Submitted:
28 April 2026
Posted:
30 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
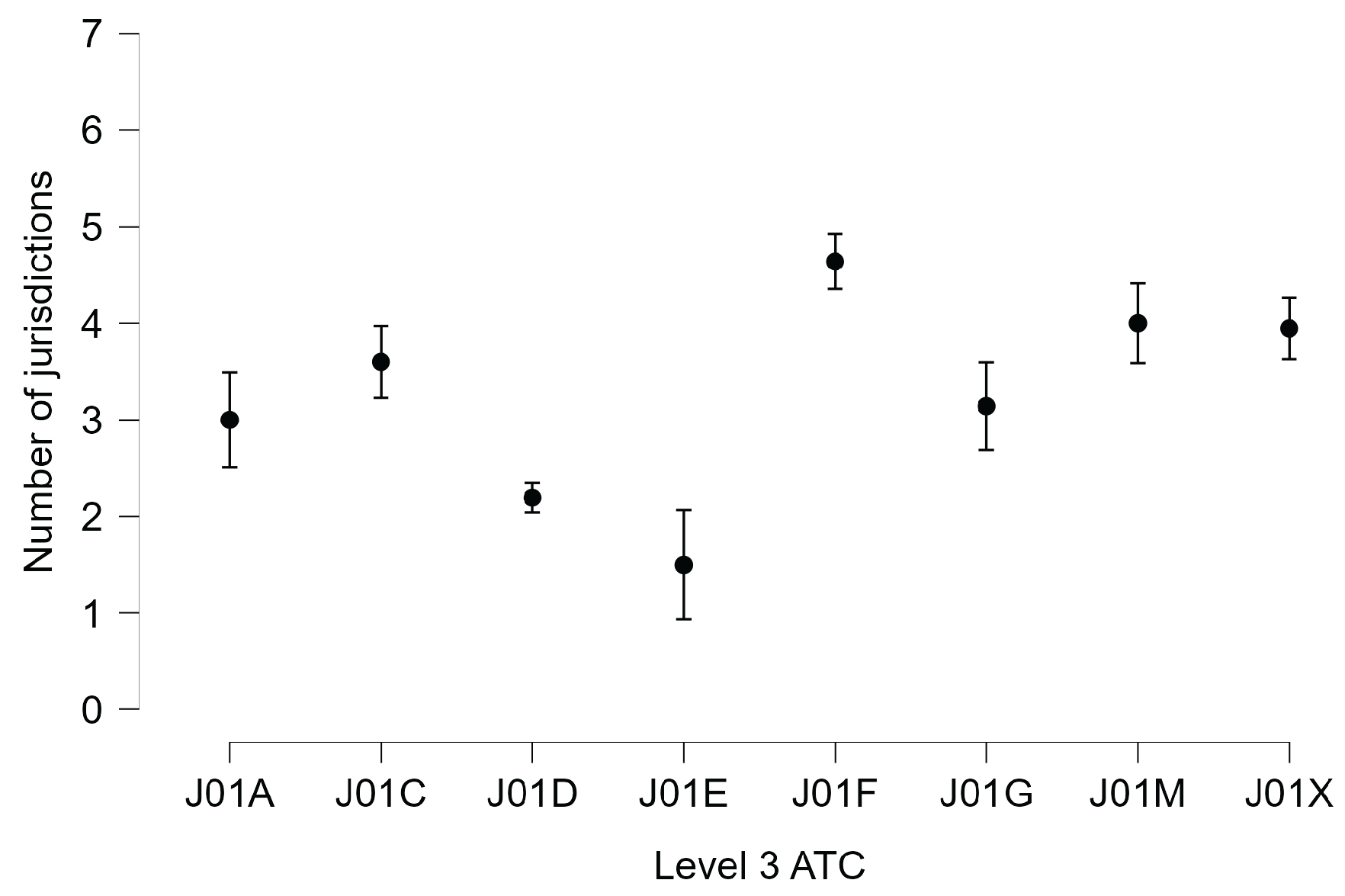
2.1. The Distribution of Antibiotic Discontinuities Across Countries and Therapeutic Classes
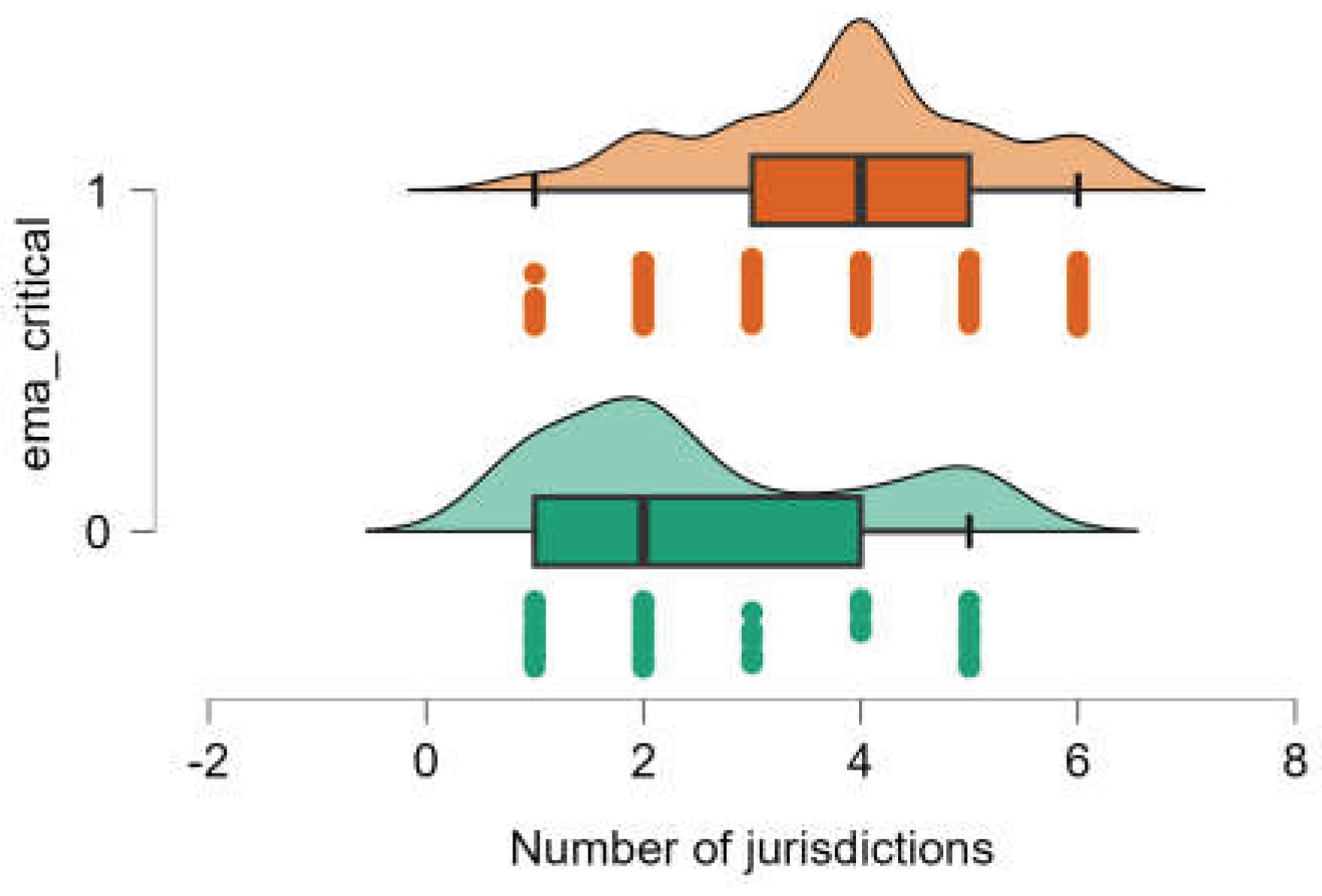
2.2. Association Between Shortages and EMA Critical Medicines
2.3. Differences in the Territorial Extent of Shortages Between EMA-Designated Critical and Non-Critical Antibiotics
2.4. Country-Level Comparison of EMA Critical Shortages
| Model | Deviance | AIC | BIC | df | ΔΧ2 | p | McFadden R2 | Nagelkerke R2 | Tjur R2 | Cox & Snell R2 |
| M0 | 432.6 | 434.567 | 438.425 | 349 | 0.000 | 0.000 | ||||
| M1 | 362.1 | 368.123 | 379.696 | 347 | 70.44 | 5.551×10-16 | 0.163 | 0.257 | 0.204 | 0.182 |
2.5. Multivariable Analysis
2.6. Sensitivity Analysis: The Effect of Variation in the Threshold on the Main Results
2.7. Data Limitations and Justification of the Type of Analysis
2.8. Policy Implications
3. Discussion
4. Materials and Methods
4.1. Study Design and Data Sources
4.2. Data Harmonization and Construction of the Master Dataset
4.3. Variables and Derived Indicators
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ATC | Anatomical Therapeutic Chemical (classification) |
| AUC | Area Under the Curve |
| AIC | Akaike Information Criterion |
| BIC | Bayesian Information Criterion |
| CI | Confidence Interval |
| EMA | European Medicines Agency |
| EU | European Union |
| FDA | Food and Drug Administration |
| INN | International Nonproprietary Name |
| LMICs | Low- and Middle-Income Countries |
| OR | Odds Ratio |
| ROC | Receiver Operating Characteristic |
| SD | Standard Deviation |
| VIF | Variance Inflation Factor |
| WHO | World Health Organization |
References
- Fox, E.R.; Sweet, B. V.; Jensen, V. Drug Shortages: A Complex Health Care Crisis. Mayo Clin. Proc. 2014, 89, 361–373. [Google Scholar] [CrossRef]
- Chirac, O.; Tăerel, A.-E.; Dinu, M.; Ancuceanu, R. SUPPLY CHAIN VULNERABILITY AND REGULATORY GAPS: AN EVALUATION OF DRUG SHORTAGES IN THE ROMANIAN PHARMACEUTICAL MARKET ( 2008-2024 ). Farmacia 2026, 74. [Google Scholar]
- Cornelissen, N.; Zielhuis, S.W.; van den Bemt, P.M.L.A.; van den Bemt, B.J.F. Causes and Management of Drug Shortages: A Scoping Review. Res. Soc. Adm. Pharm. 2026, 22, 232–253. [Google Scholar] [CrossRef]
- Phuong, J.M.; Penm, J.; Chaar, B.; Oldfield, L.D.; Moles, R. The Impacts of Medication Shortages on Patient Outcomes: A Scoping Review. PLoS ONE 2019, 14, e0215837. [Google Scholar] [CrossRef]
- Postma, D.J.; Notenboom, K.; De Smet, P.A.G.M.; Leufkens, H.G.M.; Mantel-Teeuwisse, A.K. Medicine Shortages: Impact behind Numbers. J. Pharm. Policy Pract. 2023, 16. [Google Scholar] [CrossRef]
- Shafiq, N.; Pandey, A.K.; Malhotra, S.; Holmes, A.; Mendelson, M.; Malpani, R.; Balasegaram, M.; Charani, E. Shortage of Essential Antimicrobials: A Major Challenge to Global Health Security. BMJ Glob. Heal. 2021, 6, e006961. [Google Scholar] [CrossRef]
- Skender, B.; Zhang, M. From Local Issue to Global Challenge: A Brief Overview of Antibiotic Shortages since the 1970s. Humanit. Soc. Sci. Commun. 2024, 11, 1242. [Google Scholar] [CrossRef]
- Allaw, F.; Vu Thi Lan, H.; Nagao, M.; Ndegwa, L.; Levy Hara, G.; Kanj, S.S.; Tattevin, P. Antibiotic Shortages: An Overview by the Alliance for the Prudent Use of Antibiotics (APUA). Int. J. Antimicrob. Agents 2025, 65, 107456. [Google Scholar] [CrossRef]
- Miljković, N.; Polidori, P.; Kohl, S. Managing Antibiotic Shortages: Lessons from EAHP and ECDC Surveys. Eur. J. Hosp. Pharm. Sci. Pract. 2022, 29, 90–94. [Google Scholar] [CrossRef]
- Claus, B.; Pauwels, K.; Baert, M.; Depoorter, J.; De Weerdt, E.; Boussery, K.; De Spiegeleer, B.; Commeyne, S. Drug Shortages in the Hospital: Management, Causes and Budget Impact. J. Pharm. Belg. 2015, 24–34. [Google Scholar]
- Agency, E.M. EMA Update on Shortages of Antibiotics in the EU. Available online: https://www.ema.europa.eu/en/news/ema-update-shortages-antibiotics-eu.
- Beck, M.; Buckley, J. Managing Pharmaceutical Shortages during the COVID Pandemic: An Exploratory Analysis of European Collective and National Government Responses. J. Med. Access 2022, 6. [Google Scholar] [CrossRef]
- Popp, M.; Stegemann, M.; Riemer, M.; Metzendorf, M.-I.; Romero, C.S.; Mikolajewska, A.; Kranke, P.; Meybohm, P.; Skoetz, N.; Weibel, S. Antibiotics for the Treatment of COVID-19. Cochrane Database Syst. Rev. 2021, 2022. [Google Scholar] [CrossRef]
- Shukar, S.; Zahoor, F.; Hayat, K.; Saeed, A.; Gillani, A.H.; Omer, S.; Hu, S.; Babar, Z.-U.-D.; Fang, Y.; Yang, C. Drug Shortage: Causes, Impact, and Mitigation Strategies. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Kohl, S. EMA Guidance for Industry to Prevent and Mitigate Medicine Shortages. Eur. J. Hosp. Pharm. Sci. Pract. 2023, 30, 245–246. [Google Scholar] [CrossRef]
- European Medicines Agency Union List of Critical Medicines. Available online: https://www.ema.europa.eu/en/human-regulatory-overview/post-authorisation/medicine-shortages-availability-issues/availability-medicines-during-crises/union-list-critical-medicines (accessed on 5 March 2026).
- European Medicines Agency Methodology to Identify Critical Medicines for the “Union List of Critical Medicines”. 2023.
- McGeeney, J.D.; McAden, E.; Sertkaya, A. Analysis of Drug Shortages. In Prep. by East. Res. Group, Inc. Off. ofthe Assist. Secr. Plan. Eval. (ASPE), U.S.Department Heal. Hum. Serv.; 2025. [Google Scholar]
- van Oorschot, K.E.; Van Wassenhove, L.N.; Jahre, M.; Selviaridis, K.; de Vries, H. Drug Shortages: A Systems View of the Current State. Decis. Sci. 2022, 53, 969–984. [Google Scholar] [CrossRef]
- De Weerdt, E.; Simoens, S.; Hombroeckx, L.; Casteels, M.; Huys, I. Causes of Drug Shortages in the Legal Pharmaceutical Framework. Regul. Toxicol. Pharmacol. 2015, 71, 251–258. [Google Scholar] [CrossRef]
- Balkhi, B.; Araujo-Lama, L.; Seoane-Vazquez, E.; Rodriguez-Monguio, R.; Szeinbach, S.L.; Fox, E.R. Shortages of Systemic Antibiotics in the USA: How Long Can We Wait? J. Pharm. Heal. Serv. Res. 2013, 4, 13–17. [Google Scholar] [CrossRef]
- Panteli, D.; Anderson, M.; Fieldman, T.; Baraldi, E.; Tängdén, T.; Vogler, S.; Årdal, C.; Mossialos, E. Policy Options for Sustainable Access to Off-Patent Antibiotics in Europe. Npj Antimicrob. Resist. 2024, 2, 40. [Google Scholar] [CrossRef]
- Baraldi, E.; Årdal, C.; Aho, E.; Popescu, G.-A.; Melaku, T. The Multifaceted Nature of Lack of Access to Antibiotics: Types of Shortage and Specific Causes, Consequences, and Solutions. Clin. Microbiol. Infect. 2025, 31, 333–338. [Google Scholar] [CrossRef]
- Dylst, P.; Simoens, S. Generic Medicine Pricing Policies in Europe: Current Status and Impact. Pharmaceuticals 2010, 3, 471–481. [Google Scholar] [CrossRef]
- Toshev, A.; Mihaylova, A.A.; Madzharov, V.; Petkova-Gueorguieva, E.; Kirilov, B.; Mirchev, D.; Gueorguiev, S. The European Union‘s Dependence on Imports of Active Pharmaceutical Ingredients from Third Countries – Challenges and EU Initiatives. Pharmacia 2025, 72, 1–6. [Google Scholar] [CrossRef]
- Socal, M.P.; Ahn, K.; Greene, J.A.; Anderson, G.F. Competition And Vulnerabilities In The Global Supply Chain For US Generic Active Pharmaceutical Ingredients. Health Aff. 2023, 42, 407–415. [Google Scholar] [CrossRef]
- Anderson, M.; Panteli, D.; van Kessel, R.; Ljungqvist, G.; Colombo, F.; Mossialos, E. Challenges and Opportunities for Incentivising Antibiotic Research and Development in Europe. Lancet Reg. Heal.-Eur. 2023, 33, 100705. [Google Scholar] [CrossRef]
- Laxminarayan, R.; Matsoso, P.; Pant, S.; Brower, C.; Røttingen, J.-A.; Klugman, K.; Davies, S. Access to Effective Antimicrobials: A Worldwide Challenge. Lancet 2016, 387, 168–175. [Google Scholar] [CrossRef]
- European Medicines Agency Amoxicillin and Amoxicillin/Clavulanic Acid - Supply Shortage. Available online: https://www.ema.europa.eu/en/medicines/human/shortages/amoxicillin-amoxicillin-clavulanic-acid.
- Pagano, F.; De Marco, G.; Trojano, B.; Amato, C.; Micillo, M.; Cecere, G.; Guarino, A.; Lo Vecchio, A. Impact of Amoxicillin Shortage on Pediatric Antibiotic Prescriptions in Primary Care. Antibiot. 2025, 14. [Google Scholar] [CrossRef] [PubMed]
- Marino, A.; Maniaci, A.; Lentini, M.; Ronsivalle, S.; Nunnari, G.; Cocuzza, S.; Parisi, F.M.; Cacopardo, B.; Lavalle, S.; La Via, L. The Global Burden of Multidrug-Resistant Bacteria. Epidemiologia 2025, 6, 21. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Cohn, J.; Nampoothiri, V.; Gadde, U.; Ghataure, A.; Kakkar, A.K.; Gupta; Yogendra, K.; Malhotra, S.; Mbamalu, O.; Mendelson, M.; et al. A Systematic Review of Antibiotic Drug Shortages and the Strategies Employed for Managing These Shortages. Clin. Microbiol. Infect. 2025, 31, 345–353. [Google Scholar] [CrossRef]
- Parnham, M.J.; Haber, V.E.; Giamarellos-Bourboulis, E.J.; Perletti, G.; Verleden, G.M.; Vos, R. Azithromycin: Mechanisms of Action and Their Relevance for Clinical Applications. Pharmacol. Ther. 2014, 143, 225–245. [Google Scholar] [CrossRef] [PubMed]
- Pineda-Moncusí, M.; Rekkas, A.; Martínez Pérez, Á.; Leis, A.; Lopez Gomez, C.; Fey, E.; Bruninx, E.; Maljković, F.; Sánchez-Sáez, F.; Rodeiro-Boliart, J.; et al. Changes in Use and Utilisation Patterns of Drugs with Reported Shortages between 2010 and 2024 in Europe and North America: A Network Cohort Study. Lancet Public Heal. 2025, 10, e835–e847. [Google Scholar] [CrossRef]
- Martingano, D.; Nguyen, A.; Nkeih, C.; Singh, S.; Mitrofanova, A. Clarithromycin Use for Adjunct Surgical Prophylaxis before Non-Elective Cesarean Deliveries to Adapt to Azithromycin Shortages in COVID-19 Pandemic. PLoS ONE 2020, 15, e0244266. [Google Scholar] [CrossRef]
- Radford-Smith, D.E.; Anthony, D.C. Vancomycin-Resistant E. Faecium: Addressing Global and Clinical Challenges; Antibiot.: Basel, Switzerland, 2025. [Google Scholar] [CrossRef]
- Faggioni, F.; Valerio Rossi, M.; Sestino, A. Supply Chain Resilience in the Pharmaceutical Industry: A Qualitative Analysis from Scholarly and Managerial Perspectives. Int. J. Bus. Manag. 2023, 18, 129. [Google Scholar] [CrossRef]
- Lőrinczy, L.; Turbucz, B.; Hankó, B.; Zelkó, R. Managing Antibiotic Shortages in Inpatient Care—A Review of Recent Years in Comparison with the Hungarian Status. Antibiotics 2023, 12, 1704. [Google Scholar] [CrossRef] [PubMed]
- Bade, C.; Olsacher, A.; Boehme, P.; Truebel, H.; Fehring, L. Reasons for Supply Side Driven Drug Shortages – A Mixed-Methods Study on First-Level, Higher-Level, and Root Causes from the Perspective of Marketing Authorization Holders. Res. Soc. Adm. Pharm. 2023, 19, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- FRANK, R.G.; MCGUIRE, T.G.; NASON, I. The Evolution of Supply and Demand in Markets for Generic Drugs. Milbank Q. 2021, 99, 828–852. [Google Scholar] [CrossRef] [PubMed]
- Bartoo, A.S.; Gilmer, M.A.; Tichy, E.M. Antimicrobial Shortages: A Global Issue Impacting Infectious Diseases. Clin. Infect. Dis. 2025, 80, 249–252. [Google Scholar] [CrossRef]
- Cooke, E.; Rendi-Wagner, P. Rising Antimicrobial Resistance in Europe: EMA and ECDC Outline Urgent Actions to Achieve EU Targets. Lancet Reg. Heal. Eur. 2025, 59, 101539. [Google Scholar] [CrossRef]
- Garattini, L.; Finazzi, B.; Mazzone, A. Antibiotic Shortages in Europe: Another Question of Prices? Eur. J. Intern. Med. 2024, 125, 142–144. [Google Scholar] [CrossRef]
- Lambert, M.; Taxis, K.; Pont, L. Impact of Antibiotic Shortages on Antibiotic Utilisation in the Community. Pharmacoepidemiol. Drug. Saf. 2025, 34, e70107. [Google Scholar] [CrossRef]
- Lan, Y.; Lin, X.; Chen, Q.; Wang, L.; Sun, L.; Huang, Z. Drug Supply and Assurance: A Cross-Sectional Study of Drug Shortage Monitoring Varieties in China. BMC Public Health 2024, 24, 2048. [Google Scholar] [CrossRef]
- Aronson, J.K.; Heneghan, C.; Ferner, R.E. Drug Shortages. Part 2: Trends, Causes and Solutions. Br. J. Clin. Pharmacol. 2023, 89, 2957–2963. [Google Scholar] [CrossRef]
- Francois, C.; Gawlik, G.; Mestre-Ferrandiz, J.; Pana, A.; Perelman, J.; Yfantopoulos, J.; Simoens, S. New Pricing Models for Generic Medicines to Ensure Long-Term Sustainable Competition in Europe. Front. Pharmacol. 2023, 14, 1200641. [Google Scholar] [CrossRef]
- Romanian Association of International Pharmaceutical Manufacturers (ARPIM) Pricing Policy and Clawback Tax, Causes for Drug Shortages. Available online: https://arpim.ro/politica-de-preturi-si-taxa-clawback-cauzele-pentru-lipsa-medicamentelor/ (accessed on 30 July 2025).
- Asaloș, N.; Roman, C.; Bostan, P. Pharma Sector under the Influence of the Parafiscal Mechanism: Clawback Tax. Ovidius Univ. Ann. Econ. Sci. Ser. 2023, 23, 869–875. [Google Scholar] [CrossRef]
- Roberts, M.C. Environmental Macrolide–Lincosamide–Streptogramin and Tetracycline Resistant Bacteria. Front. Microbiol. 2011, 2. [Google Scholar] [CrossRef]
- Taha, I.; Abdou, Y.; Hammad, I.; Nady, O.; Hassan, G.; Farid, M.F.; Alofi, F.S.; Alharbi, N.; Salamah, E.; Aldeeb, N.; et al. Utilization of Antibiotics for Hospitalized Patients with Severe Coronavirus Disease 2019 in Al-Madinah Al-Munawara, Saudi Arabia: A Retrospective Study. Infect. Drug. Resist. 2022, Volume 15, 7401–7411. [Google Scholar] [CrossRef]
- European Medicines Agency. Good Practices for Industry for the Prevention of Human Medicinal Product Shortages.
- World Health Organization Model List of Essential Medicines. Available online: https://list.essentialmeds.org/ (accessed on 8 March 2026).
- Des, A. fédérale; Santé, médicaments et des produits de Belgium National Register of Shortages. Available online: https://banquededonneesmedicaments.fagg-afmps.be/usage-humain (accessed on 7 January 2026).
- Agence nationale de sécurité du médicament et des produits de santé France National Register of Shortages. Available online: https://ansm.sante.fr/disponibilites-des-produits-de-sante/medicaments (accessed on 6 January 2026).
- BfArM - Federal Institute for Drugs and Medical Devices Germany National Register of Shortages. Available online: https://anwendungen.pharmnet-bund.de/lieferengpassmeldungen/faces/public/meldungen.xhtml (accessed on 7 January 2026).
- Agenția Națională a Medicamentului și a Dispozitivelor Medicale din Romania Romania National Register of Shortages. Available online: https://www.anm.ro/medicamente-de-uz-uman/autorizare-medicamente/notificari-discontinuitate-medicamente/ (accessed on 10 January 2026).
- Agencia Española de Medicamentos y Productos Sanitarios (AEMPS) Spain National Register of Shortages. Available online: https://www.aemps.gob.es/profesional-sanitario-2/problemas-de-suministro-de-medicamentos/ (accessed on 8 January 2026).
- U.S. FOOD & DRUG ADMINISTRATION Current and Resolved Drug Shortages and Discontinuations Reported to FDA. Available online: https://www.accessdata.fda.gov/scripts/drugshortages/default.cfm (accessed on 9 January 2026).
- Authority, S.F. and D. Saudi Food and Drug Authority – Currently in Shortage List. Available online: https://www.sfda.gov.sa/en/currentlyInShortageList (accessed on 9 April 2026).
- Methodology, W.C.C. for D.S. ATC/DDD Index: J01 Antibacterials for Systemic Use. Available online: https://atcddd.fhi.no/atc_ddd_index/?code=J01&showdescription=no (accessed on 23 February 2026).
- JASP Team JASP (Version 0.95.4) [Computer Software]. 2025.
- Microsoft Corporation, Redmond, WA, U. Microsoft Corporation (2019). Microsoft Excel (Version 2019, Off. Prof. Plus).
| Multinational shortage | Ema critical status | Total | ||
|---|---|---|---|---|
| No | Yes | |||
| No | Count | 66.00 | 42.00 | 108.0 |
| % within column | 62.86 % 17.14% | 30.86 % | ||
| Yes | Count | 39.00 | 203.0 | 242.0 |
| % within column | 37.14 % | 82.86 % | 69.14 % | |
| Total | Count | 105.0 | 245.0 | 350.0 |
| % within column | 100.00 % | 100.00 % | 100.00 % | |
| EMA critical status | Country | Total | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Belgium | France | Germany | Romania | Saudi | Spain | USA/FDA | |||||||||
| No | Count | 13.00 | 7.00 | 13.00 | 10.00 | 47.00 | 10.00 | 5.00 | 105.0 | ||||||
| % within column | 29.55 % | 31.82 % | 31.71 % | 22.22 % | 32.87 % | 31.25 % | 21.74 % | 30.00 % | |||||||
| Yes | Count | 31.00 | 15.00 | 28.00 | 35.00 | 96.00 | 22.00 | 18.00 | 245.0 | ||||||
| % within column | 70.45 % | 68.18 % | 68.29 % | 77.78 % | 67.13 % | 68.75 % | 78.26 % | 70.00 % | |||||||
| Total | Count | 44.00 | 22.00 | 41.00 | 45.00 | 143.0 | 32.00 | 23.00 | 350.0 | ||||||
| % within column | 100.00 % | 100.00 % | 100.00 % | 100.00 % | 100.00 % | 100.00 % | 100.00 % | 100.00 % | |||||||
| Model | Odds Ratio | p | 95% Confidence interval (odds ratio scale) |
||
| Lower bound | Upper bound | ||||
| M0 | (Intercept) | 2.241 | 3.125×10-12 | 1.786 | 2.811 |
| M1 | (Intercept) | 0.653 | 6.098×10-2 | 0.419 | 1.020 |
| EMA critical status (1) | 8.294 | 1.425×10-15 | 4.934 | 13.941 | |
| Injectable route (1) | 0.779 | 3.410×10-1 | 0.465 | 1.303 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




