Submitted:
29 April 2026
Posted:
29 April 2026
You are already at the latest version
Abstract
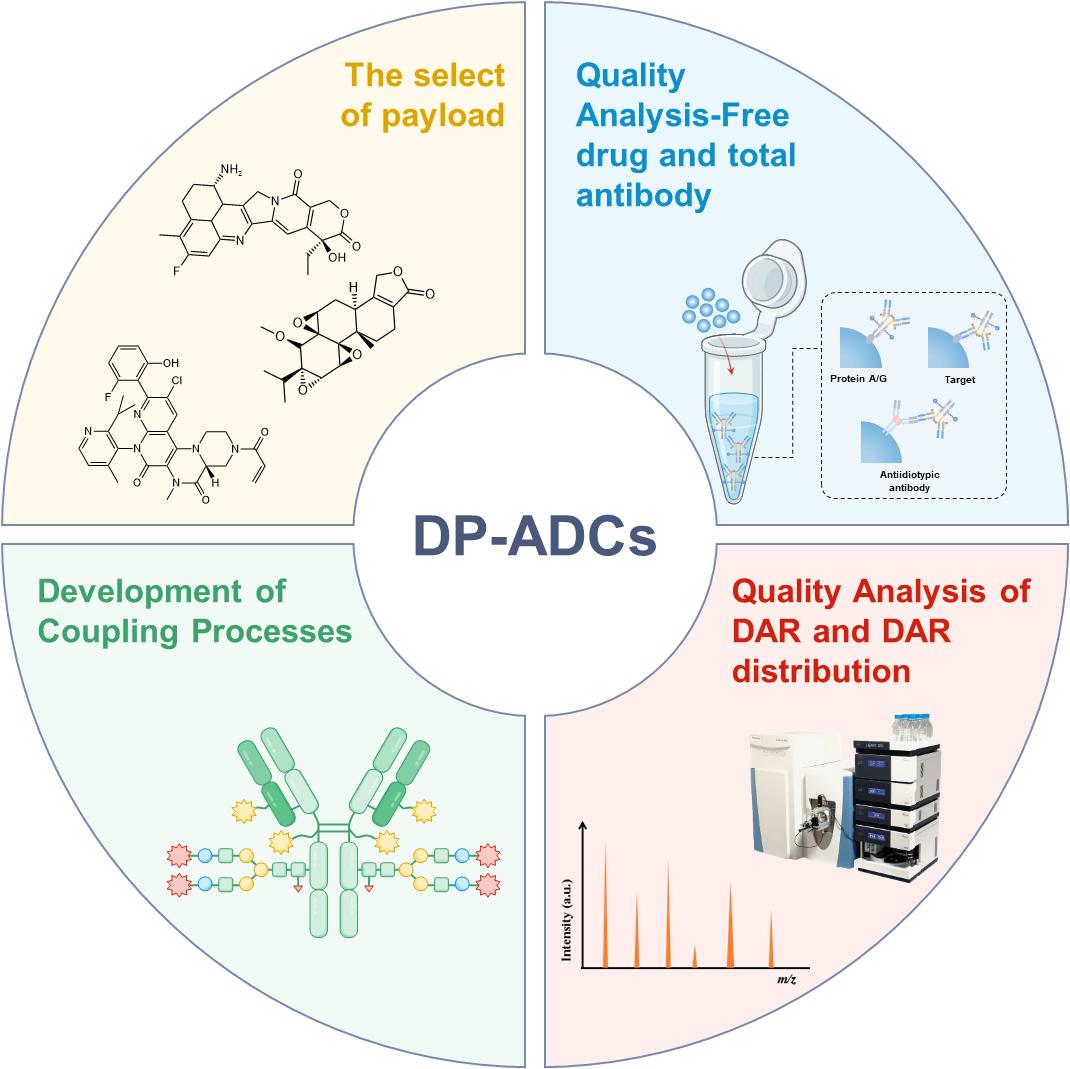
Keywords:
1. Introduction
2. Application of Payload and Coupling Technology in DP-ADCs
2.1. Auristatins
2.2. Maytansinoids
2.3. Camptothecin
2.4. Natural or Nature-Inspired Active Molecules
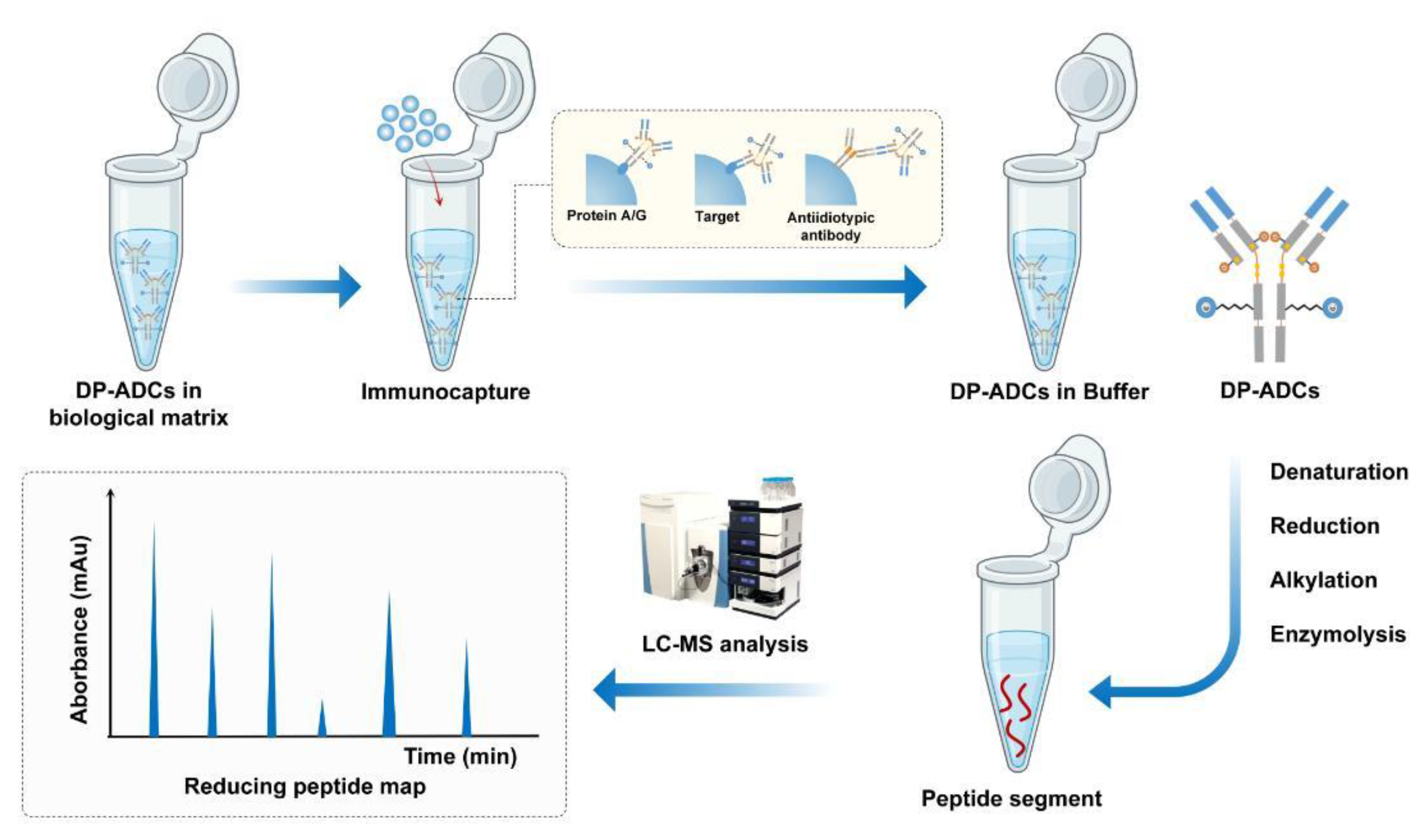
3. Challenges in the Analysis of DP-ADCs
3.1. Quantitative Analysis of the Payload and Payload-Linker
3.2. Quantitative Analysis of the Total Antibodies
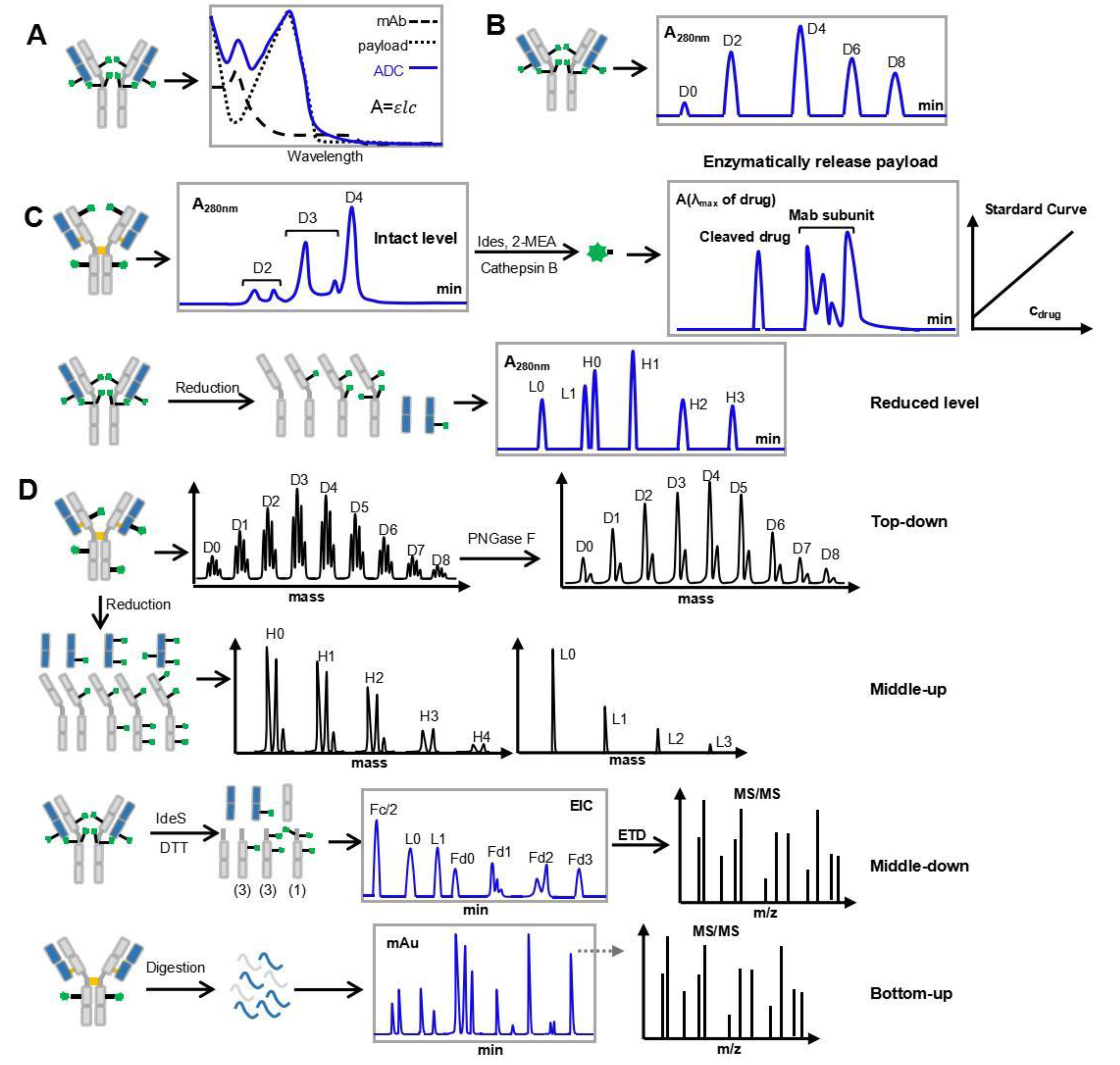
3.3. Determination of DAR and Its Distribution
3.3.1. Overview of Analytical Methods for Determination of DAR and Its Distribution
3.3.2. DAR Analytical Challenges of DP-ADCs
4. Future Landscape
4.1. Advanced Analytical Techniques for Rigorous Quality Control
4.2. Innovative Payload Combinations and Rational Molecular Design
4.3. High-Throughput Screening Platforms to Accelerate Development
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| DAR | drug-to-antibody ratio |
| DBCO | dibenzocyclooctyne |
| PEG | polyethylene glycol |
| GGFG | Gly-Gly-Phe-Gly |
| MMAF | monomethyl auristatin F |
| MMAE | monomethyl auristatin E |
| SN38 | 7-ethyl-10-hydroxycamptothecin |
| VC | Val-Cit |
| SMCC | succinimidyl4-(N-maleimidomethyl)cyclohexane-1-carboxylate |
| MTGase | microbial transglutaminase |
| PABC | p-aminobenzyloxycarbonyl |
| TCO | trans-cyclooctene |
| TOP1i | topoisomerase I inhibition |
| TKI | tyrosine kinase inhibitors |
| MC | maleimide |
| Aldox | doxorubicin |
| vcDOX | mc-vcp-doxorubicin |
| VBL | vinblastine |
| T-DA2 | two cyclopentadiene residues |
| VCP | Va-Cit-p-aminobenzyloxycarbonyl |
References
- Hafeez, U.; Parakh, S.; Gan, H.K.; Scott, A.M. Antibody-Drug Conjugates for Cancer Therapy. Molecules 2020, 25, 4764. [Google Scholar] [CrossRef]
- Hammood, M.; Craig, A.W.; Leyton, J.V. Impact of Endocytosis Mechanisms for the Receptors Targeted by the Currently Approved Antibody-Drug Conjugates (ADCs)-A Necessity for Future ADC Research and Development. Pharmaceuticals 2021, 14, 674. [Google Scholar] [CrossRef] [PubMed]
- Colombo, R.; Rich, J.R. The therapeutic window of antibody drug conjugates: A dogma in need of revision. Cancer Cell. 2022, 40, 1255–1263. [Google Scholar] [CrossRef]
- Ruan, D.-Y.; Wu, H.-X.; Meng, Q.; Xu, R.-H. Development of antibody-drug conjugates in cancer: Overview and prospects. Cancer Commun. (Lond) 2024, 44, 3–22. [Google Scholar] [CrossRef] [PubMed]
- Brignole, C.; Calarco, E.; Bensa, V.; Giusto, E.; Perri, P.; Ciampi, E.; Corrias, M.V.; Astigiano, S.; Cilli, M.; Loo, D.; Bonvini, E.; Pastorino, F.; Ponzoni, M. Antitumor activity of the investigational B7-H3 antibody-drug conjugate, vobramitamab duocarmazine, in preclinical models of neuroblastoma. J. Immunother. Cancer 2023, 11, e007174. [Google Scholar] [CrossRef]
- Murase, Y.; Nanjo, S.; Ueda, T.; Liu, Y.; Nomura, S.; Arai, S.; Terada, N.; Koba, H.; Tambo, Y.; Yano, S. Mechanisms of resistance to antibody-drug conjugates in cancers. Respir. Investig. 2025, 63, 693–698. [Google Scholar] [CrossRef] [PubMed]
- Collins, D.M.; Bossenmaier, B.; Kollmorgen, G.; Niederfellner, G. Acquired Resistance to Antibody-Drug Conjugates. Cancers 2019, 11, 394. [Google Scholar] [CrossRef]
- Pusuluri, A.; Wu, D.; Mitragotri, S. Immunological consequences of chemotherapy: Single drugs, combination therapies and nanoparticle-based treatments. J. Control Release 2019, 305, 130–154. [Google Scholar] [CrossRef]
- Núñez, C.; Capelo, J.L.; Igrejas, G.; Alfonso, A.; Botana, L.M.; Lodeiro, C. An overview of the effective combination therapies for the treatment of breast cancer. Biomaterials 2016, 97, 34–50. [Google Scholar] [CrossRef]
- Tsuchikama, K.; Anami, Y.; Ha, S.Y.Y.; Yamazaki, C.M. Exploring the next generation of antibody-drug conjugates. Nat. Rev. Clin. Oncol. 2024, 21, 203–223. [Google Scholar] [CrossRef]
- Tao, J.; Gu, Y.; Zhou, W.; Wang, Y. Dual-payload antibody-drug conjugates: Taking a dual shot. Eur. J. Med. Chem. 2025, 281, 116995. [Google Scholar] [CrossRef] [PubMed]
- Wen, M.; Yu, A.; Park, Y.; Calarese, D.; Gerber, H.-P.; Yin, G. Homogeneous antibody-drug conjugates with dual payloads: potential, methods and considerations. MAbs 2025, 17, 2498162. [Google Scholar] [CrossRef]
- Khosravifarsani, M.; Njotu, F.N.; Fon, D.A.; Fonge, H. Maximizing therapeutic potential and safety: Exploring multi/dual-payload antibody conjugates as cancer theranostics. Adv. Drug. Deliv. Rev. 2025, 222, 115608. [Google Scholar] [CrossRef]
- Moore, E.J.; Rice, M.; Roy, G.; Zhang, W.; Marelli, M. Emerging conjugation strategies and protein engineering technologies aim to improve ADCs in the fight against cancer. Xenobiotica 2024, 54, 469–491. [Google Scholar] [CrossRef]
- Tang, Y.; Tang, F.; Yang, Y.; Zhao, L.; Zhou, H.; Dong, J.; Huang, W. Real-Time Analysis on Drug-Antibody Ratio of Antibody-Drug Conjugates for Synthesis, Process Optimization, and Quality Control. Sci. Rep. 2017, 7, 7763. [Google Scholar] [CrossRef] [PubMed]
- Koganemaru, S.; Fuchigami, H.; Morizono, C.; Shinohara, H.; Kuboki, Y.; Furuuchi, K.; Uenaka, T.; Doi, T.; Yasunaga, M. Potential Mechanisms of Interstitial Lung Disease Induced by Antibody-Drug Conjugates Based on Quantitative Analysis of Drug Distribution. Mol. Cancer Ther. 2025, 24, 242–250. [Google Scholar] [CrossRef]
- Levengood, M.R.; Zhang, X.; Hunter, J.H.; Emmerton, K.K.; Miyamoto, J.B.; Lewis, T.S.; Senter, P.D. Orthogonal Cysteine Protection Enables Homogeneous Multi-Drug Antibody–Drug Conjugates. Angew. Chem. 2017, 129, 751–755. [Google Scholar] [CrossRef]
- Yamazaki, C.M.; Yamaguchi, A.; Anami, Y.; Xiong, W.; Otani, Y.; Lee, J.; Ueno, N.T.; Zhang, N.; An, Z.; Tsuchikama, K. Antibody-drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance. Nat. Commun. 2021, 12, 3528. [Google Scholar] [CrossRef]
- Tang, C.; Zeng, Y.; Zhang, J.; Zheng, X.; Tang, F.; Yao, X.; Jiang, Z.-X.; Shi, W.; Huang, W. One-Pot Assembly of Dual-Site-Specific Antibody-Drug Conjugates via Glycan Remodeling and Affinity-Directed Traceless Conjugation. Bioconjug Chem. 2023. [Google Scholar] [CrossRef] [PubMed]
- Mckertish, C.M.; Kayser, V. A Novel Dual-Payload ADC for the Treatment of HER2+ Breast and Colon Cancer. Pharmaceutics 2023, 15, 2020. [Google Scholar] [CrossRef]
- Wilski, N.A.; Haytko, P.; Zha, Z.; Wu, S.; Jin, Y.; Chen, P.; Han, C.; Chiu, M.L. A Dual-Payload Bispecific ADC Improved Potency and Efficacy over Single-Payload Bispecific ADCs. Pharmaceutics 2025, 17, 967. [Google Scholar]
- Zhang, Y.; Takeda, S.; Qi, L.; Huang, M.; Zhang, L.; Liu, W.; Mao, Y.; Chen, Z.; Hu, S. Abstract LB021: TJ102: A promising bispecific dual-payload ADC targeting CDH6 and folate receptor alpha (FRα) for the treatment of ovarian and kidney cancers. Cancer Res. 2025, 85. [Google Scholar] [CrossRef]
- Guo, H.; Du, Y.; Huang, Y.; Wang, J.; Yang, Q.; Jia, J.; Zheng, J.; Ye, H.; Du, F.; Dai, M.; Xia, H.; Chen, X.; Jiang, X.; Zhao, Z.; Liu, W.; Abstract, R.Z. 2872: DXC018: A novel HER2 dual-payload bispecific ADC with promising potential for gastrointestinal cancers and other solid tumors. Cancer Res. 2025, 85, 2872–2872. [Google Scholar] [CrossRef]
- Xu, T.; Li, F.; Lai, Z.; Abstract, R.Q. 5451: JSKN021, an innovative site-specific dual-payload bispecific antibody drug conjugate targeting EGFR and HER3 exhibits potent preclinical activities. Cancer Res. 2025, 85, 5451–5451. [Google Scholar] [CrossRef]
- Wang, L.; Xiong, M.; Gao, X.; Zhou, C.; Han, Y.; Li, Y.; Wang, J.; Shi, L.; Qin, G.; Abstract, P.H.S. 2114: Enhancing therapeutic strategies for osimertinib-resistant EGFR-mutant NSCLC: A HER3 dual-payload ADC (dpADC) with topoisomerase I and EGFR tyrosine kinase inhibitor. Cancer Res. 2024, 84, 2114. [Google Scholar] [CrossRef]
- Attinger-Toller, I.; Probst, P.; Bertrand, R.; Fay, R.; Kallenberger, L.; Maurhofer, P.; Stark, R.; Renard, E.; Santimaria, R.; Grabulovski, D.; Schlereth, B.; Abstract, P.R. S. 7334: Targeting Nectin-4 with a first-in-class triple MMAE/dual TOP1i payload ADC showing synergistic and durable activity across all target expression levels and favorable tolerability. Cancer Res. 2025, 85, 7334. [Google Scholar] [CrossRef]
- Xiong, M.; Fan, B.; Gao, X.; Sun, Y.; Lv, C.; Si, Y.; Liu, X.; Shi, L.; Song, P.H.; Abstract, G.Q. 2640: An innovative site-specific dual-payload antibody drug conjugate (dpADC) combining a novel Topo1 inhibitor and an immune agonist delivers a strong immunogenic cell death (ICD) and antitumor response in vitro and in vivo. Cancer Res. 2023, 83, 2640. [Google Scholar] [CrossRef]
- Attinger-Toller, I.; Probst, P.; Bertrand, R.; Renard, E.; Kallenberger, L.; Stark, R.; Maurhofer, P.; Grabulovski, D.; Schlereth, B.; Spycher, P. Abstract LB138: A novel dual-TOP1i ADC targeting NaPi2b designed for high efficacy and tolerability. Cancer Res. 2025, 85. [Google Scholar] [CrossRef]
- Ren, P.; Guan, M.; Tang, J.; Yin, S.; Qi, L.; Yue, J.; Li, Z.; Fan, X.; Lei, G.; Zuo, T.; Chen, J.; Xu, Y.; Ke, X.; Zhao, Y. A Novel Dual-Payload ADC Platform Integrating Exatecan and Triptolide to Enhance Antitumor Efficacy and Overcome Resistance. Mol. Cancer Ther. 2025, OF1–OF12. [CrossRef]
- Zhao, Y.; Ren, P.; Menglong, G.; Tang, J.; Qi, L.; Fan, X.; Yin, S.; Lei, G.; Abstract, X.K. 1587: A novel dual-payload HER3-directed antibody-drug conjugate, shows potent antitumor efficacy in HER3-low in vivo tumor models. Cancer Res. 2025, 85, 1587–1587. [Google Scholar] [CrossRef]
- Li, H.-K.; Lai, C.-W.; Lin, C.-Y.; Chen, Y.-J.; Leng, P.-J.; Cheng, C.-M.; Tang, S.-W.; Abstract, S.-C.H. 1785: Development of dual-payload anti-GPC3 antibody-drug conjugate by dual-payload antibody conjugation (AD2C) platform for hepatocellular carcinoma treatment. Cancer Res. 2025, 85, 1785. [Google Scholar] [CrossRef]
- Lee, C.-S.; Kim, M.J.; Kim, D.H.; Lim, S.; Lee, J.; Choi, E.; Ha, W.; Kim, H.A.; Park, J.; Abstract, S.C.K. 6755: Discovery of the synergistic dual-payload antibody-drug conjugate (CTPH-02) by combination of MMAE and novel payloads. Cancer Res. 2025, 85, 6755. [Google Scholar] [CrossRef]
- Zhou, Z.Z.; Si, Y.; Zhang, J.; Chen, K.; George, A.; Kim, S.; Zhou, L.; Liu, X.M. A Dual-Payload Antibody-Drug Conjugate Targeting CD276/B7-H3 Elicits Cytotoxicity and Immune Activation in Triple-Negative Breast Cancer. Cancer Res. 2024, 84, 3848–3863. [Google Scholar] [CrossRef] [PubMed]
- Nervig, C.S.; Rice, M.; Marelli, M.; Christie, R.J.; Owen, S.C. Modular Synthesis of Anti-HER2 Dual-Drug Antibody-Drug Conjugates Demonstrating Improved Toxicity. Bioconjug Chem. 2025, 36, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Yao, X.; Jiang, J.; Wang, X.; Huang, C.; Li, D.; Xie, K.; Xu, Q.; Li, H.; Li, Z.; Lou, L.; Fang, J. A novel humanized anti-HER2 antibody conjugated with MMAE exerts potent anti-tumor activity. Breast Cancer Res. Treat. 2015, 153, 123–133. [Google Scholar] [CrossRef]
- Best, R.L.; LaPointe, N.E.; Azarenko, O.; Miller, H.; Genualdi, C.; Chih, S.; Shen, B.-Q.; Jordan, M.A.; Wilson, L.; Feinstein, S.C.; Stagg, N.J. Microtubule and tubulin binding and regulation of microtubule dynamics by the antibody drug conjugate (ADC) payload, monomethyl auristatin E (MMAE): Mechanistic insights into MMAE ADC peripheral neuropathy. Toxicol. Appl. Pharmacol. 2021, 421, 115534. [Google Scholar] [CrossRef]
- Probst, P.; Attinger-Toller, I.; Bertrand, R.; Stark, R.; Santimaria, R.; Schlereth, B.; Grabulovski, D.; Spycher, P.R. Broadening the Therapeutic Window of ADCs Using Site-Specific Bioconjugation Showcased by an MMAE-Containing Peptide Linker in a CD79b-Targeting ADC. Mol. Cancer Ther. 2025, 24, 803–815. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Griffith, J.I.; Porath, K.A.; Rathi, S.; Le, J.; Pasa, T.I.; Decker, P.A.; Gupta, S.K.; Hu, Z.; Carlson, B.L.; Bakken, K.; Burgenske, D.M.; Feldsien, T.M.; Lefebvre, D.R.; Vaubel, R.A.; Eckel-Passow, J.E.; Reilly, E.B.; Elmquist, W.F.; Effects, J.N.S.B. Pharmacokinetics, and Linker-Payload Stability of EGFR-Targeting Antibody-Drug Conjugates Losatuxizumab Vedotin and Depatux-M in Glioblastoma Models. Clin. Cancer Res. 2024, 30, 3287–3297. [Google Scholar] [CrossRef]
- Sokka, I.K.; Imlimthan, S.; Sarparanta, M.; Maaheimo, H.; Johansson, M.P.; Ekholm, F.S. Halogenation at the Phenylalanine Residue of Monomethyl Auristatin F Leads to a Favorable cis/trans Equilibrium and Retained Cytotoxicity. Mol. Pharm. 2021, 18, 3125–3131. [Google Scholar] [CrossRef]
- N.W.C.J. van de Donk, E. Dhimolea, Brentuximab vedotin. MAbs, 4, pp. 458–465.
- Polatuzumab Vedotin, in: Drugs and Lactation Database (LactMed®), National Institute of Child Health and Human Development, Bethesda (MD), 2006. http://www.ncbi.nlm.nih.gov/books/NBK592187/ (accessed March 31, 2026).
- Enfortumab Vedotin, in: Drugs and Lactation Database (LactMed®), National Institute of Child Health and Human Development, Bethesda (MD), 2006. http://www.ncbi.nlm.nih.gov/books/NBK567878/ (accessed March 31, 2026).
- Belantamab Mafodotin, in: LiverTox: Clinical and Research Information on Drug-Induced Liver Injury, National Institute of Diabetes and Digestive and Kidney Diseases, Bethesda (MD), 2012. http://www.ncbi.nlm.nih.gov/books/NBK598877/ (accessed March 31, 2026).
- Tislelizumab, in: Drugs and Lactation Database (LactMed®), National Institute of Child Health and Human Development, Bethesda (MD), 2006.
- Kupchan, S.M.; Komoda, Y.; Court, W.A.; Thomas, G.J.; Smith, R.M.; Karim, A.; Gilmore, C.J.; Haltiwanger, R.C.; Bryan, R.F. Maytansine, a novel antileukemic ansa macrolide from Maytenus ovatus. J. Am. Chem. Soc. 1972, 94, 1354–1356. [Google Scholar] [CrossRef]
- Lewis Phillips, G.D.; Li, G.; Dugger, D.L.; Crocker, L.M.; Parsons, K.L.; Mai, E.; Blättler, W.A.; Lambert, J.M.; Chari, R.V.J.; Lutz, R.J.; Wong, W.L.T.; Jacobson, F.S.; Koeppen, H.; Schwall, R.H.; Kenkare-Mitra, S.R.; Spencer, S.D.; Sliwkowski, M.X. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody-cytotoxic drug conjugate. Cancer Res. 2008, 68, 9280–9290. [Google Scholar] [CrossRef] [PubMed]
- L. F, K. J, S. Y, T-DM1-induced thrombocytopenia in breast cancer patients: New perspectives, Biomedicine & Pharmacotherapy = Biomedecine & Pharmacotherapie 129 (2020).
- Moore, K.N.; Oza, A.M.; Colombo, N.; Oaknin, A.; Scambia, G.; Lorusso, D.; Konecny, G.E.; Banerjee, S.; Murphy, C.G.; Tanyi, J.L.; Hirte, H.; Konner, J.A.; Lim, P.C.; Prasad-Hayes, M.; Monk, B.J.; Pautier, P.; Wang, J.; Berkenblit, A.; Vergote, I.; Birrer, M.J. Phase II, randomized trial of mirvetuximab soravtansine versus chemotherapy in patients with platinum-resistant ovarian cancer: primary analysis of FORWARD I. Ann. Oncol. 2021, 32, 757–765. [Google Scholar] [CrossRef]
- Petersen, M.E.; Brant, M.G.; Lasalle, M.; Das, S.; Duan, R.; Wong, J.; Ding, T.; Wu, K.J.; Siddappa, D.; Fang, C.; Zhang, W.; Wu, A.M.L.; Hirkala-Schaefer, T.; Garnett, G.A.E.; Fung, V.; Yang, L.; Rojas, A.H.; Lawn, S.O.; Barnscher, S.D.; Rich, J.R.; Colombo, R. Design and Evaluation of ZD06519, a Novel Camptothecin Payload for Antibody Drug Conjugates. Mol. Cancer Ther. 2024, 23, 606–618. [Google Scholar] [CrossRef]
- Wang, L.; Yin, H.; Jiang, J.; Li, Q.; Gao, C.; Li, W.; Zhang, B.; Xin, Y.; Li, H.; Zhao, M.; Lu, Q. A rationally designed CD19 monoclonal antibody-triptolide conjugate for the treatment of systemic lupus erythematosus. Acta Pharm. Sin. B 2024, 14, 4560–4576. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Huangfu, S.; Wei, D.; Sun, Z.; Wu, Y.; Yu, X.; Jiang, B.; Chen, H. Triptolide-based cleavable antibody-drug conjugates for pancreatic cancer. Eur. J. Med. Chem. 2025, 295, 117798. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Ma, Y.; Guo, Y.; Sun, T.; Wu, J.; Pangeni, R.P.; Lin, M.; Li, W.; Horne, D.; Raz, D.J. Cetuximab-Triptolide Conjugate Suppresses the Growth of EGFR-Overexpressing Lung Cancers through Targeting RNA Polymerase II. Mol. Ther. Oncolytics 2020, 18, 304–316. [Google Scholar] [CrossRef]
- Pan, J. RNA polymerase - an important molecular target of triptolide in cancer cells. Cancer Lett. 2010, 292, 149–152. [Google Scholar] [CrossRef]
- Liang, X.; Xie, R.; Su, J.; Ye, B.; Wei, S.; Liang, Z.; Bai, R.; Chen, Z.; Li, Z.; Gao, X. Inhibition of RNA polymerase III transcription by Triptolide attenuates colorectal tumorigenesis. J. Exp. Clin. Cancer Res. 2019, 38, 217. [Google Scholar] [CrossRef]
- Bukhalid, R.A.; Duvall, J.R.; Lancaster, K.; Catcott, K.C.; Cetinbas, N.M.; Monnell, T.; Routhier, C.; Thomas, J.D.; Bentley, K.W.; Collins, S.D.; Ditty, E.; Eitas, T.K.; Kelleher, E.W.; Shaw, P.; Soomer-James, J.; Ter-Ovanesyan, E.; Xu, L.; Zurita, J.; Toader, D.; Damelin, M.; Lowinger, T.B. XMT-2056, a HER2-Directed STING Agonist Antibody-Drug Conjugate, Induces Innate Antitumor Immune Responses by Acting on Cancer Cells and Tumor-Resident Immune Cells. Clin. Cancer Res. 2025, 31, 1766–1782. [Google Scholar]
- Rejili, M. Synergistic strategies: ADC-PARP inhibitor combinations in triple-negative breast cancer therapy. Pathol. Res. Pract. 2025, 272, 156075. [Google Scholar]
- Semenova, G.; Frank, S.; Dumpit, R.; Han, W.; Coleman, I.; Gulati, R.; Morrissey, C.; Haffner, M.C.; Nelson, P.S.; Lee, J.K. Genotoxic antibody-drug conjugates combined with Bcl-xL inhibitors enhance therapeutic efficacy in metastatic castration-resistant prostate cancer. bioRxiv 2025, 2025.09.05.674562. [Google Scholar]
- Foreman, R.E.; Lucey, R.; Leaney, A.R.; Lee, M.-Y.; Naseer, H.; Wilson, A. Optimized LC-MS/MS methods for quantifying antibody-drug conjugate payloads in cell culture media containing phenol red. Bioanalysis 2024, 16, 575–585. [Google Scholar] [CrossRef]
- Zhu, X.; Huo, S.; Xue, C.; An, B.; Qu, J. Current LC-MS-based strategies for characterization and quantification of antibody-drug conjugates. J. Pharm. Anal. 2020, 10, 209–220. [Google Scholar] [CrossRef]
- Qin, Q.; Gong, L. Current Analytical Strategies for Antibody-Drug Conjugates in Biomatrices. Molecules 2022, 27, 6299. [Google Scholar] [CrossRef]
- Jansen, F.H.; Soomro, S.A. Chemical instability determines the biological action of the artemisinins. Curr. Med. Chem. 2007, 14, 3243–3259. [Google Scholar] [CrossRef]
- Li, Y.; Stella, C.; Zheng, L.; Bechtel, C.; Gruenhagen, J.; Jacobson, F.; Medley, C.D. Investigation of low recovery in the free drug assay for antibody drug conjugates by size exclusion—reversed phase two dimensional-liquid chromatography. J. Chromatogr. B 2016, 1032, 112–118. [Google Scholar] [CrossRef]
- Lyon, R.P.; Setter, J.R.; Bovee, T.D.; Doronina, S.O.; Hunter, J.H.; Anderson, M.E.; Balasubramanian, C.L.; Duniho, S.M.; Leiske, C.I.; Li, F.; Senter, P.D. Self-hydrolyzing maleimides improve the stability and pharmacological properties of antibody-drug conjugates. Nat. Biotechnol. 2014, 32, 1059–1062. [Google Scholar] [PubMed]
- Mak, S.Y.; Chen, S.; Fong, W.J.; Choo, A.; Ho, Y.S. A simple and highly sensitive LC-MS workflow for characterization and quantification of ADC cleavable payloads. Sci. Rep. 2024, 14, 11018. [Google Scholar] [PubMed]
- Oguma, T.; Cicci, D.; Gaudette, F.; Gauthier-Dubois, G.; Guilbaud, R.; Atsumi, R. Validation study of a method for assaying DE-310, a macromolecular carrier conjugate containing an anti-tumor camptothecin derivative, and the free drug in human plasma by HPLC and LC/MS/MS. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 818, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Zmolek, W.; Bañas, S.; Barfield, R.M.; Rabuka, D.; Drake, P.M. A simple LC/MRM-MS-based method to quantify free linker-payload in antibody-drug conjugate preparations. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 1032, 144–148. [Google Scholar]
- Wang, J.; Zhang, W.; Salter, R.; Lim, H.-K. Reductive Desulfuration as an Important Tool in Detection of Small Molecule Modifications to Payload of Antibody Drug Conjugates. Anal. Chem. 2019, 91, 2368–2375. [Google Scholar] [CrossRef]
- Li, X.; Cheng, M.; Wang, Y.; Shu, C.; Zou, B.; Song, Q.; Ding, L. LC-MS/MS method for dual-ligand peptide-drug CBP-1018 and its deconjugated payload MMAE including sample stabilization strategy for its MC-Val-Cit-PABC linker. Talanta 2024, 279, 126596. [Google Scholar] [CrossRef]
- Yin, F.; Ahsan, F.; Pinkas, J.; Das, B.; Wang, F.; Zheng, N.; Hahn, D.; Amrite, A.; Feng, J.; Adhikari, D.; Sikora, J.; Shaheen, E.; Harriman, S. A sensitive LC-MS/MS assay to quantitate free payload Aur0101 from ADC PYX-201 in rat and monkey plasma. Bioanalysis 2023, 15, 833–843. [Google Scholar] [CrossRef]
- Suh, M.-J.; Powers, J.B.; Daniels, C.M.; Wu, Y. Enhanced Pharmacokinetic Bioanalysis of Antibody-drug Conjugates using Hybrid Immunoaffinity Capture and Microflow LC-MS/MS. AAPS J. 2023, 25, 68. [Google Scholar] [CrossRef] [PubMed]
- Heudi, O.; Barteau, S.; Picard, F.; Kretz, O. Quantitative analysis of maytansinoid (DM1) in human serum by on-line solid phase extraction coupled with liquid chromatography tandem mass spectrometry - Method validation and its application to clinical samples. J. Pharm. Biomed. Anal. 2016, 120, 322–332. [Google Scholar] [CrossRef]
- Yin, F.; Adhikari, D.; Peay, M.; Cortes, D.; Garada, M.; Woolf, M.S.; Ma, E.; Lebarbenchon, D.; Mylott, W.; Dyszel, M.; Harriman, S.; Pinkas, J. Development and validation of a hybrid immunoaffinity LC-MS/MS assay for quantitation of total antibody (TAb) from an antibody drug conjugate (ADC) PYX-201 in human plasma. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2023, 1228, 123844. [Google Scholar] [CrossRef]
- Yin, F.; Adhikari, D.; Sun, M.; Woolf, M.S.; Ma, E.; Mylott, W.; Shaheen, E.; Harriman, S.; Pinkas, J. Bioanalysis of an antibody drug conjugate (ADC) PYX-201 in human plasma using a hybrid immunoaffinity LC-MS/MS approach. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2023, 1223, 123715. [Google Scholar] [CrossRef]
- Kumar, D.; Trivedi, N. Advances in LC-MS strategies for comprehensive characterization and quantification of antibody-drug conjugates in preclinical and clinical settings. J. Chromatogr. A 2026, 1766, 466606. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.M.N.; Zeng, Y.; Lanihun, A.-A.A.; Arisa, O.T.; Horner, J.L.; Figg, W.D. Recent Advances in Bioanalytical Methods for Quantification and Pharmacokinetic Analyses of Antibody-Drug Conjugates. AAPS J. 2025, 27, 138. [Google Scholar] [CrossRef]
- Mou, S.; Huang, Y.; Rosenbaum, A.I. ADME Considerations and Bioanalytical Strategies for Pharmacokinetic Assessments of Antibody-Drug Conjugates. Antibodies 2018, 7, 41. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; An, B.; Shen, S.; Zhang, M.; Shen, X.; Duan, X.; Balthasar, J.P.; Qu, J. Qualitative and quantitative characterization of protein biotherapeutics with liquid chromatography mass spectrometry. Mass. Spectrom. Rev. 2017, 36, 734–754. [Google Scholar] [CrossRef]
- Li, X.; Chen, X.; Zhong, D. [Bioanalysis in the development of antibody-drug conjugates]. Yao Xue Xue Bao 2016, 51, 517–528. [Google Scholar]
- Wang, J.; Gu, H.; Liu, A.; Kozhich, A.; Rangan, V.; Myler, H.; Luo, L.; Wong, R.; Sun, H.; Wang, B.; Vezina, H.E.; Deshpande, S.; Zhang, Y.; Yang, Z.; Olah, T.V.; Aubry, A.-F.; Arnold, M.E.; Pillutla, R.; DeSilva, B. Antibody-drug conjugate bioanalysis using LB-LC-MS/MS hybrid assays: strategies, methodology and correlation to ligand-binding assays. Bioanalysis 2016, 8, 1383–1401. [Google Scholar] [CrossRef]
- Han, L.; Zhao, Y.; Zhang, Q. Conjugation Site Analysis by MS/MS Protein Sequencing. Methods Mol. Biol. 2020, 2078, 221–233. [Google Scholar] [PubMed]
- Triebel, L.J.; Wissenbach, D.; Peters, F.T.; Wissenbach, D.K. Adduct formation by coeluting endogenous biomolecules using ESI-MS: A comprehensive study on ESI efficiency. Anal. Bioanal. Chem. 2025, 417, 6327–6337. [Google Scholar] [CrossRef]
- Zhou, W.; Gong, M.; Mao, Y.; Yuan, X.; Deng, Y.; Zhang, Q.; Guo, W.; Qiu, L.; Huang, X.; Cao, Z.; Xia, J.; She, X.; Cong, Y.; Zhang, C.; Liu, H.; Chen, W. Analytical performance evaluation and optimization of serum 25(OH)D LC-MS/MS measurement. Clin. Chem. Lab. Med. 2025, 63, 1354–1365. [Google Scholar] [CrossRef]
- Yu, X.; Li, W.; Huang, W.; Xiao, B.; Long, J.; Wang, Q.; Wang, G.; Wang, C.; Yu, M.; Yu, J.; Diao, X. Simultaneous Quantification of Total Antibody and Conjugated Payload for DS001 in Rat Serum Using a Hybrid Immuno-Capture LC–MS/MS. AAPS J. 2025, 27, 23. [Google Scholar] [CrossRef]
- Kristensen, D.B.; Eskesen, N.S.; Coll-Satue, C.; Nicolas, A.; Simonsen, J.K.; Rasmussen, L.; Sloth, T.M.; Ørgaard, M.; Madzharova, E.; Krabbe, S.; Leth, K.Z.; Jensen, P.F.; Beck, A. Rapid and selective characterization of antibody-drug conjugates in complex sample matrices by native affinity liquid chromatography-mass spectrometry. mAbs 2026, 18, 2618314. [Google Scholar] [CrossRef] [PubMed]
- Ieki, K.; Fukuda, S.; Miyawaki, S.; Hirowatari, K. Regulated bioanalysis of antibody-drug conjugates using LC-MS. Bioanalysis 2025, 17, 549–560. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Wang, C.; Wu, Y.; Dong, L.; Chen, F.; Dong, K.; Song, H. Simple and Rapid LC–MS/MS Methods for Quantifying Catabolites of Antibody-Drug Conjugates with SMCC Linker. J. Chromatogr. Sci. 2021, 59, 642–649. [Google Scholar] [CrossRef]
- Hashii, N.; Haruyama, Y.; Hirayama, R.; Kajita, R.; Kishino, Y.; Mochizuki, T.; Inoue, K.; Goda, R.; Hoshino, M.; Kuroiwa, I.; Aikawa, H.; Ueda, N.; Nagumo, K.; Oda, Y.; Saito, Y.; Ishii-Watabe, A. Versatile intact LC–MS method for evaluating the drug–antibody ratio and drug load distribution of antibody–drug conjugates in human plasma. J. Chromatogr. B 2025, 1258, 124608. [Google Scholar]
- Mckertish, C.; Kayser, V. Advances and Limitations of Antibody Drug Conjugates for Cancer. Biomedicines 2021, 9, 872. [Google Scholar] [CrossRef]
- Hamblett, K.J.; Senter, P.D.; Chace, D.F.; Sun, M.M.C.; Lenox, J.; Cerveny, C.G.; Kissler, K.M.; Bernhardt, S.X.; Kopcha, A.K.; Zabinski, R.F.; Meyer, D.L.; Francisco, J.A. Effects of Drug Loading on the Antitumor Activity of a Monoclonal Antibody Drug Conjugate. Clin. Cancer Res. 2004, 10, 7063–7070. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, C.M.; Yamaguchi, A.; Anami, Y.; Xiong, W.; Otani, Y.; Lee, J.; Ueno, N.T.; Zhang, N.; An, Z.; Tsuchikama, K. Antibody-drug conjugates with dual payloads for combating breast tumor heterogeneity and drug resistance. Nat. Commun. 2021, 12, 3528. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, Y.; Mendelsohn, B.A. Recent Advances in Drug–Antibody Ratio Determination of Antibody–Drug Conjugates. Chem. Pharm. Bull. 2021, 69, 976–983. [Google Scholar] [CrossRef]
- Y. Chen, Drug-to-Antibody Ratio (DAR) by UV/Vis Spectroscopy, in: L. Ducry (Ed.).
- Wagh, A.; Song, H.; Zeng, M.; Tao, L.; Das, T.K. Challenges and new frontiers in analytical characterization of antibody-drug conjugates. mAbs 2018, 10, 222–243. [Google Scholar] [CrossRef]
- Liu, T.; Tao, Y.; Xia, X.; Zhang, Y.; Deng, R.; Wang, Y. Analytical tools for antibody–drug conjugates: From in vitro to in vivo. TrAC. Trends Anal. Chem. 2022, 152, 116621. [Google Scholar] [CrossRef]
- Matsuda, Y.; Tawfiq, Z.; Leung, M.; Mendelsohn, B.A. Insight into Temperature Dependency and Design of Experiments towards Process Development for Cysteine-Based Antibody-Drug Conjugates. ChemistrySelect 2020, 5, 8435–8439. [Google Scholar]
- Källsten, M.; Hartmann, R.; Artemenko, K.; Lind, S.B.; Lehmann, F.; Bergquist, J. Qualitative analysis of antibody–drug conjugates (ADCs): an experimental comparison of analytical techniques of cysteine-linked ADCs. Analyst 2018, 143, 5487–5496. [Google Scholar]
- Zhou, Q.; Stefano, J.E.; Manning, C.; Kyazike, J.; Chen, B.; Gianolio, D.A.; Park, A.; Busch, M.; Bird, J.; Zheng, X.; Simonds-Mannes, H.; Kim, J.; Gregory, R.C.; Miller, R.J.; Brondyk, W.H.; Dhal, P.K.; Pan, C.Q. Site-Specific Antibody–Drug Conjugation through Glycoengineering. Bioconjugate Chem. 2014, 25, 510–520. [Google Scholar]
- Matsuda, Y.; Robles, V.; Malinao, M.-C.; Song, J.; Mendelsohn, B.A. Comparison of Analytical Methods for Antibody–Drug Conjugates Produced by Chemical Site-Specific Conjugation: First-Generation AJICAP. Anal. Chem. 2019, 91, 12724–12732. [Google Scholar] [CrossRef]
- Haverick, M.; Mengisen, S.; Shameem, M.; Ambrogelly, A. Separation of mAbs molecular variants by analytical hydrophobic interaction chromatography HPLC: overview and applications. mAbs 2014, 6, 852–858. [Google Scholar] [CrossRef]
- Bobaly, B.; Beck, A.; Veuthey, J.-L.; Guillarme, D.; Fekete, S. Impact of organic modifier and temperature on protein denaturation in hydrophobic interaction chromatography. J. Pharm. Biomed. Anal. 2016, 131, 124–132. [Google Scholar] [CrossRef]
- Bobály, B.; Randazzo, G.M.; Rudaz, S.; Guillarme, D.; Fekete, S. Optimization of non-linear gradient in hydrophobic interaction chromatography for the analytical characterization of antibody-drug conjugates. J. Chromatogr. A 2017, 1481, 82–91. [Google Scholar]
- Andris, S.; Hubbuch, J. Modeling of hydrophobic interaction chromatography for the separation of antibody-drug conjugates and its application towards quality by design. J. Biotechnol. 2020, 317, 48–58. [Google Scholar] [CrossRef]
- Wiggins, B.; Liu-Shin, L.; Yamaguchi, H.; Ratnaswamy, G. Characterization of Cysteine-Linked Conjugation Profiles of Immunoglobulin G1 and Immunoglobulin G2 Antibody–Drug Conjugates. J. Pharm. Sci. 2015, 104, 1362–1372. [Google Scholar] [PubMed]
- D’Atri, V.; Pell, R.; Clarke, A.; Guillarme, D.; Fekete, S. Is hydrophobic interaction chromatography the most suitable technique to characterize site-specific antibody-drug conjugates? J. Chromatogr. A 2019, 1586, 149–153. [Google Scholar] [CrossRef]
- Fekete, S.; Molnár, I.; Guillarme, D. Separation of antibody drug conjugate species by RPLC: A generic method development approach. J. Pharm. Biomed. Anal. 2017, 137, 60–69. [Google Scholar] [CrossRef] [PubMed]
- Apffel, A.; Fischer, S.; Goldberg, G.; Goodley, P.C.; Kuhlmann, F.E. Enhanced sensitivity for peptide mapping with electrospray liquid chromatography-mass spectrometry in the presence of signal suppression due to trifluoroacetic acid-containing mobile phases. J. Chromatogr. A 1995, 712, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Bobály, B.; Beck, A.; Fekete, J.; Guillarme, D.; Fekete, S. Systematic evaluation of mobile phase additives for the LC–MS characterization of therapeutic proteins. Talanta 2015, 136, 60–67. [Google Scholar] [CrossRef]
- Tawfiq, Z.; Matsuda, Y.; Alfonso, M.J.; Clancy, C.; Robles, V.; Leung, M.; Mendelsohn, B.A. Analytical Comparison of Antibody-drug Conjugates Based on Good Manufacturing Practice Strategies. ANAL. SCI. 2020, 36, 871–875. [Google Scholar] [CrossRef] [PubMed]
- Tawfiq, Z.; Caiazza, N.C.; Kambourakis, S.; Matsuda, Y.; Griffin, B.; Lippmeier, J.C.; Mendelsohn, B.A. Synthesis and Biological Evaluation of Antibody Drug Conjugates Based on an Antibody Expression System: Conamax. ACS Omega 2020, 5, 7193–7200. [Google Scholar] [CrossRef]
- Li, Y.; Gu, C.; Gruenhagen, J.; Yehl, P.; Chetwyn, N.P.; Medley, C.D. An enzymatic deconjugation method for the analysis of small molecule active drugs on antibody-drug conjugates. mAbs 2016, 8, 698–705. [Google Scholar] [CrossRef]
- Gikanga, B.; Adeniji, N.S.; Patapoff, T.W.; Chih, H.-W.; Yi, L. Cathepsin B Cleavage of vcMMAE-Based Antibody–Drug Conjugate Is Not Drug Location or Monoclonal Antibody Carrier Specific. Bioconjugate Chem. 2016, 27, 1040–1049. [Google Scholar] [CrossRef]
- Adamo, M.; Sun, G.; Qiu, D.; Valente, J.; Lan, W.; Song, H.; Bolgar, M.; Katiyar, A.; Krishnamurthy, G. Drug-to-antibody determination for an antibody-drug-conjugate utilizing cathepsin B digestion coupled with reversed-phase high-pressure liquid chromatography analysis. J. Chromatogr. A 2017, 1481, 44–52. [Google Scholar] [CrossRef]
- Jeffrey, S.C.; De Brabander, J.; Miyamoto, J.; Senter, P.D. Expanded Utility of the β-Glucuronide Linker: ADCs That Deliver Phenolic Cytotoxic Agents. ACS Med. Chem. Lett. 2010, 1, 277–280. [Google Scholar] [CrossRef]
- Valliere-Douglass, J.F.; McFee, W.A.; Salas-Solano, O. Native Intact Mass Determination of Antibodies Conjugated with Monomethyl Auristatin E and F at Interchain Cysteine Residues. Anal. Chem. 2012, 84, 2843–2849. [Google Scholar] [CrossRef]
- Jones, J.; Pack, L.; Hunter, J.H.; Valliere-Douglass, J.F. Native size-exclusion chromatography-mass spectrometry: suitability for antibody–drug conjugate drug-to-antibody ratio quantitation across a range of chemotypes and drug-loading levels. mAbs 2020, 12, 1682895. [Google Scholar]
- Nguyen, M.; Smith, J.; Rzewuski, S.; Legido-Quigley, C.; Lauber, M.A. High sensitivity LC-MS profiling of antibody-drug conjugates with difluoroacetic acid ion pairing. mAbs 2019, 11, 1358–1366. [Google Scholar]
- Sarrut, M.; Corgier, A.; Fekete, S.; Guillarme, D.; Lascoux, D.; Janin-Bussat, M.-C.; Beck, A.; Heinisch, S. Analysis of antibody-drug conjugates by comprehensive on-line two-dimensional hydrophobic interaction chromatography x reversed phase liquid chromatography hyphenated to high resolution mass spectrometry. I − Optimization of separation conditions. J. Chromatogr. B 2016, 1032, 103–111. [Google Scholar]
- Källsten, M.; Pijnappel, M.; Hartmann, R.; Lehmann, F.; Kovac, L.; Lind, S.B.; Bergquist, J. Application of triple quadrupole mass spectrometry for the characterization of antibody–drug conjugates. Anal. Bioanal. Chem. 2019, 411, 2569–2576. [Google Scholar] [CrossRef]
- Larson, E.J.; Zhu, Y.; Wu, Z.; Chen, B.; Zhang, Z.; Zhou, S.; Han, L.; Zhang, Q.; Ge, Y. Rapid Analysis of Reduced Antibody Drug Conjugate by Online LC-MS/MS with Fourier Transform Ion Cyclotron Resonance Mass Spectrometry. Anal. Chem. 2020, 92, 15096–15103. [Google Scholar] [CrossRef] [PubMed]
- Todoroki, K.; Mizuno, H.; Sugiyama, E.; Toyo’oka, T. Bioanalytical methods for therapeutic monoclonal antibodies and antibody–drug conjugates: A review of recent advances and future perspectives. J. Pharm. Biomed. Anal. 2020, 179, 112991. [Google Scholar] [CrossRef]
- Wang, J.; Gu, H.; Liu, A.; Kozhich, A.; Rangan, V.; Myler, H.; Luo, L.; Wong, R.; Sun, H.; Wang, B.; Vezina, H.E.; Deshpande, S.; Zhang, Y.; Yang, Z.; Olah, T.V.; Aubry, A.-F.; Arnold, M.E.; Pillutla, R.; DeSilva, B. Antibody–Drug Conjugate Bioanalysis Using Lb-LC–MS/MS Hybrid Assays: Strategies, Methodology and Correlation to Ligand-Binding Assays. Bioanalysis 2016, 8, 1383–1401. [Google Scholar] [CrossRef] [PubMed]
- Von Pawel-Rammingen, U. IdeS, a novel streptococcal cysteine proteinase with unique specificity for immunoglobulin G. EMBO J. 2002, 21, 1607–1615. [Google Scholar] [CrossRef]
- Duivelshof, B.L.; Designer, E.; Hernandez-Alba, O.; Ehkirch, A.; Toftevall, H.; Sjögren, J.; Cianferani, S.; Beck, A.; Guillarme, D.; D’Atri, V. Glycan-Mediated Technology for Obtaining Homogeneous Site-Specific Conjugated Antibody–Drug Conjugates: Synthesis and Analytical Characterization by Using Complementary Middle-up LC/HRMS Analysis. Anal. Chem. 2020, 92, 8170–8177. [Google Scholar] [PubMed]
- Chen, B.; Lin, Z.; Zhu, Y.; Jin, Y.; Larson, E.; Xu, Q.; Fu, C.; Zhang, Z.; Zhang, Q.; Pritts, W.A.; Ge, Y. Middle-Down Multi-Attribute Analysis of Antibody-Drug Conjugates with Electron Transfer Dissociation. Anal. Chem. 2019, 91, 11661–11669. [Google Scholar]
- Sang, H.; Lu, G.; Liu, Y.; Hu, Q.; Xing, W.; Cui, D.; Zhou, F.; Zhang, J.; Hao, H.; Wang, G.; Ye, H. Conjugation site analysis of antibody-drug-conjugates (ADCs) by signature ion fingerprinting and normalized area quantitation approach using nano-liquid chromatography coupled to high resolution mass spectrometry. Anal. Chim. Acta 2017, 955, 67–78. [Google Scholar] [CrossRef]
- Chen, J.; Yin, S.; Wu, Y.; Ouyang, J. Development of a Native Nanoelectrospray Mass Spectrometry Method for Determination of the Drug-to-Antibody Ratio of Antibody–Drug Conjugates. Anal. Chem. 2013, 85, 1699–1704. [Google Scholar] [CrossRef]
- Yang, X.; Seol, H.; Lin, W.; Xu, X.; Shen, B.; Qiu, H.; Li, N. Site-Specific Quantitation of Drug Conjugations on Antibody–Drug Conjugates (ADCs) Using a Protease-Assisted Drug Deconjugation and Linker-like Labeling (PADDLL) Method. Anal. Chem. 2021, 93, 9549–9558. [Google Scholar] [CrossRef]
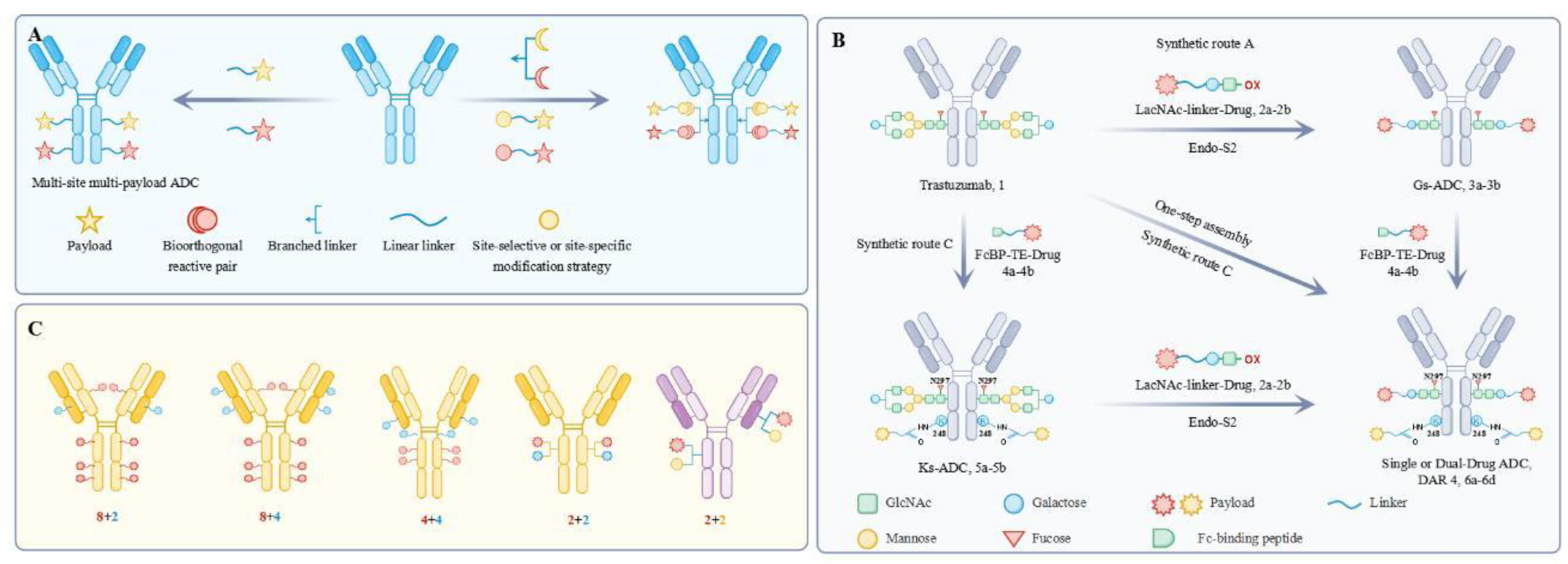
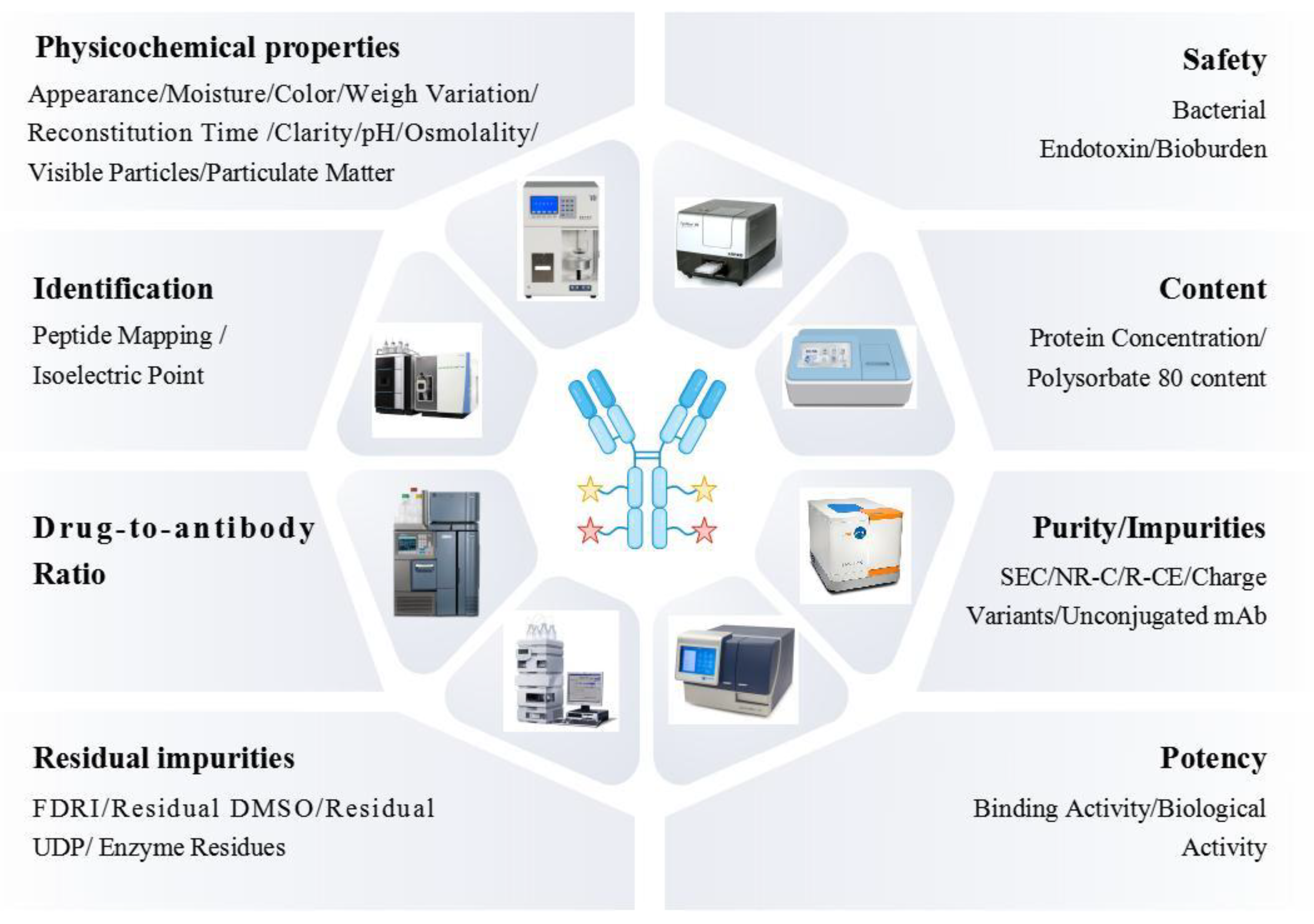
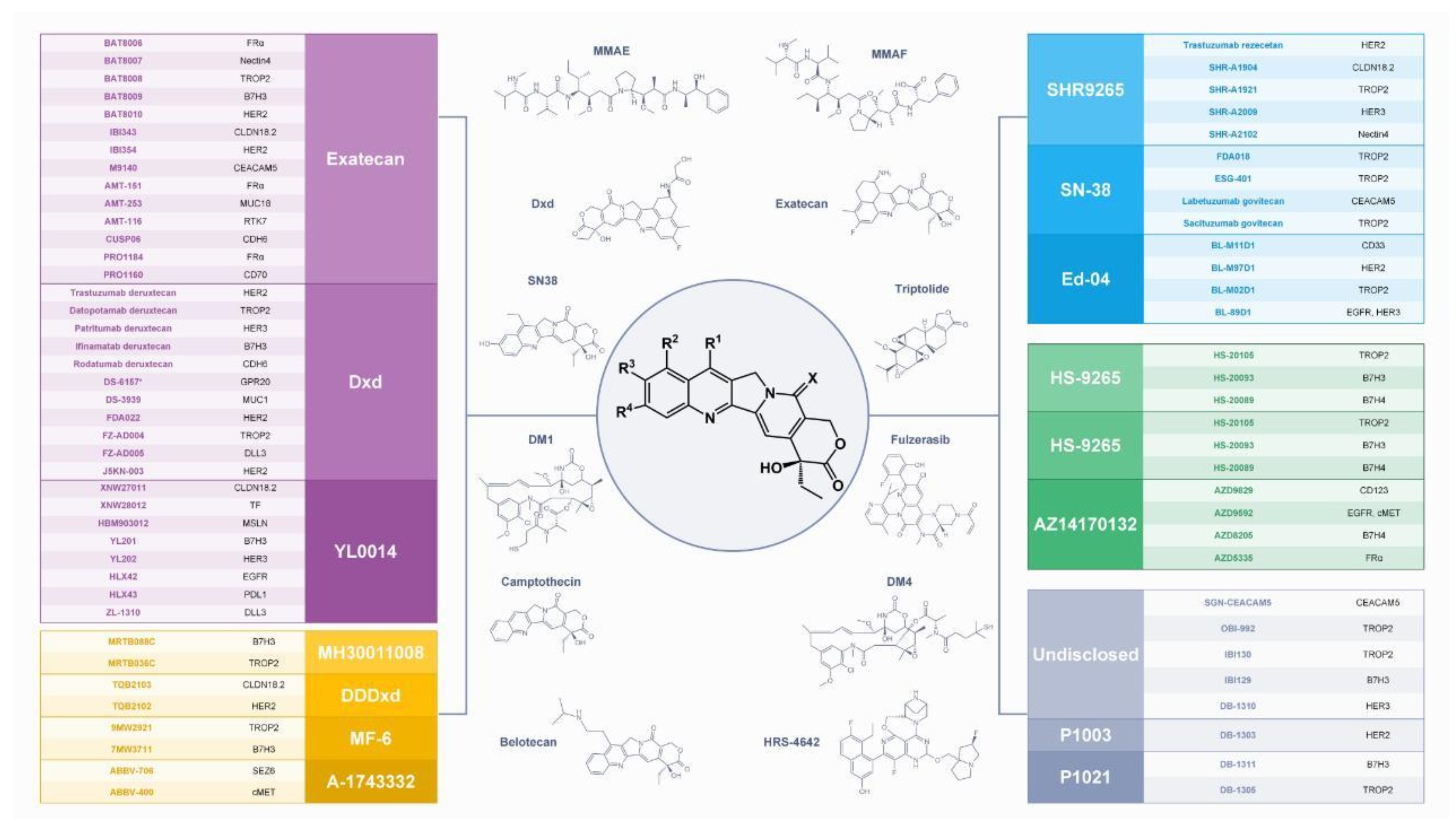
| Code name | Payload | Linker | Linker-conjugation | Antibody | DAR | Reference |
|---|---|---|---|---|---|---|
| MMAE/F ADC | MMAE and MMAF | MC-Cys(SiPr)-Cys(Acm)-PEG24 | Maleimide | Anti-CD30 | 8 + 8 | [1] |
| MMAE/F 4 + 2 ADC, MMAE/F 2 + 2 ADC, MMAE/F 2 + 4 ADC | MMAE and MMAF | DBCO-PEG3-GluValCit-PABC-MMAE, TCO-PEG3-GluValCit-PABC-MMAF | MTGase-mediated transpeptidation | Anti-HER2, Anti-Trop2 | 4 + 2, 2 + 2, 2 + 4 | [2] |
| ADC-6b, ADC-6c | MMAE and MMAF | MMAF-PAB-VC-NH-LacNAc oxazoline, MMAE-PAB-VC-NH-LacNAc oxazoline |
Endo-S2-mediated glycosite-specific, thioester-based K248 site-specific | Anti-HER2 | 2 + 2 | [3] |
| Tmab-VcMMAE-SMCC-DM1 | MMAE and DM1 | MC-VcMMAE, SMCC-DM1 | Maleimide and Lys residues |
Anti-Tmab | 2.83 + 5.25 | [4] |
| 412a-MMAF-SN38 | MMAF and SN38 | MV-PEG8-VC-PABC-SN38 and MC-PEG4-MMAF | Maleimide | Anti-EGFR, anti-cMET | 2.4 + 1.7 | [5] |
| TJ102 | PY-4CAR2 (TOP1i) and PE-E2K (microtubule inhibitor) | Undisclosed | Undisclosed | Anti-CDH6 and anti-FRα | Undisclosed | [6] |
| DXC018 | Antimetabolite inhibitor and TOP1i | Undisclosed | Undisclosed | Anti-HER2 | Undisclosed | [7] |
| JSKN021 | T01 (Alphatecan TOP1i) and MMAE | Undisclosed | Undisclosed | Anti-HER3 and anti-EGFR | 4 + 2 | [8] |
| HER3 dpADC | TOP1i and EGFR TKI | Undisclosed | Enzymatic site-specific | Anti-HER3 | Undisclosed | [9] |
| MMAE (DAR 2) and TOPi (DAR 2+2) ADCs | MMAE/dual TOP1i | Undisclosed | Undisclosed | Anti-Nectin-4 | 2 + 2 + 2 | [10] |
| dpADC | TOP1i and an immune agonist | Undisclosed | Undisclosed | Anti-Trop2 | Undisclosed | [11] |
| LB138 | Two different TOP1i | Undisclosed | Undisclosed | Anti-NaPi2b | 2 + 2 | [12] |
| KH815 | Triptolide and exatecan | DBCO-PEG4-GGFG-Triptolide and MC-PEG2-VC-PABC-Exatecan | Maleimide and glycosite-specific | Anti-Trop2 | 4 + 4 | [13] |
| KHN922 | Triptolide and exatecan | DBCO-PEG4-GGFG-Triptolide and MC-PEG2-VC-PABC-Exatecan | Maleimide and glycosite-specific | Anti-HER3 | 4 + 4 | [14] |
| AD2C | Undisclosed | Undisclosed | Undisclosed | Anti-GPC3 | Undisclosed | [15] |
| CTPH-02 | MMAE and novel payloads | Undisclosed | Undisclosed | Anti-HER2 | Undisclosed | [16] |
| MAb-MMAF/IMQ | MMAF and IMQ | MC-C6-MMAF, phosphine-azide-IMQ | Maleimide and Lys residues | Anti-CD276/B7-H3 | 3–4 + 7–14 | [17] |
| T-DA2(MMAE)-Cys(MMAE) | MMAE, Aldox, VBL, vcDox | MC-PEG3-VCP-payload MC-VCP-payload | Maleimide and cyclopentadiene | Anti-HER2 | 2 or 4 + 1.8–7.1 | [18] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




