Submitted:
26 April 2026
Posted:
28 April 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Healthy Ocular Neurovascular: Architecture and Blood Flow Regulation
Where Do Things Go Wrong in Glaucoma?
Vascular Compromise: Cause or Consequence of Neurodegeneration?
Pathologic Changes in Vascular Structure and Function
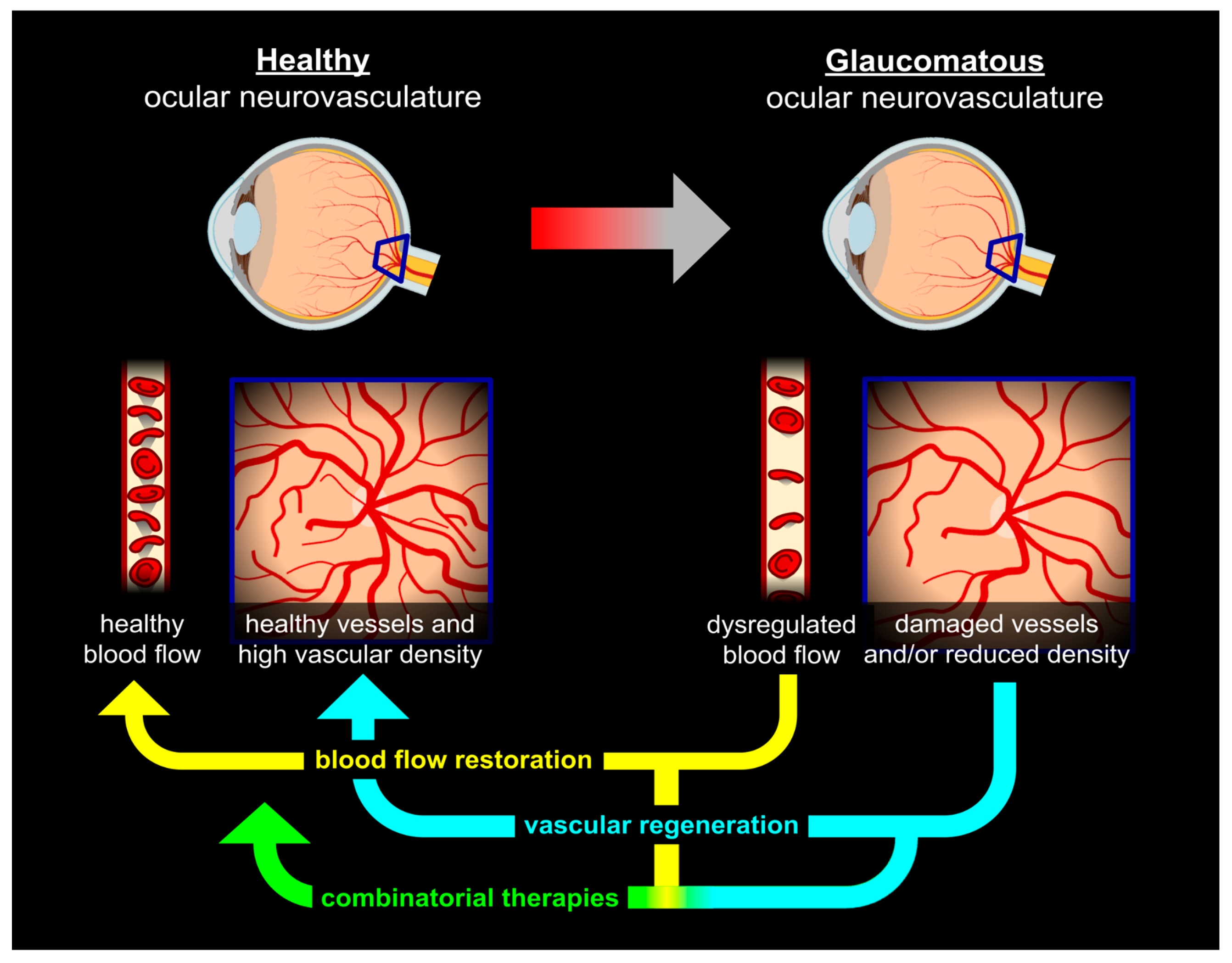
Regenerating Vascular Architecture
Restoring Functional Blood Flow
Conclusions and Future Directions
Funding
References
- Riva, C. E.; Sinclair, S. H.; Grunwald, J. E. Autoregulation of retinal circulation in response to decrease of perfusion pressure. Invest. Ophthalmol. Vis. Sci. 1981, 21, 34–38. [Google Scholar] [PubMed]
- Prada, D.; et al. Autoregulation and neurovascular coupling in the optic nerve head. Surv. Ophthalmol. 2016, 61, 164–186. [Google Scholar] [CrossRef]
- Garhöfer, G.; et al. Diffuse luminance flicker increases blood flow in major retinal arteries and veins. Vis. Res. 2004, 44, 833–838. [Google Scholar] [CrossRef]
- Warner, R. L.; et al. Full-field flicker evoked changes in parafoveal retinal blood flow. Sci. Rep. 2020, 10, 16051. [Google Scholar] [CrossRef]
- Garhöfer, G.; Huemer, K. H.; Zawinka, C.; Schmetterer, L.; Dorner, G. T. Influence of diffuse luminance flicker on choroidal and optic nerve head blood flow. Curr. Eye Res. 2002, 24, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Riva, C. E.; Logean, E.; Falsini, B. Visually evoked hemodynamical response and assessment of neurovascular coupling in the optic nerve and retina. Prog. Retin. Eye Res. 2005, 24, 183–215. [Google Scholar] [CrossRef]
- Mishra, A.; Hamid, A.; Newman, E. A. Oxygen modulation of neurovascular coupling in the retina. Proc. Natl. Acad. Sci. U. S. A. 2011, 108, 17827–17831. [Google Scholar] [CrossRef]
- Roy, C. S.; Sherrington, C. S. On the regulation of the blood-supply of the brain. J. Physiol. 1890, 11, 85–158.17. [Google Scholar] [CrossRef]
- Iadecola, C. The neurovascular unit coming of age: A journey through neurovascular coupling in health and disease. Neuron 2017, 96, 17–42. [Google Scholar] [CrossRef] [PubMed]
- Wareham, L. K.; Calkins, D. J. The neurovascular unit in glaucomatous neurodegeneration. Front. Cell. Dev. Biol. 2020, 8, 452. [Google Scholar] [CrossRef]
- Alarcon-Martinez, L.; et al. Neurovascular dysfunction in glaucoma. Prog. Retin. Eye Res. 2023, 97, 101217. [Google Scholar] [CrossRef]
- Li, M.; et al. Microvascular and cellular dysfunctions in Alzheimer’s disease: an integrative analysis perspective. Sci. Rep. 2024, 14, 20944. [Google Scholar] [CrossRef]
- de la Torre, J. C.; Stefano, G. B. Evidence that Alzheimer’s disease is a microvascular disorder: the role of constitutive nitric oxide. Brain Res. Brain Res. Rev. 2000, 34, 119–136. [Google Scholar] [CrossRef]
- Shi, H.; et al. Retinal vasculopathy in Alzheimer’s disease. Front. Neurosci. 2021, 15, 731614. [Google Scholar] [CrossRef]
- Vautier, A.; et al. Retinal vessels as a window on amyotrophic lateral sclerosis pathophysiology: A systematic review. Rev. Neurol. 2023, 179, 548–562. [Google Scholar] [CrossRef] [PubMed]
- Garbuzova-Davis, S.; et al. Amyotrophic lateral sclerosis: a neurovascular disease. Brain Res. 2011, 1398, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Soldatov, V. O.; Kukharsky, M. S.; Belykh, A. E.; Sobolev, A. M.; Deykin, A. V. Retinal damage in amyotrophic lateral sclerosis: Underlying mechanisms. Eye Brain 2021, 13, 131–146. [Google Scholar] [CrossRef]
- Lin, C.-Y.; et al. Neurovascular abnormalities in humans and mice with Huntington’s disease. Exp. Neurol. 2013, 250, 20–30. [Google Scholar] [CrossRef]
- Garcia, F. J.; et al. Single-cell dissection of the human brain vasculature. Nature 2022, 603, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Amini, E.; et al. Huntington’s disease and neurovascular structure of retina. Neurol. Sci. 2022, 43, 5933–5941. [Google Scholar] [CrossRef]
- Pitha, I.; Du, L.; Nguyen, T. D.; Quigley, H. IOP and glaucoma damage: The essential role of optic nerve head and retinal mechanosensors. Prog. Retin. Eye Res. 2024, 99, 101232. [Google Scholar] [CrossRef]
- Stowell, C.; Burgoyne, C. F.; Tamm, E. R.; Ethier, C. R. Lasker/IRRF Initiative on Astrocytes and Glaucomatous Neurodegeneration Participants. Biomechanical aspects of axonal damage in glaucoma: A brief review. Exp. Eye Res. 2017, 157, 13–19. [Google Scholar] [CrossRef]
- Sigal, I. A.; Ethier, C. R. Biomechanics of the optic nerve head. Exp. Eye Res. 2009, 88, 799–807. [Google Scholar] [CrossRef]
- Quigley, H. A.; Addicks, E. M.; Green, W. R.; Maumenee, A. E. Optic nerve damage in human glaucoma. II. The site of injury and susceptibility to damage: II. The site of injury and susceptibility to damage. Arch. Ophthalmol. 1981, 99, 635–649. [Google Scholar] [CrossRef]
- Asrani, S. G.; et al. The relationship between intraocular pressure and glaucoma: An evolving concept. Prog. Retin. Eye Res. 2024, 103, 101303. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y. I.; et al. Longitudinal evaluation of advanced glaucoma: ten year follow-up cohort study. Sci. Rep. 2024, 14, 476. [Google Scholar] [CrossRef]
- Leske, M. C.; et al. Factors for glaucoma progression and the effect of treatment: the early manifest glaucoma trial: The early manifest glaucoma trial. Arch. Ophthalmol. 2003, 121, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Killer, H. E.; Pircher, A. Normal tension glaucoma: review of current understanding and mechanisms of the pathogenesis. Eye (Lond.) 2018, 32, 924–930. [Google Scholar] [CrossRef] [PubMed]
- Pitale, P. M.; et al. Selective vulnerability of the intermediate retinal capillary plexus precedes retinal ganglion cell loss in ocular hypertension. Front. Cell. Neurosci. 2022, 16, 1073786. [Google Scholar] [CrossRef]
- Waxman, S.; et al. Pericytes in the optic nerve head. Prog. Retin. Eye Res. 2025, 107, 101375. [Google Scholar] [CrossRef]
- Alarcon-Martinez, L.; et al. Pericyte dysfunction and loss of interpericyte tunneling nanotubes promote neurovascular deficits in glaucoma. Proc. Natl. Acad. Sci. U. S. A. 2022, 119. [Google Scholar] [CrossRef]
- Lavery, W. J.; Muir, E. R.; Kiel, J. W.; Duong, T. Q. Magnetic resonance imaging indicates decreased choroidal and retinal blood flow in the DBA/2J mouse model of glaucoma. Invest. Ophthalmol. Vis. Sci. 2012, 53, 560–564. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; et al. Microglia drive peripapillary vascular density reduction in normal tension glaucoma by regulating the Rpl17/Stat5b/Apoa1 axis. Adv. Sci. (Weinh.) 2025, 12, e07894. [Google Scholar] [CrossRef]
- Dunn, M.; et al. Microvascular volume loss exceeds nerve fiber layer but not neuroretinal rim tissue loss during progression of nonhuman primate experimental glaucoma. Invest. Ophthalmol. Vis. Sci. 2026, 67, 24. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; et al. Associations of retinal microvascular density and fractal dimension with glaucoma: A prospective study from UK Biobank. Ophthalmol. Sci. 2025, 5, 100661. [Google Scholar] [CrossRef]
- Yarmohammadi, A.; et al. Relationship between optical coherence tomography angiography vessel density and severity of visual field loss in glaucoma. Ophthalmology 2016, 123, 2498–2508. [Google Scholar] [CrossRef]
- Hou, H.; et al. Ganglion cell complex thickness and macular vessel density loss in primary open-angle glaucoma. Ophthalmology 2020, 127, 1043–1052. [Google Scholar] [CrossRef]
- Wu, J.-H.; et al. Longitudinal OCTA vessel density loss in macula and optic nerve head in healthy, glaucoma suspect and established glaucoma eyes. Br. J. Ophthalmol. 2025, 109, 490–496. [Google Scholar] [CrossRef] [PubMed]
- Sugiyama, T. Basic technology and clinical applications of the updated model of laser speckle flowgraphy to ocular diseases. Photonics 2014, 1, 220–234. [Google Scholar] [CrossRef]
- Gu, C.; Li, A.; Yu, L. Diagnostic performance of laser speckle flowgraphy in glaucoma: a systematic review and meta-analysis. Int. Ophthalmol. 2021, 41, 3877–3888. [Google Scholar] [CrossRef]
- Mursch-Edlmayr, A. S.; et al. Laser speckle flowgraphy derived characteristics of optic nerve head perfusion in normal tension glaucoma and healthy individuals: a Pilot study. Sci. Rep. 2018, 8, 5343. [Google Scholar] [CrossRef]
- Shiga, Y.; et al. Optic nerve head blood flow, as measured by laser speckle flowgraphy, is significantly reduced in preperimetric glaucoma. Curr. Eye Res. 2016, 41, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, F.; Iwase, T.; Yamamoto, K.; Ra, E.; Terasaki, H. Correlation between blood flow on optic nerve head and structural and functional changes in eyes with glaucoma. Sci. Rep. 2020, 10, 729. [Google Scholar] [CrossRef]
- Hayreh, S. S. The blood supply of the optic nerve head and the evaluation of it - myth and reality. Prog. Retin. Eye Res. 2001, 20, 563–593. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D. R.; Braverman, S. Reevaluation of the optic disk vasculature. Am. J. Ophthalmol. 1976, 82, 165–174. [Google Scholar] [CrossRef]
- Lieberman, M. F.; Maumenee, A. E.; Green, W. R. Histologic studies of the vasculature of the anterior optic nerve. Am. J. Ophthalmol. 1976, 82, 405–423. [Google Scholar] [CrossRef]
- Mackenzie, P. J.; Cioffi, G. A. Vascular anatomy of the optic nerve head. Can. J. Ophthalmol. 2008, 43, 308–312. [Google Scholar] [CrossRef]
- Kang, M. H.; et al. Microvascular density is associated with retinal ganglion cell axonal volume in the laminar compartments of the human optic nerve head. Invest. Ophthalmol. Vis. Sci. 2018, 59, 1562–1570. [Google Scholar] [CrossRef]
- Waxman, S.; et al. Lamina cribrosa vessel and collagen beam networks are distinct. Exp. Eye Res. 2022, 215, 108916. [Google Scholar] [CrossRef]
- Lee, P.-Y.; et al. A workflow for three-dimensional reconstruction and quantification of the monkey optic nerve head vascular network. J. Biomech. Eng. 2022, 144. [Google Scholar]
- Lu, Y.; et al. The robust lamina cribrosa vasculature: Perfusion and oxygenation under elevated intraocular pressure. Invest. Ophthalmol. Vis. Sci. 2024, 65, 1. [Google Scholar] [CrossRef]
- Waxman, S.; et al. High-resolution, three-dimensional reconstruction of the outflow tract demonstrates segmental differences in cleared eyes. Invest. Ophthalmol. Vis. Sci. 2018, 59, 2371–2380. [Google Scholar] [CrossRef]
- Darche, M.; et al. Three-dimensional characterization of developing and adult ocular vasculature in mice using in toto clearing. Commun. Biol. 2022, 5, 1135. [Google Scholar] [CrossRef] [PubMed]
- Darche, M.; et al. Light sheet fluorescence microscopy of cleared human eyes. Commun. Biol. 2023, 6, 1025. [Google Scholar] [CrossRef]
- Krasniqi, D.; Richardson, D. S.; Jakobs, T.; Rizzo, J. F. Light sheet fluorescence microscopy of cleared human orbital arteries. Invest. Ophthalmol. Vis. Sci. 2024, 65, PB0046–PB0046. [Google Scholar]
- Waxman, S.; Linton, A.; Di Polo, A.; Fortune, B.; Sigal, I. A. High-resolution 3D imaging of the optically cleared ONH reveals the intact structure of collagen and vascular networks. Invest. Ophthalmol. Vis. Sci. 2025, 66, PP009–PP009. [Google Scholar]
- Williamson, M. R.; et al. A window of vascular plasticity coupled to behavioral recovery after stroke. J. Neurosci. 2020, 40, 7651–7667. [Google Scholar] [CrossRef]
- Lapi, D.; Colantuoni, A. Remodeling of cerebral microcirculation after ischemia-reperfusion. J. Vasc. Res. 2015, 52, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Fawcett, J. W. The struggle to make CNS axons regenerate: Why has it been so difficult? Neurochem. Res. 2020, 45, 144–158. [Google Scholar] [CrossRef]
- Lee, C.; et al. Vascular endothelial growth factor signaling in health and disease: from molecular mechanisms to therapeutic perspectives. Signal Transduct. Target. Ther. 2025, 10, 170. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Zechariah, A.; Qu, Y.; Hermann, D. M. Effects of vascular endothelial growth factor in ischemic stroke. J. Neurosci. Res. 2012, 90, 1873–1882. [Google Scholar] [CrossRef] [PubMed]
- Foxton, R. H.; et al. VEGF-A is necessary and sufficient for retinal neuroprotection in models of experimental glaucoma. Am. J. Pathol. 2013, 182, 1379–1390. [Google Scholar] [CrossRef] [PubMed]
- Jin, K. L.; Mao, X. O.; Greenberg, D. A. Vascular endothelial growth factor: direct neuroprotective effect in in vitro ischemia. Proc. Natl. Acad. Sci. U. S. A. 2000, 97, 10242–10247. [Google Scholar] [CrossRef]
- Wang, D.-P.; et al. Neuroprotective effects of VEGF-A nanofiber membrane and FAAH inhibitor URB597 against oxygen-glucose deprivation-induced ischemic neuronal injury. Int. J. Nanomed. 2021, 16, 3661–3678. [Google Scholar] [CrossRef]
- Kilic, U.; et al. Human vascular endothelial growth factor protects axotomized retinal ganglion cells in vivo by activating ERK-1/2 and Akt pathways. J. Neurosci. 2006, 26, 12439–12446. [Google Scholar] [CrossRef]
- Tripathi, R. C.; Li, J.; Tripathi, B. J.; Chalam, K. V.; Adamis, A. P. Increased level of vascular endothelial growth factor in aqueous humor of patients with neovascular glaucoma. Ophthalmology 1998, 105, 232–237. [Google Scholar] [CrossRef]
- Tolentino, M. J.; et al. Vascular endothelial growth factor is sufficient to produce iris neovascularization and neovascular glaucoma in a nonhuman primate. Arch. Ophthalmol. 1996, 114, 964–970. [Google Scholar] [CrossRef]
- Moshfeghi, A. A.; et al. Impact of anti-VEGF treatment on development of proliferative diabetic retinopathy in routine clinical practice. BMC Ophthalmol. 2024, 24, 229. [Google Scholar] [CrossRef] [PubMed]
- Stiver, S. I.; Tan, X.; Brown, L. F.; Hedley-Whyte, E. T.; Dvorak, H. F. VEGF-A angiogenesis induces a stable neovasculature in adult murine brain. J. Neuropathol. Exp. Neurol. 2004, 63, 841–855. [Google Scholar] [CrossRef]
- Beheshtizadeh, N.; et al. Vascular endothelial growth factor (VEGF) delivery approaches in regenerative medicine. Biomed. Pharmacother. 2023, 166, 115301. [Google Scholar] [CrossRef]
- Gerhardt, H. VEGF and endothelial guidance in angiogenic sprouting. Organogenesis 2008, 4, 241–246. [Google Scholar] [CrossRef] [PubMed]
- Hellström, M.; Kalén, M.; Lindahl, P.; Abramsson, A.; Betsholtz, C. Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 1999, 126, 3047–3055. [Google Scholar] [CrossRef]
- Hellberg, C.; Ostman, A.; Heldin, C.-H. PDGF and vessel maturation. Recent Results Cancer Res. 2010, 180, 103–114. [Google Scholar]
- Liu, Y.; et al. Sequential release of vascular endothelial growth factor and platelet-derived growth factor at the appropriate time for improved angiogenesis. Acta Biomater. 2025, 196, 171–181. [Google Scholar] [CrossRef]
- Zhang, Z.; Warner, K. A.; Mantesso, A.; Nör, J. E. PDGF-BB signaling via PDGFR-β regulates the maturation of blood vessels generated upon vasculogenic differentiation of dental pulp stem cells. Front. Cell. Dev. Biol. 2022, 10, 977725. [Google Scholar] [CrossRef]
- Li, S. Y.; Johnson, R.; Smyth, L. C.; Dragunow, M. Platelet-derived growth factor signalling in neurovascular function and disease. Int. J. Biochem. Cell. Biol. 2022, 145, 106187. [Google Scholar] [CrossRef]
- Andrae, J.; Gallini, R.; Betsholtz, C. Role of platelet-derived growth factors in physiology and medicine. Genes. Dev. 2008, 22, 1276–1312. [Google Scholar] [CrossRef]
- Brudno, Y.; Ennett-Shepard, A. B.; Chen, R. R.; Aizenberg, M.; Mooney, D. J. Enhancing microvascular formation and vessel maturation through temporal control over multiple pro-angiogenic and pro-maturation factors. Biomaterials 2013, 34, 9201–9209. [Google Scholar] [CrossRef]
- Uemura, A.; et al. Recombinant angiopoietin-1 restores higher-order architecture of growing blood vessels in mice in the absence of mural cells. J. Clin. Invest. 2002, 110, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Gavard, J.; Patel, V.; Gutkind, J. S. Angiopoietin-1 prevents VEGF-induced endothelial permeability by sequestering Src through mDia. Dev. Cell. 2008, 14, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Wang, L.; Hu, A.; Zhang, S. Mechanisms of astrocyte action in the blood brain barrier: From structural support to dynamic regulation. J. Integr. Neurosci. 2025, 24, 45223. [Google Scholar] [CrossRef]
- Bilal, A.; Constantin, F.; Chirila, S.; Hangan, T. New trends in the treatment of open-angle glaucoma: a critical review. Int. Ophthalmol. 2025, 45, 381. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, M.; Xin, C.; Martin, E.; Wang, R. Trabecular meshwork movement controls distal valves and chambers: New glaucoma medical and surgical targets. J. Clin. Med. 2023, 12, 6599. [Google Scholar] [CrossRef]
- Kizhatil, K.; Ryan, M.; Marchant, J. K.; Henrich, S.; John, S. W. M. Schlemm’s canal is a unique vessel with a combination of blood vascular and lymphatic phenotypes that forms by a novel developmental process. PLoS Biol. 2014, 12, e1001912. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Cull, G. A.; Piper, C.; Burgoyne, C. F.; Fortune, B. Anterior and posterior optic nerve head blood flow in nonhuman primate experimental glaucoma model measured by laser speckle imaging technique and microsphere method. Invest. Ophthalmol. Vis. Sci. 2012, 53, 8303–8309. [Google Scholar] [CrossRef] [PubMed]
- Iwase, T.; Akahori, T.; Yamamoto, K.; Ra, E.; Terasaki, H. Evaluation of optic nerve head blood flow in response to increase of intraocular pressure. Sci. Rep. 2018, 8, 17235. [Google Scholar] [CrossRef]
- Kiyota, N.; et al. The impact of intraocular pressure elevation on optic nerve head and choroidal blood flow. Invest. Ophthalmol. Vis. Sci. 2018, 59, 3488–3496. [Google Scholar] [CrossRef]
- Gherghel, D.; Hosking, S. L.; Cunliffe, I. A.; Armstrong, R. A. First-line therapy with latanoprost 0.005% results in improved ocular circulation in newly diagnosed primary open-angle glaucoma patients: a prospective, 6-month, open-label study. Eye (Lond.) 2008, 22, 363–369. [Google Scholar] [CrossRef]
- Iida, Y.; et al. Retinal blood flow velocity change in parafoveal capillary after topical tafluprost treatment in eyes with primary open-angle glaucoma. Sci. Rep. 2017, 7, 5019. [Google Scholar] [CrossRef]
- Schallek, J.; Geng, Y.; Nguyen, H.; Williams, D. R. Morphology and topography of retinal pericytes in the living mouse retina using in vivo adaptive optics imaging and ex vivo characterization. Invest. Ophthalmol. Vis. Sci. 2013, 54, 8237–8250. [Google Scholar] [CrossRef]
- Rouget, C. Memoire sur le develloppment, la structure et les propietes physiologiques des capillaries senguins et lymphatiques. Arch. Physiol. Norm. Pathol. 1873, 5, 603–663. [Google Scholar]
- Trost, A.; Bruckner, D.; Rivera, F. J.; Reitsamer, H. A. Pericytes in the retina. Adv. Exp. Med. Biol. 2019, 1122, 1–26. [Google Scholar]
- Adamiec-Mroczek, J.; Oficjalska-Młyńczak, J.; Misiuk-Hojło, M. Roles of endothelin-1 and selected proinflammatory cytokines in the pathogenesis of proliferative diabetic retinopathy: Analysis of vitreous samples. Cytokine 2010, 49, 269–274. [Google Scholar] [CrossRef]
- He, S.; Prasanna, G.; Yorio, T. Endothelin-1-mediated signaling in the expression of matrix metalloproteinases and tissue inhibitors of metalloproteinases in astrocytes. Invest. Ophthalmol. Vis. Sci. 2007, 48, 3737–3745. [Google Scholar] [CrossRef]
- Mansberger, S. L.; et al. PER-001, an endothelin antagonist, increased optic nerve head blood flow with structural and functional improvements in patients with glaucoma. Invest. Ophthalmol. Vis. Sci. 2025, 66, 3322–3322. [Google Scholar]
- Gray, D. Perfuse Therapeutics Announces Positive Results from Phase 2 Clinical Trials in Glaucoma and Diabetic Retinopathy Patients. Perfuse Therapeutics. 2025. Available online: https://perfusetherapeutics.com/perfuse-therapeutics-announces-positive-results-from-phase-2-clinical-trials-in-glaucoma-and-diabetic-retinopathy-patients/.
- Rao, V. P.; Epstein, D. L. Rho GTPase/Rho kinase inhibition as a novel target for the treatment of glaucoma. BioDrugs 2007, 21, 167–177. [Google Scholar] [CrossRef]
- Reboussin, É.; et al. Evaluation of Rho kinase inhibitor effects on neuroprotection and neuroinflammation in an ex-vivo retinal explant model. Acta Neuropathol. Commun. 2024, 12, 150. [Google Scholar] [CrossRef]
- Wang, J.; Wang, H.; Dang, Y. Rho-kinase inhibitors as emerging targets for glaucoma therapy. Ophthalmol. Ther. 2023, 12, 2943–2957. [Google Scholar] [CrossRef]
- Lonfat, N.; Moreno-Leon, L.; Punzo, C.; Khanna, H. Update on gene therapy clinical trials for eye diseases. Hum. Gene Ther. 2025, 36, 1287–1300. [Google Scholar] [CrossRef] [PubMed]
- Butt, F. R.; et al. Gene therapy for inherited retinal disease: Current strategies, personalized medicine, and future implications-A comprehensive review. J. Pers. Med. 2025, 15, 619. [Google Scholar] [CrossRef] [PubMed]
- Reid, C. A.; Nettesheim, E. R.; Connor, T. B.; Lipinski, D. M. Development of an inducible anti-VEGF rAAV gene therapy strategy for the treatment of wet AMD. Sci. Rep. 2018, 8, 11763. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Available online: https://clinicaltrials.gov/study/NCT06856577.
- Abbas, F.; et al. Revival of light signalling in the postmortem mouse and human retina. Nature 2022, 606, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J. W.; Chan, D.; Sivak, J. M. A novel ex-vivo model for studying early pressure-induced retinal and optic nerve head responses in the human eye. Invest. Ophthalmol. Vis. Sci. 2022, 63, 923–A0392. [Google Scholar]
- Chan, D.; Zhang, J. W.; Won, G.-J.; Sivak, J. M. Retinal cytoarchitecture is preserved in an organotypic perfused human and porcine eye model. Acta Neuropathol. Commun. 2024, 12, 186. [Google Scholar] [CrossRef]
- Lohss, M.; et al. A mobile system for whole eye perfusion supporting retinal function and surgery. Front. Bioeng. Biotechnol. 2025, 13, 1699876. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



