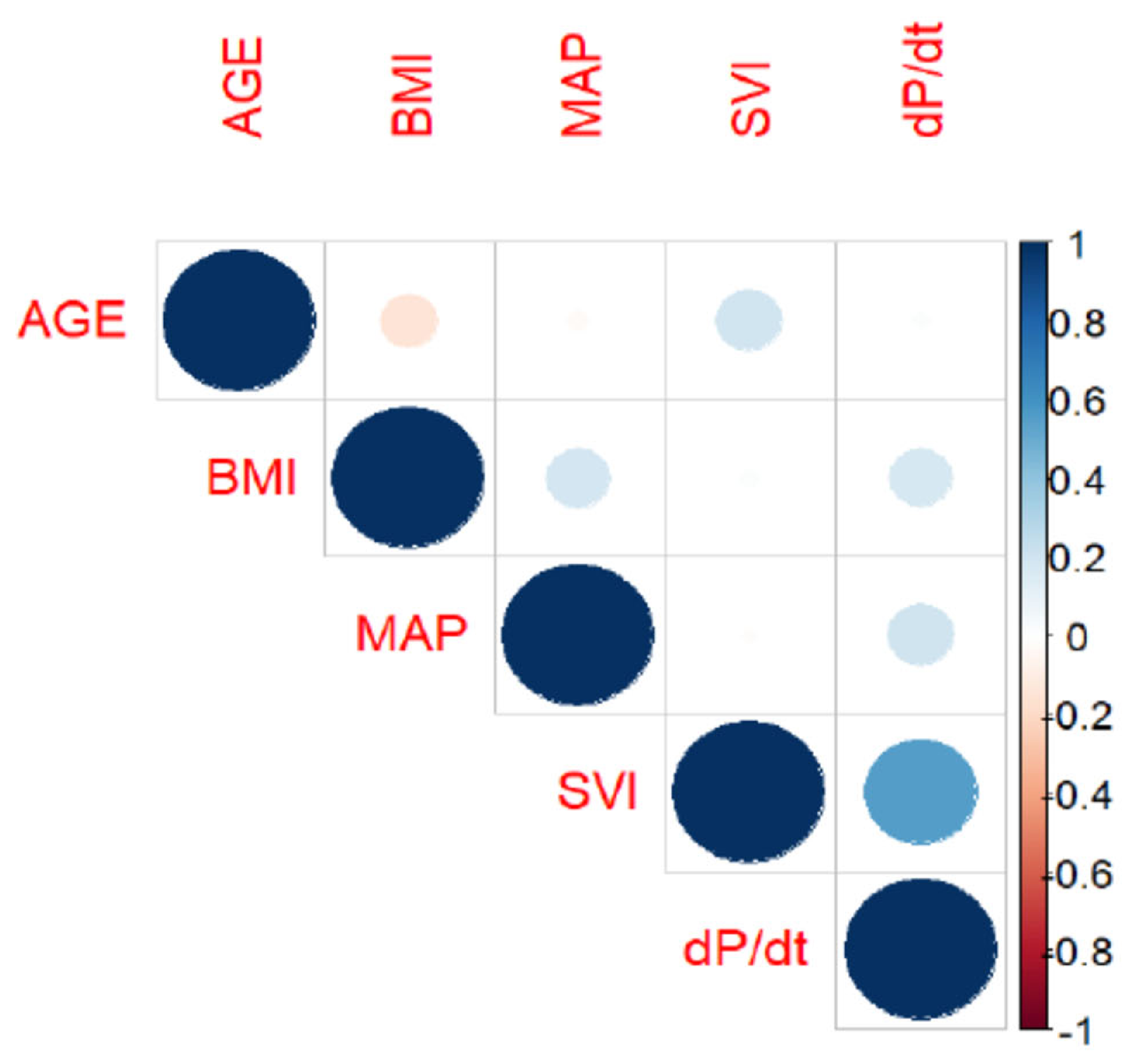
2.1. Statistical Analysis
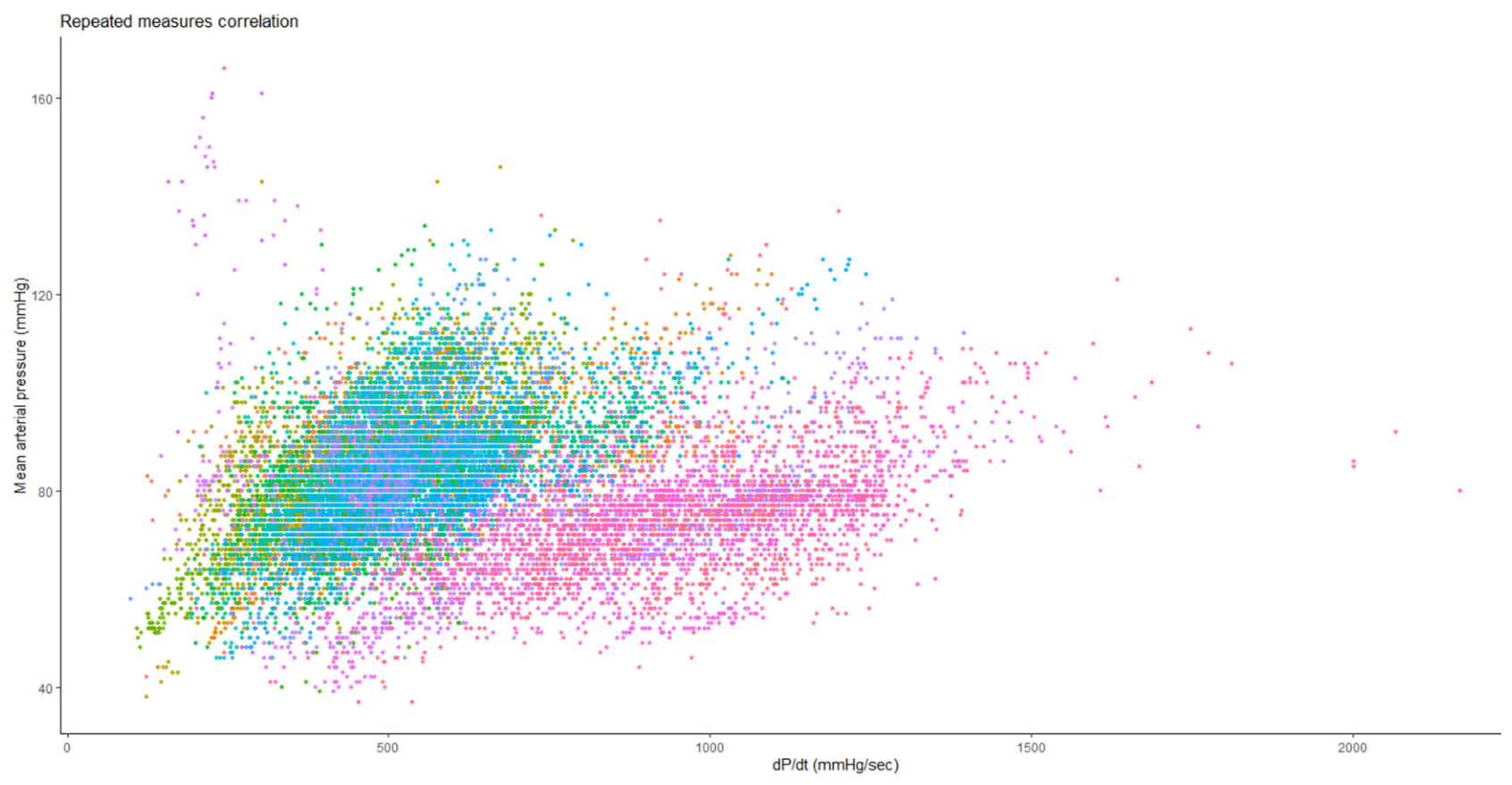
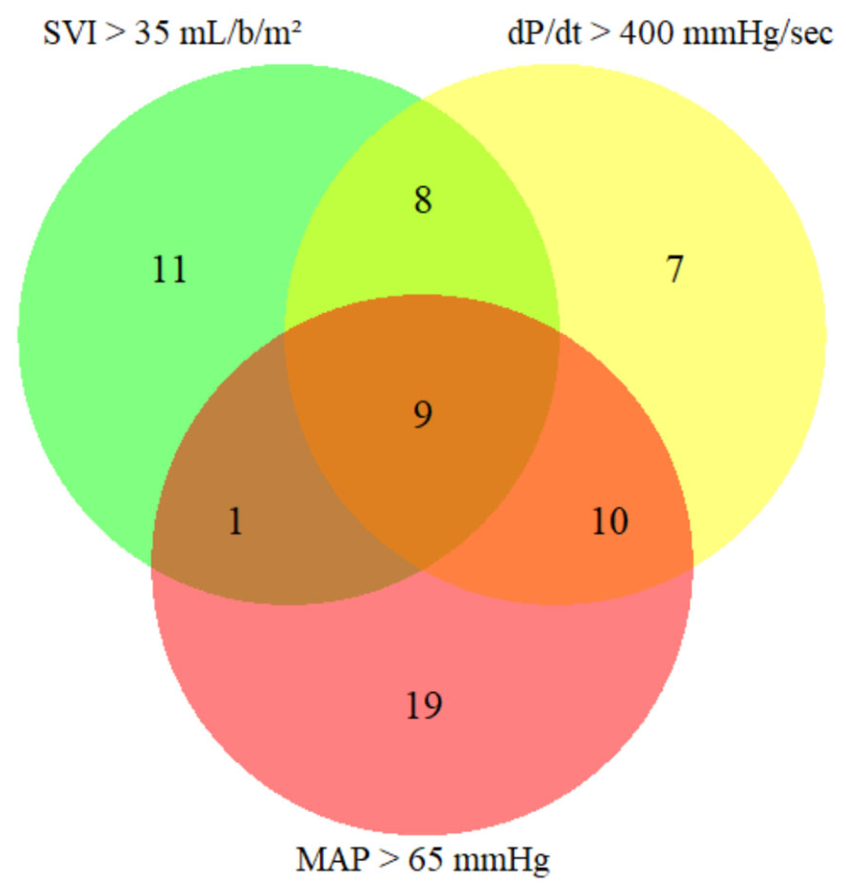
3. Results
3.1. Intraoperative Hypotension
3.2. Primary Outcome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| SA | Spinal anesthesia |
| CD | cesarean delivery |
| CO | cardiac output |
| HR | heart rate |
| SV | stroke volume |
| NIBP | non-invasive blood pressure |
| SVI | stroke volume index |
| dP/dtmax | rise of the arterial pressure |
| ASA | American Society of Anesthesiologists |
| BMI | body mass index |
| IQR | interquartile range |
| TWA | time weighed average |
References
- Landau R, Sultan P. Neuraxial anesthesia and pain management for cesarean delivery. Am J Obstet Gynecol. 2026;233(6S):S135-S152. [CrossRef]
- Ngan Kee WD. Prevention of maternal hypotension after regional anaesthesia for caesarean section. Curr Opin Anaesthesiol. 2010;23(3):304–309.
- Kinsella SM, Carvalho B, Dyer RA, et al. International consensus statement on the management of hypotension with vasopressors during caesarean section under spinal anaesthesia. Anaesthesia. 2018;73(1):71–92. [CrossRef]
- Dyer RA, Reed AR, van Dyk D, et al. Hemodynamic effects of ephedrine, phenylephrine, and the coadministration of phenylephrine with oxytocin during spinal anesthesia for elective cesarean delivery. Anesthesiology. 2009;111(4):753-65. [CrossRef]
- Tsen LC, Gelman S. The venous system during pregnancy. Part 1: physiologic considerations. Int J Obstet Anesth. 2022;50:103273. [CrossRef]
- Tsen LC, Gelman S. The venous system during pregnancy. Part 2: clinical implications. Int J Obstet Anesth. 2022;50:103274. [CrossRef]
- van Oppen AC, van der Tweel I, Alsbach GP, Heethaar RM, Bruinse HW. A longitudinal study of maternal hemodynamics during normal pregnancy. Obstet Gynecol. 1996;88(1):40–6.
- Sanghavi M, Rutherford JD. Cardiovascular physiology of pregnancy. Circulation. 2014;130(12):1003–8.
- Robson SC, Hunter S, Boys RJ, Dunlop W. Serial study of factors influencing changes in cardiac output during human pregnancy. Am J Physiol. 1989;256(4 Pt 2):H1060–H1065. [CrossRef]
- Ueland K, Metcalfe J. Circulatory changes in pregnancy. Clin Obstet Gynecol. 1975;18(1):41–50.
- Aya AG, Vialles N, Tanoubi I, et al. Spinal anesthesia-induced hypotension: a risk comparison between patients with severe preeclampsia and healthy women undergoing cesarean delivery. Anesth Analg. 2005;101(3):869–875. [CrossRef]
- Ilies C, Gruenewald M, Ludwigs J, et al. Evaluation of the CNAP™ system for continuous non-invasive blood pressure monitoring in parturients undergoing caesarean section. Br J Anaesth. 2012;108(3):413–418.
- Vos JJ, Poterman M, Mooyaart EAQ, et al. Non-invasive continuous arterial pressure and cardiac output during caesarean section: a comparison of Nexfin and LiDCOrapid. Br J Anaesth. 2014;112(4):609–615.
- Vasile F, La Via L, Murabito P, et al. Non-Invasive Monitoring during Caesarean Delivery: Prevalence of Hypotension and Impact on the Newborn. J Clin Med. 2023;12(23):7295. [CrossRef]
- Scolletta S, Bodson L, Donadello K, et al. Assessment of left ventricular systolic function by pulse wave analysis in critically ill patients. Anaesthesia. 2013;68(7):700–707. [CrossRef]
- Ostadal P, Vondrakova D, Krüger A, Janotka M, Naar J. Continual measurement of arterial dP/dtmax enables minimally invasive monitoring of left ventricular contractility in patients with acute heart failure. Crit Care. 2019;23(1):364. [CrossRef]
- Monge García MI, Jian Z, Settels JJ, et al. Performance comparison of ventricular and arterial dP/dtmax for assessing left ventricular systolic function during different experimental loading and contractile conditions. Crit Care. 2018;22(1):325.
- Guinot PG, Marienne JP, Signouret T, et al. Ability of arterial dP/dtmax to predict fluid responsiveness in the operating room. Br J Anaesth. 2013;110(4):615–622.
- Frassanito L, Sonnino C, Piersanti A, et al. Performance of the Hypotension Prediction Index With Noninvasive Arterial Pressure Waveforms in Awake Cesarean Delivery Patients Under Spinal Anesthesia. Anesth Analg. 2022;134(3):633-643. [CrossRef]
- Heerman JR, Segers P, Roosens CD, Gasthuys F, Verdonck PR, Poelaert JI. Echocardiographic assessment of aortic elastic properties with automated border detection in an ICU: in vivo application of the arctangent Langewouters model. Am J Physiol Heart Circ Physiol. 2005;288(5):H2504-11.
- Wesseling KH, de Wit B, van der Hoeven GMA, van Goudoever J, Settels JJ. Physiocal, calibrating finger vascular physiology for finapres. Homeostasis in Health and Disease. 1995;36(2-3),67-82.
- Maheshwari K, Khanna S, Bajracharya GR, et al. A Randomized Trial of Continuous Noninvasive Blood Pressure Monitoring During Noncardiac Surgery. Anesth Analg. 2018;127(2):424-431. [CrossRef]
- Langesaeter E, Dyer RA. Maternal haemodynamic changes during spinal anaesthesia for caesarean section. Curr Opin Anaesthesiol. 2011;24(3):242-248.
- Dyer RA, Farina Z, Joubert IA, et al. Crystalloid preload versus rapid crystalloid administration after induction of spinal anaesthesia (co-load) for elective caesarean section. Anaesth Intensive Care. 2004;32(3):351–357. [CrossRef]
- Langesæter E, Gibbs M, Dyer RA. The role of cardiac output monitoring in obstetric anesthesia. Curr Opin Anaesthesiol. 2015;28(3):247-53. [CrossRef]
- Tawfik MM, Hafez SM, Abdelmohaymen HA, Ismail OM. Serial echocardiographic measurements of cardiac output after spinal anesthesia for scheduled cesarean delivery in healthy patients: a prospective observational study. Int J Obstet Anesth. 2025;64:104752. [CrossRef]
- Juri T, Suehiro K, Kimura A, et al. Impact of non-invasive continuous blood pressure monitoring on maternal hypotension during cesarean delivery: a randomized-controlled study. J Anesth. 2018;32(6):822-830. [CrossRef]
- Shih PY, Wei TJ, Lee CT, et al. Proactive haemodynamic management using the hypotension prediction index during caesarean section: a randomised controlled study. Anaesthesia. 2026 Feb 15. [CrossRef]
- Higgins N, Fitzgerald PC, van Dyk D, et al. The Effect of Prophylactic Phenylephrine and Ephedrine Infusions on Umbilical Artery Blood pH in Women With Preeclampsia Undergoing Cesarean Delivery With Spinal Anesthesia: A Randomized, Double-Blind Trial. Anesth Analg. 2018;126(6):1999-2006.
- Hasanin A, Amin S, Agiza A, et al. Norepinephrine versus ephedrine to maintain arterial blood pressure during spinal anesthesia for cesarean delivery: a prospective double-blinded trial. Int J Obstet Anesth. 2019;39:81-87.
- Ngan Kee WD, Lee SWY, Ng FF, Khaw KS. Prophylactic Norepinephrine Infusion for Preventing Hypotension During Spinal Anesthesia for Cesarean Delivery. Anesth Analg. 2018;126(6):1989-1994.
- Aguilar Molina O, Barbosa Balaguera S, Campo-Rivera N, et al. Normal echocardiographic findings in healthy pregnant women: A narrative review of the literature. Curr Probl Cardiol. 2025;50(3):102969.
- Melchiorre K, Sharma R, Khalil A, Thilaganathan B. Maternal Cardiovascular Function in Normal Pregnancy: Evidence of Maladaptation to Chronic Volume Overload. Hypertension. 2016;67(4):754-62.
- Estensen ME, Beitnes JO, Grindheim G, et al. Altered maternal left ventricular contractility and function during normal pregnancy. Ultrasound Obstet Gynecol. 2013;41(6):659-66. [CrossRef]
- Sengupta SP, Bansal M, Hofstra L, Sengupta PP, Narula J. Gestational changes in left ventricular myocardial contractile function: new insights from two-dimensional speckle tracking echocardiography. Int J Cardiovasc Imaging. 2017;33(1):69-82. [CrossRef]
- Kuck K, Baker PD. Perioperative Noninvasive Blood Pressure Monitoring. Anesth Analg. 2018;127(2):408-411. [CrossRef]
- Boisson M, Poignard ME, Pontier B, Mimoz O, Debaene B, Frasca D. Cardiac output monitoring with thermodilution pulse-contour analysis vs. non-invasive pulse-contour analysis. Anaesthesia. 2019;74(6):735-740. [CrossRef]
- D’Amico F, Pruna A, Putowski Z, et al. Low Versus High Blood Pressure Targets in Critically Ill and Surgical Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Crit Care Med. 2024;52(9):1427-1438. [CrossRef]
- Kant M, van Klei WA, Hollmann MW, et al. Proactive vs Reactive Treatment of Hypotension During Surgery: The PRETREAT Randomized Clinical Trial. JAMA. 2025;334(21):1905-1914.
- Vaquer S, Chemla D, Teboul JL, et al. Influence of changes in ventricular systolic function and loading conditions on pulse contour analysis-derived femoral dP/dtmax. Ann Intensive Care. 2019;9(1):61.
- Vaquer S, Chemla D, Teboul JL, et al. Volume Infusion Markedly Increases Femoral dP/dtmax in Fluid-Responsive Patients Only. Crit Care Med. 2020 Oct;48(10):1487-1493.
| Characteristic | Total patients (N=95) |
|---|---|
| Age, years | 35 (5) |
| Height, cm | 163 (7) |
| Weight, kg | 72 (66, 84) |
| Body mass index, kg/m2 | 27 (24, 29) |
| Body surface area, m2 | 1.8 (0.2) |
| Medical history | |
| None | 40 (42) |
| Class I obesity | 25 (26) |
| Gestational diabetes | 15 (16) |
| Hypothyroidism | 15 (16) |
| Hypertension | 1 (1) |
| Maternal cholestasis of pregnancy | 1 (1) |
| Cancer | 4 (4) |
| Other* | 6 (6) |
| Twin pregnancy | 8 (8) |
| Indications to Cesarean section | |
| Previous Cesarean delivery | 52 (55) |
| Multiple gestation ± malpresentation | 19 (20) |
| Prior myomectomy | 9 (9) |
| Fetal macrosomia | 1 (1) |
| Placenta previa/accreta | 2 (2) |
| Other maternal conditions | 6 (6) |
| Maternal request | 6 (6) |
| Characteristic | Total patients (N=95) |
|---|---|
| Duration of surgery, min | 83 (69, 95) |
| dP/dtmax | |
| Patients with mean dP/dtmax<400 mmHg/sec | 60 (63) |
| Total number of events with dP/dtmax<400 mmHg/sec | 305 |
| TWA of area of dP/dtmax<400 mmHg/sec, mmHg/sec | 12.71 (3.41, 27.54) |
| Area for dP/dtmax<400 mmHg/sec, mmHg/sec·min | 1051 (333, 2248) |
| Mean dP/dtmax<400 mmHg/sec, mmHg/sec | 346 (326, 366) |
| Total duration of events with dP/dtmax<400 mmHg/sec, sec | 17 (5, 29) |
| Average duration of events with dP/dtmax<400 mmHg/sec, sec | 3 (2, 4) |
| SVI | |
| Patients with SVI <35 mL/b/m2 | 66 (69) |
| Total number of events with SVI <35 mL/b/m2 | 336 |
| TWA of area of SVI <35 mL/b/m2, mL/b/m2 | 1.12 (0.44, 3.44) |
| Area for SVI <35 mL/b/m2, mL/b/m²·min | 106.16 (31.42, 293.91) |
| Mean SVI <35 mL/b/m2, mL/b/m2 | 30.18 (28.53, 31.81) |
| Total duration of events with SVI <35 mL/b/m2, sec | 24 (7, 42) |
| Average duration of events with SVI <35 mL/b/m2, sec | 4 (2, 8) |
| MAP | |
| Total number of events with MAP <65 mmHg | 209 |
| Patients with MAP <65 mmHg | 56 (59) |
| Number of events with MAP <65 mmHg per patient | 2 (1, 5) |
| TWA-MAP <65 mmHg per patient, mmHg | 0.55 (0.21, 1.13) |
| Area for MAP <65 mmHg per patient, mmHg·min | 36 (17, 123) |
| Duration of hypotensive events <65 mmHg per patient, min | 4 (2, 17) |
| Percentage of time with MAP <65 mmHg | 5 (2, 15) |
| Vasopressors | |
| Patients who received norepinephrine | 57 (60) |
| Total administered norepinephrine dose, mcg | 15 (10, 35) |
| Patients who received atropine | 2 (2) |
| Total administered atropine dose, mg | 0.5 (0.5, 0.5) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




