Submitted:
28 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
Aim
2. Materials and Methods
Inclusion Criteria
Exclusion Criteria
Blood Sample Collection and Storage
Total Oxidant Status (TOS) Measurement
Total Antioxidant Status (TAS) Measurement
Oxidative Stress Index (OSI) Calculation
Assessment of DNA Damage by the Comet Assay
Urinary 8-Hydroxy-2′-Deoxyguanosine (8-OHdG) Measurement
Statistical Analysis
3. Results
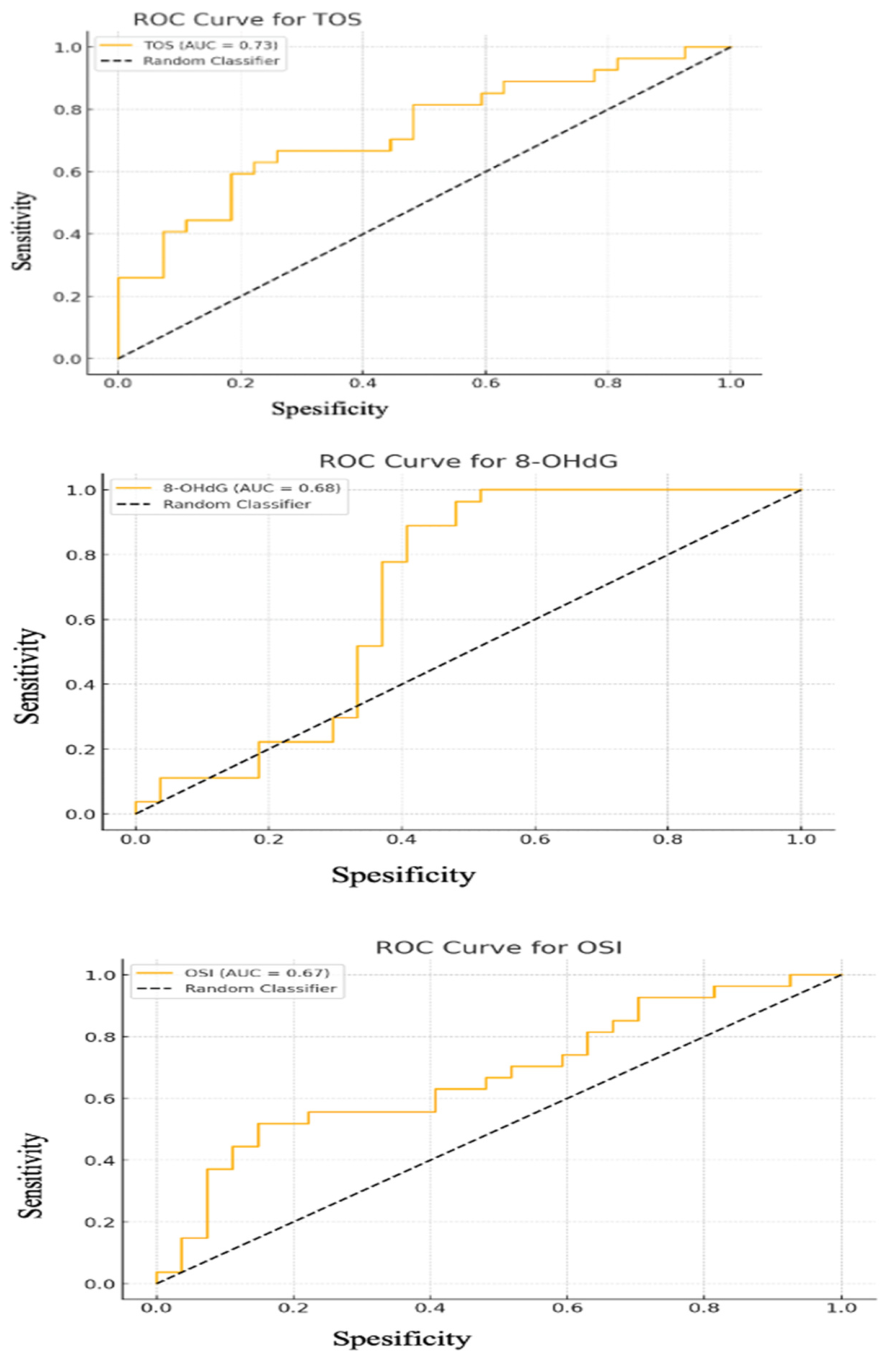
ROC Analysis and Correlations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgements
Conflict of Interest
Abbreviations
| IGM | Idiopathic granulomatous mastitis |
| TOS | Total oxidant status |
| TAS | Total antioxidant status |
| OSI | Oxidative stress index |
| 8-OhdG | 8-hydroxy-2′-deoxyguanosine |
| DNA | Deoxyribonucleic acid |
| ROS | Reactive oxygen species |
| ROC | Receiver operating characteristic |
| ELISA | Enzyme-linked immunosorbent assay |
| WBC | White blood cell |
| NLR | Neutrophil-to-lymphocyte ratio |
| Hb | Hemoglobin |
| Hct | Hematocrit |
| NK | Natural killer cell |
| TREM-1 | Triggering receptor expressed on myeloid cells-1 |
| SLE | Systemic lupus erythematosus |
References
- Kessler, E.; Wolloch, Y. Granulomatous Mastitis: A Lesion Clinically Simulating Carcinoma. Am. J. Clin. Pathol. 1972, 58, 642–646. [CrossRef]
- Altintoprak, F.; Karakece, E.; Kivilcim, T.; Dikicier, E.; Cakmak, G.; Celebi, F.; Ciftci, I.H. Idiopathic Granulomatous Mastitis: An Autoimmune Disease?. Sci. World J. 2013, 2013, 148727. [CrossRef]
- D. Y. Kehribar, T. I. Duran, A. K. Polat, and M. Ozgen,“Efectiveness of methotrexate in idiopathic granulomatousmastitis treatment,” Te American Journal of the MedicalSciences, vol. 360, no. 5, pp. 560–565, 2020.
- Cohen C. Granulomatous mastitis. A review of 5 cases. S Afr Med J. 1977 Jul 2;52(1):14–6.
- Altintoprak, F.; Kivilcim, T.; Ozkan, O.V. Aetiology of idiopathic granulomatous mastitis. World J. Clin. Cases 2014, 2, 852–8. [CrossRef]
- Al-Khaffaf, B.; Knox, F.; Bundred, N.J. Idiopathic Granulomatous Mastitis: A 25-Year Experience. J. Am. Coll. Surg. 2008, 206, 269–273. [CrossRef]
- Benson JR, Dumitru D. Idiopathic granulomatous mastitis: Presentation, investigation and management. Vol. 12, Future Oncology. Future Medicine Ltd.; 2016. p. 1381–94.
- Heer, R.; Shrimankar, J.; Griffith, C. Granulomatous mastitis can mimic breast cancer on clinical, radiological or cytological examination: a cautionary tale. Breast 2003, 12, 283–286. [CrossRef]
- Zhang, M.; Pu, D.; Feng, D.; Shi, G.; Li, J. Rare and Complicated Granulomatous Lobular Mastitis (2000–2023): A Bibliometrics Study and Visualization Analysis. J. Inflamm. Res. 2024, ume 17, 3709–3724. [CrossRef]
- Sarmadian, R.; Safi, F.; Sarmadian, H.; Shokrpour, M.; Almasi-Hashiani, A. Treatment modalities for granulomatous mastitis, seeking the most appropriate treatment with the least recurrence rate: a systematic review and meta-analysis. Eur. J. Med Res. 2024, 29, 1–21. [CrossRef]
- Velidedeoglu, M.; Kilic, F.; Mete, B.; Yemisen, M.; Celik, V.; Gazioglu, E.; Ferahman, M.; Ozaras, R.; Yilmaz, M.H.; Aydogan, F. Bilateral idiopathic granulomatous mastitis. Asian J. Surg. 2016, 39, 12–20. [CrossRef]
- Basu, A.K. DNA Damage, Mutagenesis and Cancer. Int. J. Mol. Sci. 2018, 19, 970. [CrossRef]
- Altindag, O.; Karakoc, M.; Kocyigit, A.; Celik, H.; Soran, N. Increased DNA damage and oxidative stress in patients with rheumatoid arthritis. Clin. Biochem. 2007, 40, 167–171. [CrossRef]
- Kocyigit, A.; Keles, H.; Selek, S.; Guzel, S.; Celik, H.; Erel, O. Increased DNA damage and oxidative stress in patients with cutaneous leishmaniasis. Mutat. Res. Toxicol. Environ. Mutagen. 2005, 585, 71–78. [CrossRef]
- Dizdaroglu, M. Oxidatively induced DNA damage: Mechanisms, repair and disease. Cancer Lett. 2012, 327, 26–47. [CrossRef]
- Collins, A.R. The Comet Assay for DNA Damage and Repair: Principles, Applications, and Limitations. Mol. Biotechnol. 2004, 26, 249–261. [CrossRef]
- Valavanidis, A.; Vlachogianni, T.; Fiotakis, C. 8-hydroxy-2′ -deoxyguanosine (8-OHdG): A Critical Biomarker of Oxidative Stress and Carcinogenesis. J. Environ. Sci. Heal. Part C 2009, 27, 120–139. [CrossRef]
- Velidedeoglu, M.; Kundaktepe, B.P.; Aksan, H.; Uzun, H. Preoperative Fibrinogen and Hematological Indexes in the Differential Diagnosis of Idiopathic Granulomatous Mastitis and Breast Cancer. Medicina 2021, 57, 698. [CrossRef]
- Emsen, A.; Köksal, H.; Uçaryilmaz, H.; Kadoglou, N.; Artaç, H. The alteration of lymphocyte subsets in idiopathic granulomatous mastitis. Turk. J. Med Sci. 2021, 51, 1905–1911. [CrossRef]
- Velidedeoglu M, Umman V, Kilic F, Celik V, Gazioglu E, Hatipoglu E, et al. Idiopathic granulomatous mastitis: introducing a diagnostic algorithm based on 5 years of follow-up of 152 cases from Turkey and a review of the literature. Surg Today. 2022 Apr 21;52(4):668–80.
- Martinez-Ramos, D.; Simon-Monterde, L.; Suelves-Piqueres, C.; Queralt-Martin, R.; Granel-Villach, L.; Laguna-Sastre, J.M.; Nicolau, M.J.; Escrig-Sos, J. Idiopathic granulomatous mastitis: A systematic review of 3060 patients. Breast J. 2019, 25, 1245–1250. [CrossRef]
- Dogan, S.; Dal, F.; Guler, M.; Sevik, H.; Idiz, U.O. Is peripheral blood immunophenotyping useful to understand the etiology of Idiopathic Granulomatous?. Hum. Immunol. 2023, 84, 315–319. [CrossRef]
- Baslaim, M.M.; Khayat, H.A.; Al-Amoudi, S.A. Idiopathic Granulomatous Mastitis: A Heterogeneous Disease with Variable Clinical Presentation. World J. Surg. 2007, 31, 1677–1681. [CrossRef]
- Rowe, P.H. Granulomatous Mastitis Associated with a Pituitary Prolactinoma. Int. J. Clin. Pr. 1984, 38, 32–34. [CrossRef]
- Çetinkaya, G.; Kozan, R.; Emral, A.C.; Tezel, E. Granulomatous mastitis, watch and wait is a good option. Ir. J. Med Sci. (1971 -) 2020, 190, 1117–1122. [CrossRef]
- Fattahi, A.S.; Amini, G.; Sajedi, F.; Mehrad-Majd, H. Factors Affecting Recurrence of Idiopathic Granulomatous Mastitis: A Systematic Review. Breast J. 2023, 2023, 1–12. [CrossRef]
- Erozgen, F.; Ersoy, Y.E.; Akaydin, M.; Memmi, N.; Celik, A.S.; Celebi, F.; Guzey, D.; Kaplan, R. Corticosteroid treatment and timing of surgery in idiopathic granulomatous mastitis confusing with breast carcinoma. Breast Cancer Res. Treat. 2010, 123, 447–452. [CrossRef]
- Mizrakli, T.; Velidedeoglu, M.; Yemisen, M.; Mete, B.; Kilic, F.; Yilmaz, H.; Ozturk, T.; Ozaras, R.; Aydogan, F.; Perek, A. Corticosteroid treatment in the management of idiopathic granulomatous mastitis to avoid unnecessary surgery. Surg. Today 2014, 45, 457–465. [CrossRef]
- Toktas, O.; Konca, C.; Trabulus, D.C.; Soyder, A.; Koksal, H.; Karanlik, H.; Polat, A.K.; Ozbas, S.; Yormaz, S.; Isik, A.; et al. A Novel First-Line Treatment Alternative for Noncomplicated Idiopathic Granulomatous Mastitis: Combined İntralesional Steroid İnjection with Topical Steroid Administration. Breast Care 2020, 16, 181–187. [CrossRef]
- Papila Kundaktepe B, Velidedeoğlu M, Mete B. The effect of methotrexate monotherapy on treatment-resistant idiopathic granulomatous mastitis patients. The Surgeon. 2022 Jun;20(3):e13–9.
- Zeng, Y.; Zhang, D.; Fu, N.; Zhao, W.; Huang, Q.; Cui, J.; Chen, Y.; Liu, Z.; Zhang, X.; Zhang, S.; et al. Risk Factors for Granulomatous Mastitis and Establishment and Validation of a Clinical Prediction Model (Nomogram). Risk Manag. Heal. Policy 2023, ume 16, 2209–2222. [CrossRef]
- Afeiche, M.C.; Iroz, A.; Thielecke, F.; De Castro, A.C.; Lefebvre, G.; Draper, C.F.; Martínez-Costa, C.; Haaland, K.; Marchini, G.; Agosti, M.; et al. The Dietary Inflammatory Index Is Associated with Subclinical Mastitis in Lactating European Women. Nutrients 2022, 14, 4719. [CrossRef]
- Ramos-Lopez, O.; Martinez-Urbistondo, D.; Vargas-Nuñez, J.A.; Martinez, J.A. The Role of Nutrition on Meta-inflammation: Insights and Potential Targets in Communicable and Chronic Disease Management. Curr. Obes. Rep. 2022, 11, 305–335. [CrossRef]
- Wang, X.; He, X.; Liu, J.; Zhang, H.; Wan, H.; Luo, J.; Yang, J. Immune pathogenesis of idiopathic granulomatous mastitis: from etiology toward therapeutic approaches. Front. Immunol. 2024, 15, 1295759. [CrossRef]
- Koksal, H.; Vatansev, H.; Artac, H.; Kadoglou, N. The clinical value of interleukins-8, -10, and -17 in idiopathic granulomatous mastitis. Clin. Rheumatol. 2020, 39, 1671–1677. [CrossRef]
- Ates, D.; Doner, H.C.; Kurban, S.; Koksal, H. The Effect of Soluble TREM-1 in Idiopathic Granulomatous Mastitis. Immunol. Investig. 2021, 51, 839–850. [CrossRef]
- Cetinkaya ÖA, Çelik SU, Terzioğlu SG, Eroğlu A. The Predictive Value of the Neutrophil-to-Lymphocyte and Platelet-to-Lymphocyte Ratio in Patients with Recurrent Idiopathic Granulomatous Mastitis. Eur J Breast Health. 2020 Jan;16(1):61–5.
- Zhu, Q.; Wang, L.; Wang, P. The Identification of Gene Expression Profiles Associated with Granulomatous Mastitis. Breast Care 2020, 16, 319–327. [CrossRef]
- Ozcinar, B.; Ocak, Z.; Billur, D.; Ertugrul, B.; Timirci-Kahraman, O. Whole-Exome Sequencing: Discovering Genetic Causes of Granulomatous Mastitis. Int. J. Mol. Sci. 2025, 26, 425. [CrossRef]
- Kurien, B.T.; Hensley, K.; Bachmann, M.; Scofield, R.H. Oxidatively modified autoantigens in autoimmune diseases. Free. Radic. Biol. Med. 2006, 41, 549–556. [CrossRef]
- Buczyńska A, Sidorkiewicz I, Wiatr A, Adamska A, Siewko K, Krętowski A, et al. The role of oxidative stress in Hashimoto’s disease. Endocrine Abstracts. 2023 May 2;
- da Silva, G.B.; Yamauchi, M.A.; Bagatini, M.D. Oxidative stress in Hashimoto’s thyroiditis: possible adjuvant therapies to attenuate deleterious effects. Mol. Cell. Biochem. 2022, 478, 949–966. [CrossRef]
- Muro, P.; Zhang, L.; Li, S.; Zhao, Z.; Jin, T.; Mao, F.; Mao, Z. The emerging role of oxidative stress in inflammatory bowel disease. Front. Endocrinol. 2024, 15, 1390351. [CrossRef]
- Aslan M, Nazligul Y, Bolukbas C, Bolukbas FF, Horoz M, Dulger AC, et al. Peripheral lymphocyte DNA damage and oxidative stress in patients with ulcerative colitis. Pol Arch Intern Med. 2011 Jul 1;121(7–8):223–9.
- Kundu, S.; Ghosh, P.; Datta, S.; Ghosh, A.; Chattopadhyay, S.; Chatterjee, M. Oxidative stress as a potential biomarker for determining disease activity in patients with Rheumatoid Arthritis. Free. Radic. Res. 2012, 46, 1482–1489. [CrossRef]
- Souliotis, V.L.; Sfikakis, P.P. Increased DNA double-strand breaks and enhanced apoptosis in patients with lupus nephritis. Lupus 2014, 24, 804–815. [CrossRef]
| Control group (n=27) | Patient Group (n=28) | p | |||||||||
| Mean±SD/n-% | Median | Mean±SD/n-% | Median | ||||||||
| Age | 35.4 | ± | 8.6 | 32.0 | 37.3 | ± | 5.3 | 37.5 | 0.081 | m | |
| Age at menarche | 13.7 | ± | 1.4 | 14.0 | 12.8 | ± | 1.1 | 13.0 | 0.006 | m | |
| Parity | (-) | 9 | 33.3% | 4 | 14.3% | 0.179 | X² | ||||
| (+) | 18 | 66.7% | 24 | 85.7% | |||||||
| Age at first birth | 23.8 | ± | 5.5 | 21.0 | 23.5 | ± | 4.6 | 23.5 | 0.901 | m | |
| Number of births | 1.8 | ± | 0.7 | 2.0 | 2.6 | ± | 1.1 | 2.0 | 0.055 | m | |
| Spontaneous abortion | (-) | 26 | 96.3% | 21 | 75.0% | 0.025 | X² | ||||
| I | 1 | 3.7% | 6 | 21.4% | |||||||
| II | 0 | 0.0% | 1 | 3.6% | |||||||
| Induced abortion | (-) | 25 | 92.6% | 23 | 82.1% | 0.245 | X² | ||||
| I | 1 | 3.7% | 4 | 14.3% | |||||||
| II | 1 | 3.7% | 1 | 3.6% | |||||||
| History of breastfeeding | (-) (+) |
9 18 |
33.3% 66.7% |
4 24 |
14.3% 85.7% |
0.179 | X² | ||||
| Presence of autoimmune disease | (-) (+) |
20 7 |
74.1% 25.9% |
22 6 |
78.6% 21.4% |
0.695 | X² | ||||
| Family history of breast cancer | (-) (+) |
24 3 |
88.9% 11.1% |
25 3 |
89.3% 10.7% |
0.962 | X² | ||||
|
Menopausal status Premenopausal Postmenopasal |
24 3 |
88.9% 11.1% |
280 | 100.0% 0.0% |
0.111 | X² | |||||
| Smoking status | (-) (+) |
14 13 |
51.9% 48.1% |
18 10 |
64.3% 35.7% |
0.350 | X² | ||||
| Oral contraceptive use | (-) (+) |
26 1 |
96.3% 3.7% |
26 2 |
92.9% 7.1% |
1.000 | X² | ||||
| Number of patient | n-% | ||
| History of abscess drainage | (-) | 7 | 25.0% |
| (+) | 21 | 75.0% | |
| Presence of axillary lymphadenopathy | (-) | 7 | 25.0% |
| (+) | 21 | 75.0% | |
| Presence of breast fistula | (-) | 13 | 46.4% |
| (+) | 15 | 53.6% | |
| Prior antibiotic use | (-) | 4 | 14.3% |
| (+) | 24 | 85.7% | |
| History of systemic steroid use | (-) | 16 | 57.1% |
| (+) | 12 | 42.9% | |
| History of topical steroid use | (-) | 18 | 64.3% |
| (+) | 10 | 35.7% | |
| Biopsy method | Core needle biopsy | 22 | 78.6% |
| Excisional biopsy | 5 | 17.8% | |
| Fine-needle aspiration biopsy (FNAB) | 1 | 3.6% |
| Control Group (n=27) | Patient Group (n=28) | p | |||||||||
| Mean±SD | Median | Mean±SD | Median | ||||||||
| WBC (×10⁹/L) | 7.5 | ± | 1.1 | 7.8 | 8.7 | ± | 1.4 | 8.61 | 0.002 | t | |
| Neutrophil (×10⁹/L) | 3.9 | ± | 1.1 | 3.8 | 6.5 | ± | 1.4 | 6.3 | 0.000 | t | |
| Lymphocyte (×10⁹/L) | 1.8 | ± | 0.4 | 1.8 | 2.2 | ± | 0.7 | 2.2 | 0.027 | m | |
| NLR | 2.04 | ± | 0.57 | 2.06 | 3.02 | ± | 1.1 | 3.05 | 0.007 | m | |
| HB (g/dL) | 13.5 | ± | 1.0 | 13.6 | 12.5 | ± | 1.2 | 12.6 | 0.003 | t | |
| HCT (%) | 43.3 | ± | 2.8 | 44.4 | 40.4 | ± | 4.2 | 41.1 | 0.008 | m | |
| PLT (×10⁹/L) | 301.7 | ± | 44.4 | 312.0 | 291.0 | ± | 47.4 | 276.0 | 0.395 | t | |
| MCV (fL) | 88.4 | ± | 2.8 | 88.6 | 88.2 | ± | 2.8 | 87.9 | 0.844 | t | |
| MCH (pg) | 30.0 | ± | 1.6 | 29.9 | 29.9 | ± | 1.6 | 29.7 | 0.730 | t | |
| MCHC (g/dL) | 33.6 | ± | 1.3 | 33.8 | 33.6 | ± | 1.2 | 33.7 | 0.821 | t | |
| Urea (mg/dL) | 31.0 | ± | 5.5 | 29.7 | 47.1 | ± | 6.0 | 44.4 | 0.000 | m | |
| Creatinine (mg/dL) | 0.78 | ± | 0.20 | 0.74 | 1.14 | ± | 0.32 | 1.28 | 0.000 | m | |
| AST (U/L) | 20.1 | ± | 6.1 | 19.0 | 21.5 | ± | 6.4 | 21.5 | 0.418 | m | |
| ALT (U/L) | 29.9 | ± | 10.7 | 31.0 | 30.0 | ± | 11.1 | 29.5 | 0.970 | t | |
| LDH (U/L) | 157.5 | ± | 35.9 | 149.0 | 274.8 | ± | 35.6 | 267.0 | 0.000 | t | |
| Serum albumin (g/dL) | 3.9 | ± | 0.5 | 3.9 | 3.9 | ± | 0.6 | 3.9 | 0.993 | t | |
| Serum sodium (mmol/L) | 139.4 | ± | 3.8 | 139.0 | 138.9 | ± | 3.6 | 139.0 | 0.563 | t | |
| Serum potassium (mmol/L) | 3.8 | ± | 0.3 | 3.7 | 4.9 | ± | 0.5 | 4.9 | 0.000 | m | |
| TOS (µmol H2O2 Equiv./L) | 1.04 | ± | 0.71 | 0.81 | 2.39 | ± | 2.27 | 1.43 | 0.009 | m | |
| TAS (mmol Trolox Equiv./L) | 0.85 | ± | 0.32 | 0.91 | 0.89 | ± | 0.22 | 0.90 | 0.534 | t | |
| Urine 8-OHdG (ng/ml) | 23.62 | ± | 19.2 | 25.63 | 36.3 | ± | 8.5 | 35.0 | 0.024 | m | |
| OSI(arbitrary unit,AU) | 155.2 | ± | 168.2 | 116.7 | 284.7 | ± | 296.5 | 178.3 | 0.028 | m | |
| Min-Max | Median | Mean±SD | |||||||||||
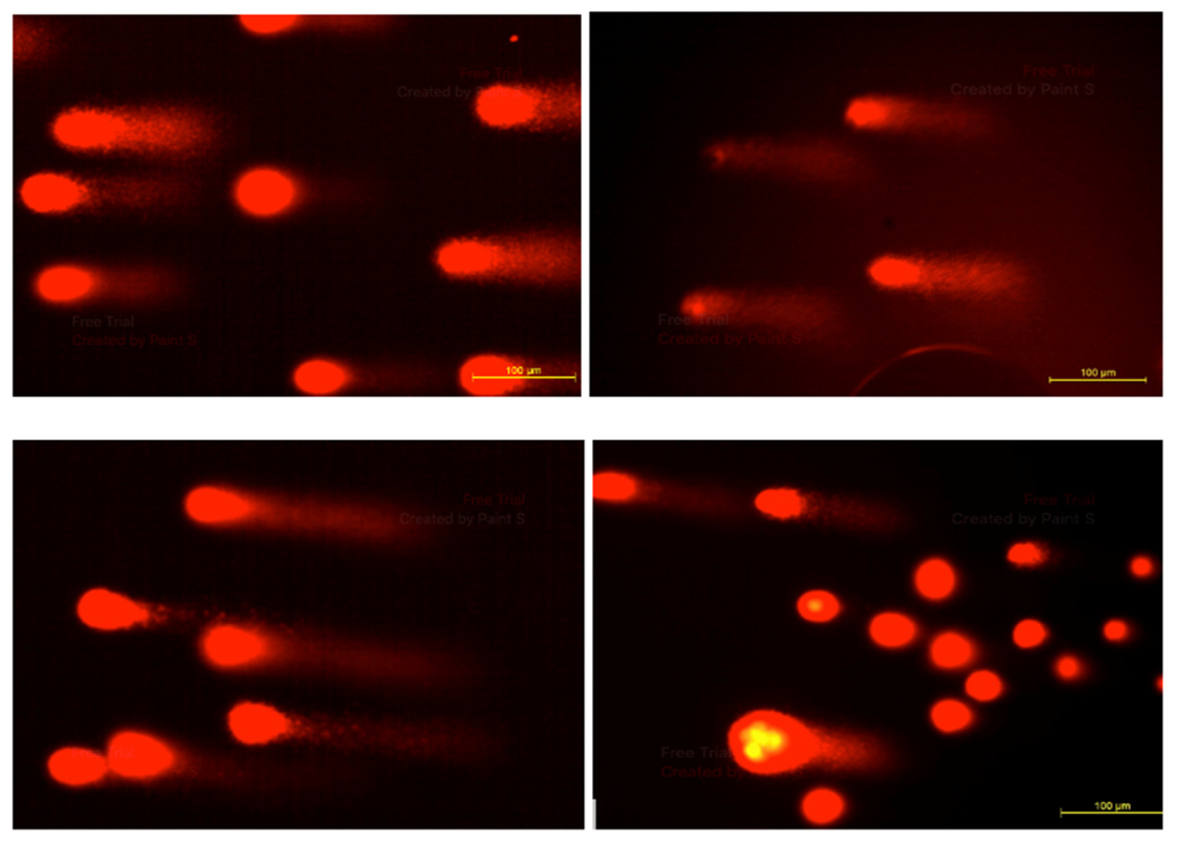
| Tail İntensity % | 0.003 | - | 16.58 | 3.45 | 4.84 | ± | 5.0 | ||||||
| Tail Moment | 0.0001 | - | 51.74 | 16.51 | 17.16 | ± | 14.7 | ||||||
| Control Group (n=15) | Patient Group (n=14) | p | |||||||||||
| Mean±SD | Median | Mean±SD | Median | ||||||||||
| Tail İntensity % | 3.15 | ± | 4.1 | 1.80 | 6.78 | ± | 5.3 | 5.26 | 0.029 | m | |||
| Tail Moment | 10.8 | ± | 11.9 | 7.71 | 24.0 | ± | 15.0 | 22.29 | 0.016 | m | |||
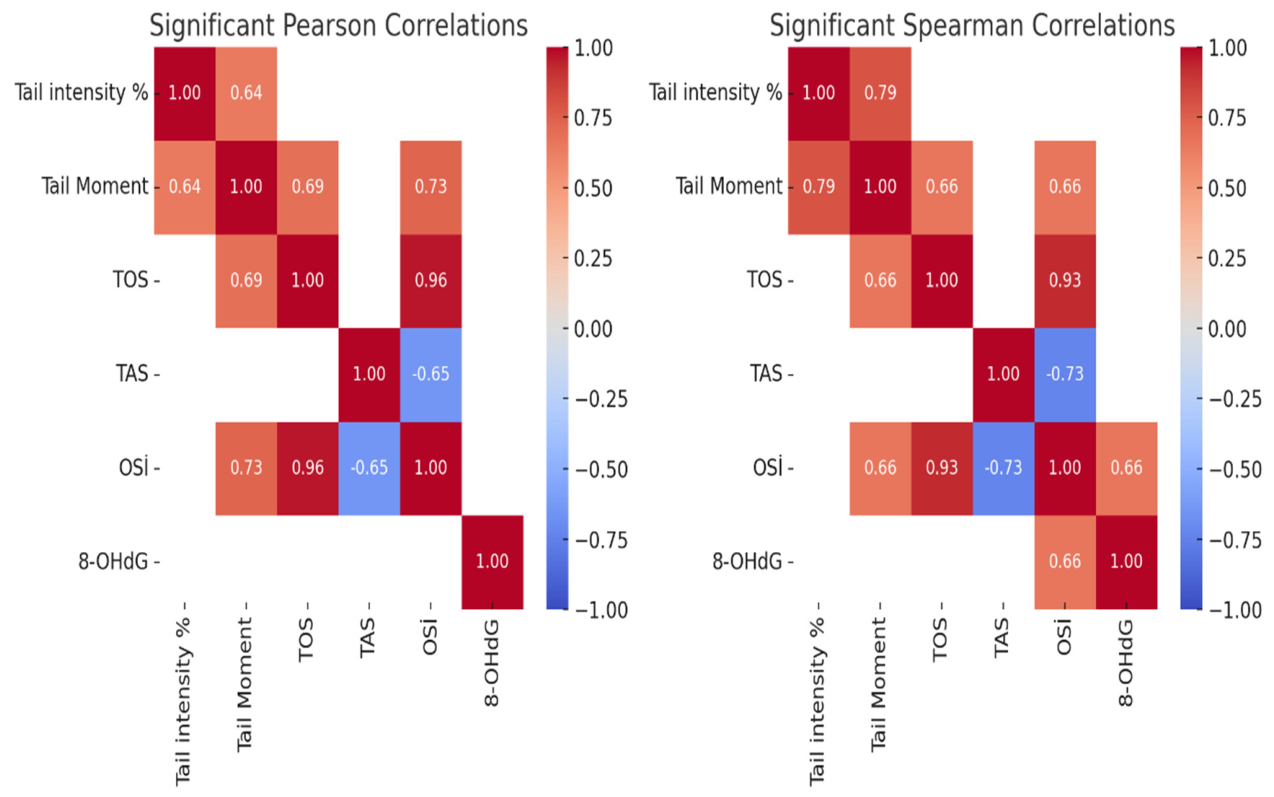
| Tail İntensity % | Tail Moment | TOS | TAS | OSİ | ||
| Tail Moment | r | 0.64 | ||||
| p | 0.0007 | |||||
| TOS | r | 0.729 | 0.69 | |||
| p | 0.000 | 0.000 | ||||
| TAS | r | -0.511 | -0.528 | -0.529 | ||
| p | 0.005 | 0.003 | 0.003 | |||
| OSİ | r | 0.735 | 0.734 | 0.961 | -0.727 | |
| p | 0.000 | 0.002 | 0.001 | 0.001 | ||
| 8-OHDG | r | 0.449 | 0.556 | 0.664 | -0.522 | 0.662 |
| p | 0.015 | 0.002 | 0.000 | 0.004 | 0.000 | |
| Pearson-Spearman Correlation | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




