Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Study Population
2.2. Cryoballoon Ablation Procedure
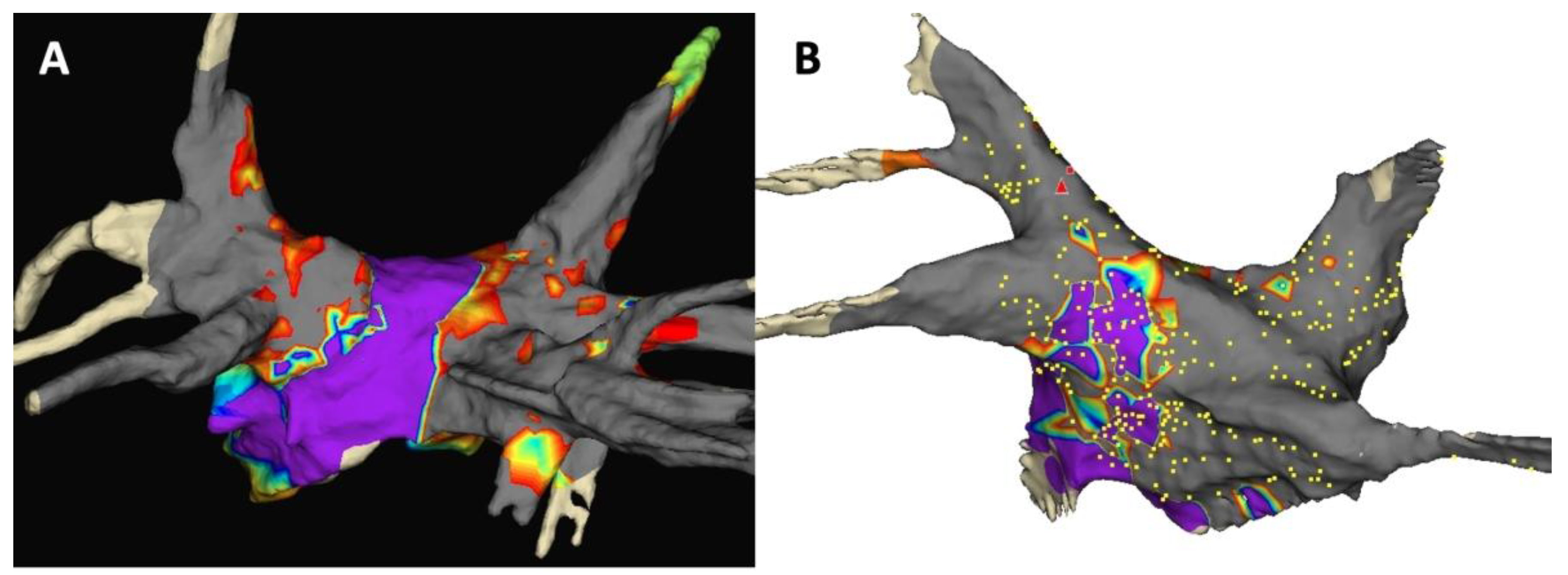
2.3. Perprocedural Electroanatomical Mapping
2.4. Post-Procedural Management and Follow-Up
2.5. Repeat Ablation Procedure
2.6. Statistical Analysis
3. Results
3.1. Clinical Characteristics
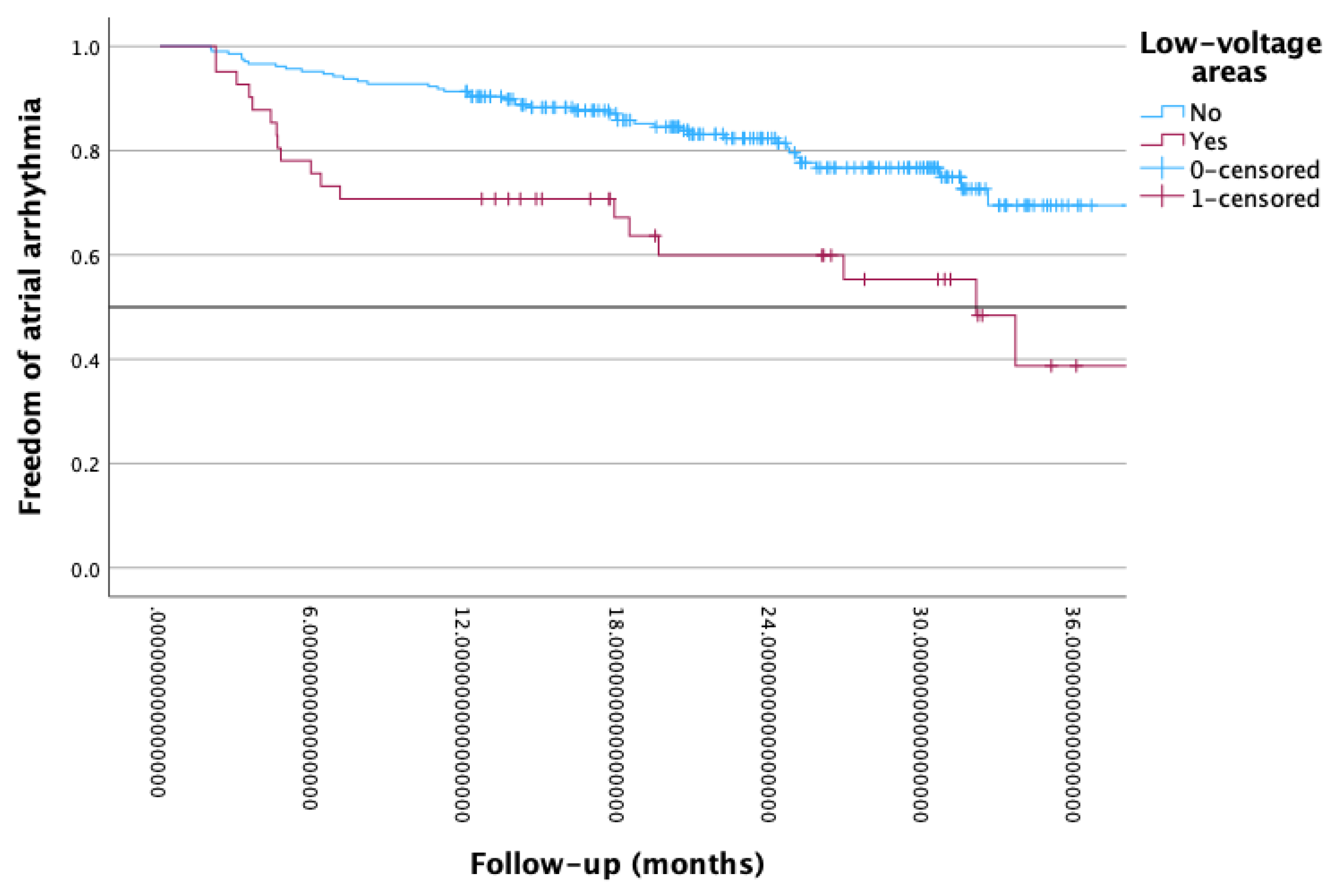
3.2. Clinical Outcome and Predictors
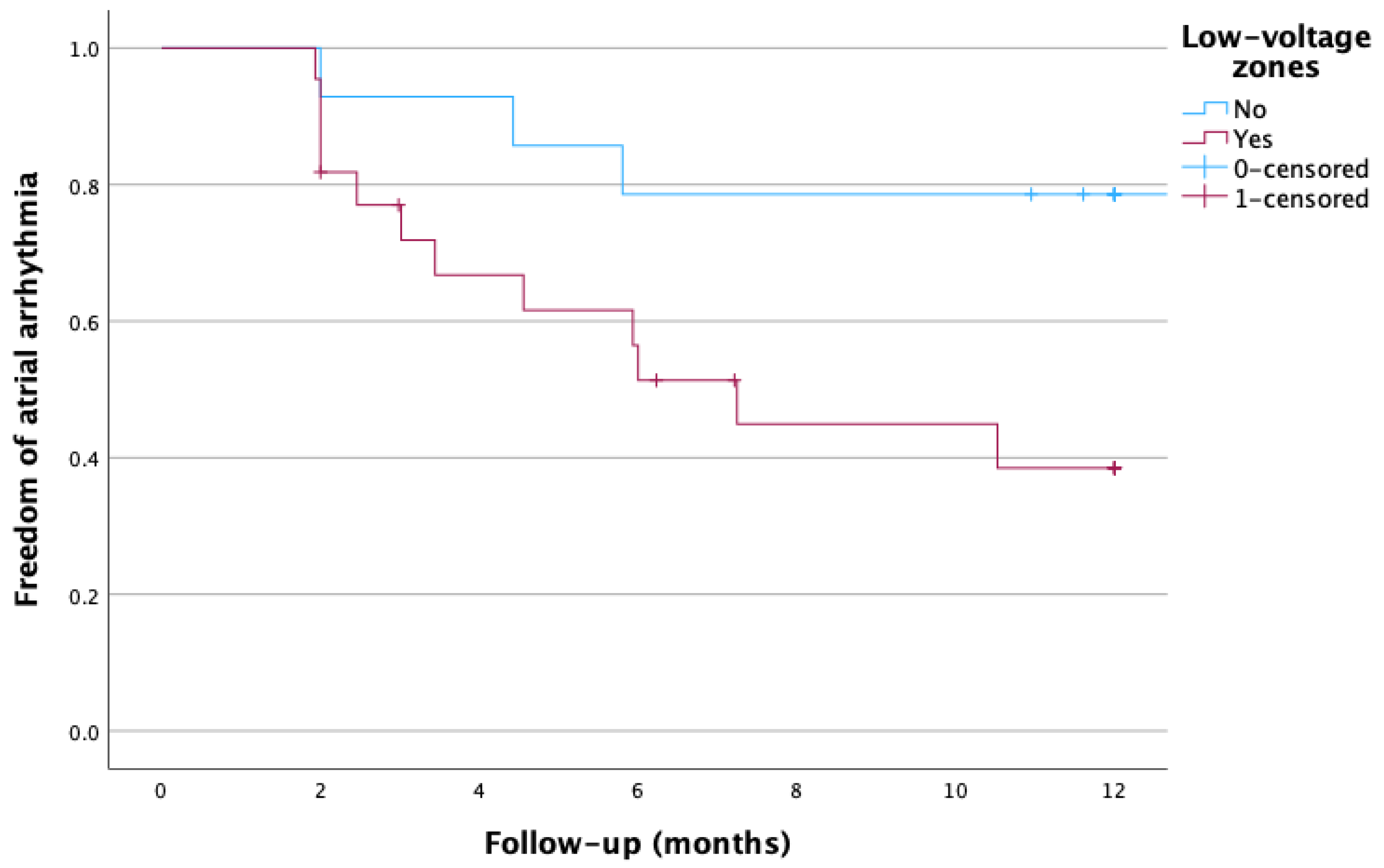
3.3. Findings During Repeat Procedures
4. Discussion
4.1. Main Findings
- 1)
- Patients with pwLVZ were older, more frequent female, had higher CHA2DS2-VASc scores and a higher incidence of obesity, structural heart disease and persistent AF.
- 2)
- Patients with pwLVZ had a worse outcome at 1 year after index ablation and the presence of pwLVZ was a predictor of recurrence.
- 3)
- During repeat procedure, patients with pwLVZ were more likely to undergo empirical substrate ablation. Patients with pwLVZ at index procedure had a worse outcome at 1 year after first repeat ablation.
4.2. Addition of Electroanatomical Mapping to Cryoballoon Ablation
4.3. Posterior Wall Low-Voltage Zones
4.4. Posterior Wall Low-Voltage Zones During Cryoballoon Ablation Predict Worse Clinical Outcome
4.5. Clinical Implications and Future Perspectives
4.6. Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allessie, M.; Ausma, J.; Schotten, U. Electrical, contractile and structural remodeling during atrial fibrillation. Cardiovasc. Res. 2002, 54, 230–246. [Google Scholar] [CrossRef] [PubMed]
- Bergonti, M.; Spera, F.R.; Ferrero, T.G.; Nsahlai, M.; Bonomi, A.; Tijskens, M.; Boris, W.; Saenen, J.; Huybrechts, W.; Miljoen, H.; et al. Characterization of Atrial Substrate to Predict the Success of Pulmonary Vein Isolation: The Prospective, Multicenter MASH-AF II (Multipolar Atrial Substrate High Density Mapping in Atrial Fibrillation) Study. J. Am. Hear. Assoc. 2023, 12. [Google Scholar] [CrossRef] [PubMed]
- Masuda, M.; Fujita, M.; Iida, O.; Okamoto, S.; Ishihara, T.; Nanto, K.; Kanda, T.; Tsujimura, T.; Matsuda, Y.; Okuno, S.; et al. Left atrial low-voltage areas predict atrial fibrillation recurrence after catheter ablation in patients with paroxysmal atrial fibrillation. Int. J. Cardiol. 2018, 257, 97–101. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Wazni, O.M.; Marrouche, N.F.; Martin, D.O.; Kilicaslan, F.; Minor, S.; Schweikert, R.A.; Saliba, W.; Cummings, J.; Burkhardt, J.D.; Bhargava, M.; Belden, W.A.; Abdul-Karim, A.; Natale, A. Pre-existent left atrial scarring in patients undergoing pulmonary vein antrum isolation: an independent predictor of procedural failure. J. Am. Coll. Cardiol. 2005, 45(2), 285–292. [Google Scholar] [CrossRef] [PubMed]
- Vlachos, K.; Efremidis, M.; Letsas, K.P.; Bazoukis, G.; Martin, R.; Kalafateli, M.; Lioni, L.; Georgopoulos, S.; Saplaouras, A.; Efremidis, T.; et al. Low-voltage areas detected by high-density electroanatomical mapping predict recurrence after ablation for paroxysmal atrial fibrillation. J. Cardiovasc. Electrophysiol. 2017, 28, 1393–1402. [Google Scholar] [CrossRef] [PubMed]
- Tijskens, M.; Abugattas, J.P.; Thoen, H.; Strazdas, A.; Schwagten, B.; Wolf, M.; De Greef, Y. Adding Electroanatomical Mapping to Cryoballoon Pulmonary Vein Isolation Improves 1-Year Clinical Outcome and Durability of Pulmonary Vein Isolation: A Propensity Score-Matched Analysis. J. Cardiovasc. Dev. Dis. 2024, 11, 57. [Google Scholar] [CrossRef] [PubMed]
- De Greef, Y.; Tijskens, M.; De Torres, J.P.A.; Sofianos, D.; Cecchini, F.; De Schouwer, K.; De Cocker, J.; Buysschaert, I.; Varnavas, V.; Wolf, M. Electroanatomical mapping improves procedural outcomes of cryoballoon pulmonary vein isolation (the Achieve Plus study). J. Interv. Card. Electrophysiol. 2022, 66, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Ströker, E.; De Greef, Y.; Schwagten, B.; Kupics, K.; Coutiño, H.E.; Takarada, K.; Abugattas, J.P.; Salghetti, F.; De Cocker, J.; Stockman, D.; et al. Over-the-needle trans-septal access using the cryoballoon delivery sheath and dilator in atrial fibrillation ablation. Pacing Clin. Electrophysiol. 2019, 42, 868–873. [Google Scholar] [CrossRef] [PubMed]
- Sanders, P.; Morton, J.B.; Kistler, P.M.; Davidson, N.C.; Hussin, A.; Spence, S.J.; Lindsay, C.B.; Vohra, J.K.; Sparks, P.B.; Kalman, J.M. Electrophysiologic and electroanatomic characterization of the atria in sinus node disease: Evidence of diffuse atrial remodeling. JACC 2003, 41, 121. [Google Scholar] [CrossRef]
- Tzeis, S.; Gerstenfeld, E.P.; Kalman, J.; Saad, E.B.; Sepehri Shamloo, A.; Andrade, J.G.; Barbhaiya, C.R.; Baykaner, T.; Boveda, S.; Calkins, H.; Chan, N.Y.; Chen, M.; Chen, S.A.; Dagres, N.; Damiano, R.J.; De Potter, T.; Deisenhofer, I.; Derval, N.; Di Biase, L.; Duytschaever, M.; Dyrda, K.; Hindricks, G.; Hocini, M.; Kim, Y.H.; la Meir, M.; Merino, J.L.; Michaud, G.F.; Natale, A.; Nault, I.; Nava, S.; Nitta, T.; O’Neill, M.; Pak, H.N.; Piccini, J.P.; Purerfellner, H.; Reichlin, T.; Saenz, L.C.; Sanders, P.; Schilling, R.; Schmidt, B.; Supple, G.E.; Thomas, K.L.; Tondo, C.; Verma, A.; Wan, E.Y.; Steven, D.; Agbayani, M.J.; Jared Bunch, T.; Chugh, A.; Diaz, J.C.; Freeman, J.V.; Hardy, C.A.; Heidbuchel, H.; Johar, S.; Linz, D.; Maesen, B.; Noseworthy, P.A.; Oh, S.; Porta-Sanchez, A.; Potpara, T.; Rodriguez-Diez, G.; Sacher, F.; Suwalski, P.; Trines, S.A. 2024 European Heart Rhythm Association/Heart Rhythm Society/Asia Pacific Heart Rhythm Society/Latin American Heart Rhythm Society expert consensus statement on catheter and surgical ablation of atrial fibrillation. Europace 2024, 26(4). [Google Scholar] [CrossRef] [PubMed]
- Tijskens, M.B.d.B.; Wolf, M.; Schwagten, B.; De Greef, Y. Cryoballoon Pulmonary Vein Isolation Facilitated by Integrated Electroanatomical Mapping for Paroxysmal Atrial Fibrillation in a Patient with Kartagener’s Syndrome. Case Rep. Clin. Cardiol. J. 2026, 6(1), 170. [Google Scholar]
- Huo, Y.; Gaspar, T.; Schönbauer, R.; Wójcik, M.; Fiedler, L.; Roithinger, F.X.; Martinek, M.; Pürerfellner, H.; Kirstein, B.; Richter, U.; et al. Low-Voltage Myocardium-Guided Ablation Trial of Persistent Atrial Fibrillation. NEJM Évid. 2022, 1. [Google Scholar] [CrossRef] [PubMed]
- Van Leuven, O.; Bergonti, M.; Spera, F.R.; Ferrero, T.G.; Nsahlai, M.; Bilotta, G.; Tijskens, M.; Boris, W.; Saenen, J.; Huybrechts, W.; Miljoen, H.; Gonzalez-Juanatey, J.R.; Martinez-Sande, J.L.; Vandaele, L.; Wittock, A.; Heidbuchel, H.; Valderrabano, M.; Rodriguez-Manero, M.; Sarkozy, A. Gender-Related Differences in Atrial Substrate in Patients with Atrial Fibrillation. Am. J. Cardiol. 2023, 203, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Tijskens, M.; Abugattas, J.P.; Wolf, M.; Schwagten, B.; De Greef, Y. Recurrence of Atrial Fibrillation After Cryoballoon Ablation in Women Is Less Pulmonary Vein Mediated as Compared to Men: Lessons From Repeat Ablation Procedures. J. Cardiovasc. Electrophysiol. 2025, 36, 1395–1405. [Google Scholar] [CrossRef] [PubMed]
- Schade, A.; Costello-Boerrigter, L.; Steinborn, F.; Bayri, A.H.; Chapran, M.; Surber, R.; Schulze, P.C.; Mattea, V. Voltage-guided ablation in persistent atrial fibrillation—favorable 1-year outcome and predictors. J. Interv. Card. Electrophysiol. 2020, 62, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Chieng, D.; Sugumar, H.; Hunt, A.; Ling, L.-H.; Segan, L.; Al-Kaisey, A.; Hawson, J.; Prabhu, S.; Voskoboinik, A.; Wong, G.; et al. Impact of Posterior Left Atrial Voltage on Ablation Outcomes in Persistent Atrial Fibrillation. JACC Clin. Electrophysiol. 2023, 9, 2291–2299. [Google Scholar] [CrossRef] [PubMed]
- Pranata, R.; Kamarullah, W.; Karwiky, G.; Achmad, C.; Iqbal, M. Left atrial posterior wall isolation in addition to pulmonary vein isolation using a pentaspline catheter in pulsed-field ablation for atrial fibrillation: A systematic review and meta-analysis. Hear. Rhythm. O2 2024, 5, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.A.; Elaraby, A.; Almaadawy, O.; Abuelazm, M.; Hassan, A.R.; Bakr, A.; Husain, M.A.; Elsayed, H.; Abdelazeem, B. Adjunctive left atrial posterior wall isolation for atrial fibrillation: An updated systematic review and meta-analysis. Pacing Clin. Electrophysiol. 2024, 47, 1108–1123. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Geng, Y.; Liu, Y.; Lv, T.; Zhang, P. The Value of Adjunctive Left Atrial Posterior Wall Isolation on Clinical Outcomes in Atrial Fibrillation Patients: A Systematic Review and Meta-Analysis. Rev. Cardiovasc. Med. 2024, 25, 210. [Google Scholar] [CrossRef] [PubMed]
| No pwLVZ (N=209) | pwLVZ (N=41) | P-value | |
|---|---|---|---|
| Age | 64.2±10.4 | 69.3±8.5 | 0.003 |
| Female, n (%) | 68 (32.5%) | 26/41 (63.4%) | <0.001 |
| Obesitas | 54 (25.8%) | 13/41 (31.7%) | 0.048 |
| Arterial hypertension | 109 (52.4%) | 23/41 (56.1%) | 0.665 |
| Diabetes | 20/209 (9.7%) | 6/41 (14.6%) | 0.348 |
| History of stroke | 17/209 (8.2%) | 3/41 (7.3%) | 0.847 |
| CHADS-VASc score | 2.0±1.5 | 3.0±1.6 | <0.001 |
| Sleep apnea | 16/209 (7.7%) | 2/41 (4.9%) | 0.525 |
| Structural heart disease | 29/209 (17.4%) | 11/41 (35.5%) | 0.021 |
| Left atrial diameter (mm) | 38.9±7.5 | 40.6±7.1 | 0.260 |
| Diagnosis-to-ablation time (months) | 28.3±42.1 | 16.5±25.8 | 0.086 |
| Persistent AF | 91/209 (43.8%) | 28/41 (68.3%) | 0.004 |
| Univariate analysis | Multivariate analysis | |||||
|---|---|---|---|---|---|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value | |
| Persistent AF | 1.288 | 0.774 – 2.144 | 0.331 | |||
| Diagnosis-to-ablation time | 1.004 | 0.998 – 1.009 | 0.158 | |||
| CHADS-VASc score | 1.258 | 1.084 – 1.461 | 0.003 | 1.160 | 0.973 – 1.382 | 0.098 |
| Obesity | 0.862 | 0.480 – 1.547 | 0.618 | |||
| Left atrial diameter | 1.063 | 1.023 – 1.105 | 0.002 | 1.054 | 1.014 – 1.097 | 0.008 |
| Structural heart disease | 1.257 | 0.637 – 2.483 | 0.510 | |||
| Posterior wall LVZ | 2.520 | 1.448 – 4.386 | 0.001 | 2.583 | 1.334 – 5.002 | 0.005 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




