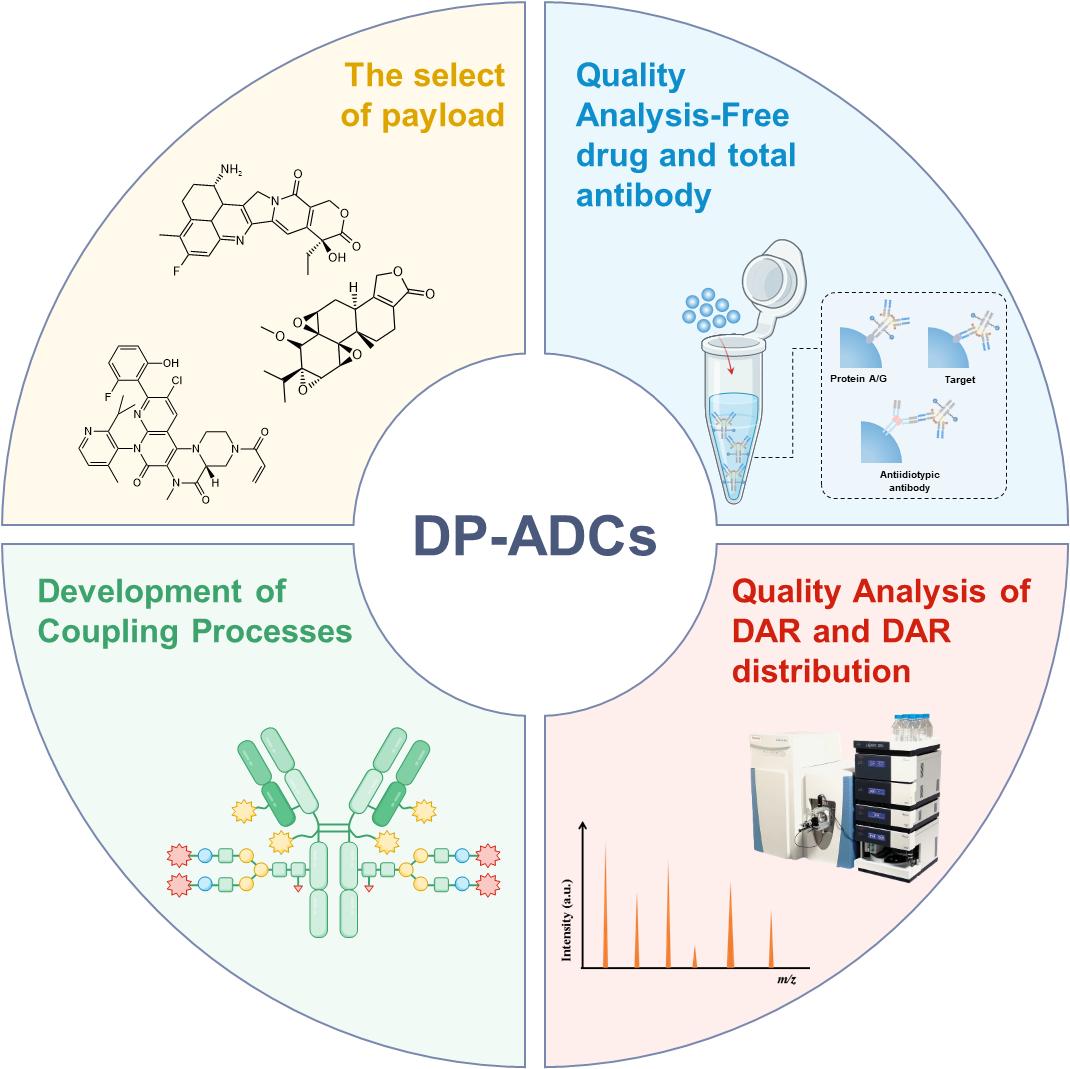
Antibody–drug conjugates (ADCs) are a pivotal technology for precision cancer therapy, harnessing the synergistic effects of antibody targeting and toxin delivery. However, traditional ADCs encounter limitations in efficacy that stem from tumor resistance, heterogeneity, and intense target competition. Dual-payload ADCs (DP-ADCs) represent a promising solution to these challenges, as they leverage dual mechanisms of action that mitigate acquired drug resistance and enhance adaptability to tumor heterogeneity. The complex structure of DP-ADCs presents substantial quality control hurdles. In this manuscript, we review the current payload selection and conjugation strategies of DP-ADCs and examine recent advances in quality control research. Specifically, we analyze the analytical challenges related to the quantification of free toxins, the determination of the total antibody content, and the characterization of the drug-to-antibody ratio and its distribution. Ultimately, the aim of this work is to provide valuable guidance for future DP-ADC quality control analyses to facilitate their clinical translation and application.